Issue 33 November 2012
Worcestershire Record | 33 (Nov 2012) page: 27-29 | Worcestershire Biological Records Centre & Worcestershire Recorders
Moth Recording in the Teme Valley 2012
Danny Arnold
Teme Valley Wildlife Group
Birchfield is an 18 acre smallholding in the Teme Valley that I am fortunate enough to call home. Not used for making a living, it has become a personal mini wildlife reserve. Almost every night when I’m here throughout the year, I run at least one moth trap in a fixed location, which is supplemented at least once a week by four further traps in four other fixed locations for nine months of the year. So typically having run circa 450 – 500 moth light traps per year for the past five years or so I guess it could quite legitimately be termed, a “constant effort” site, at least for moth data.
And the Teme Valley is proving to be one Worcestershire’s moth ‘hot spots’ owing much to the exceptional quality and diversity of habitat found in the area. Much of the habitat is pristine arising in part from the difficulty of the terrain for farming the land. On the Valley floor and on the Valley sides fields tend to be small and often undulating, a resulting legacy of moraines produced from the receding glaciers of the last ice age. This has tended to restrict arable crops in favour of livestock farming, typically sheep. As such, good hedges are required to contain livestock, which of course, makes for good connectivity corridors for all sorts of wildlife throughout the Valley.
With a good number of springs and water courses coming off the higher ground, dingles have been hewn into the Valley slopes at irregular intervals. These are often deep and relatively inaccessible places resulting in farms fencing off these areas in a bid to keep livestock out. Often left to their own devices, these dingles are relatively unmanaged and somewhat neglected tracts of land that evolve almost ad-hoc, further adding to the biodiversity found in the area. Over the years, they become very important mini ecosystems in their own right, supporting a wide range of often quite specialised flora and fauna.
The ecology of dingles in the area is complex and scantily documented. They are unique in that they cover a full spectrum of changing habitat, turning from dried up gullies in the height of summer to damp, mossy, shaded ravines trickling with spring water, to raging torrents of water during a flash floods, carrying vast quantities of scouring water down into the waiting out-flow into the River Teme below.
It is this tremendous diversity of habitat within the Teme Valley which over the past five years or so has produced almost 800 species of moth, including seven “first records for Worcestershire”. And indeed, every year, sees new species added to the ever increasing area list, that covers the predominantly SO66 Ordnance Survey hectad (10×10 km square)
The latest ‘first record for Worcestershire’ fell to Ken Willetts living on the Highwood at Eastham. Not even having completed his first ever year of moth trapping, his new addiction to the world of moths had him sweep netting his garden flowers for even the smallest quarry. One such specimen caught his attention owing to its striking colouration. Ken forwarded me a photograph of the specimen which I presumed to be a possible new record for Worcestershire 902 Chrysoclista lathamella (Figs. 1& 2). This was later confirmed as such by County Moth recorder Tony Simpson.
Other relative rarities turning up in the Teme Valley this year have been 887 Mompha lacteella, (confirmed by dissection) 463 Ypsolopha vittella and 1272 Pammene aurana all taken by Ken Willetts, confirmed Tony Simpson.
Also 469 Eidophasia messingiella , 891 Mompha sturnipennella (Fig 3), 280 Caloptilia cuculipennella (Fig. 4.) and 515 Coleophora albitarsella have all turned up for the Author, all being confirmed by dissection.
Also of note is the seemingly increasing occurrence of 289 Caloptilia falconipennella (Fig. 5.), unknown in Worcestershire pre 2009, it is now appearing with increased regularity in the moth traps of the Teme Valley. The larva of this species is an Alder feeder, so, with plenty of Alder in the Teme Valley, local habitat profile fits well with this species presence.
But even with these encouraging signs of hitherto non recorded moth species in the Teme Valley, on the Moth front generally the summer of 2012 has been a poor one for Lepidoptera, reflecting the general status nationally. Moth numbers are down. Species numbers recorded are down. Looking at the data from Birchfield over the summer months from the past five years Graph 1 and 2 (Figs. 6 & 7) clearly outline just how poor a year it has been for moths.
Graph 1 (Fig. 6.) shows the number of spring and summer moth species trapped each month this year (2012), against the average number of moth species trapped over the past four years for the same monthly periods.
This clearly shows a marked decline in the number of species being seen and recorded this year from the Teme Valley. (And if the figures are further drilled down into, but not shown here, it can be seen there is even an apparent general downward trend almost every year over the past five years, indicating this is a continuing trend with 2012 being the worst year in a set of five).
With the exception of a slight recovery in June species numbers recorded would appear to be down around 50% per month, based on monthly average numbers over the previous four years.
Similar occurrences are also apparent with moth numbers trapped as is seen from Graph 2 (Fig. 7.). There is clearly a significant decline in the volume of moths around as seen from the perspective of the past five years.
Obviously a five year period is only a tiny reflection of a much bigger picture, but underlying trends are at this point undeniable. Lepidoptera populations are in trouble.
The inspiring rewards of the effort of finding new species in the Teme Valley area is contrasted by the downward trend in general species recorded and numbers.
There are no easy answers or quick fixes to the issues. We believe we ‘manage’ our 18 acres with our sights set firmly on wildlife prosperity but even with that focus we appear to be falling short.
Mini studies such as this, with concentrated monitoring and data production from a site such as this, clearly helps to demonstrate trends in the Teme Valley area and may possibly even reflect trends further afield on an even greater scale.
Images
 Fig. 1. 902 Chrysoclista lathamella. Ken Willetts
Fig. 1. 902 Chrysoclista lathamella. Ken Willetts
 Fig. 2. 902 Chrysoclista lathamella. Ken Willetts
Fig. 2. 902 Chrysoclista lathamella. Ken Willetts
 Fig. 3. 891 Mompha sturnipennella. Danny Arnold
Fig. 3. 891 Mompha sturnipennella. Danny Arnold
 Fig. 4. 280 Caloptilia cuculipennella. Danny Arnold
Fig. 4. 280 Caloptilia cuculipennella. Danny Arnold
 Fig. 5. 289 Caloptilia falconipennella. Danny Arnold
Fig. 5. 289 Caloptilia falconipennella. Danny Arnold
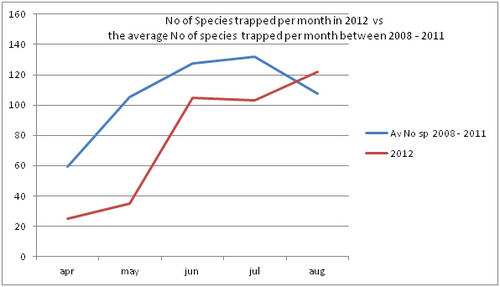 Fig. 6. Graph 1 number of moth species per month
Fig. 6. Graph 1 number of moth species per month
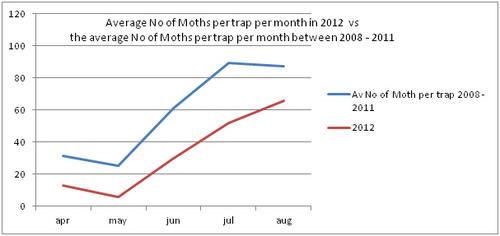 Fig. 7. Graph 2 number of moths per month
Fig. 7. Graph 2 number of moths per month
Worcestershire Record | 33 (Nov 2012) page: 55-57 | Worcestershire Biological Records Centre & Worcestershire Recorders
An account of one Swift nest in Kidderminster
Mike Averill
This is not an in-depth study, but observations of a Swift nest assisted by the installation of a camera. In 2010 a box designed for Swift use (see Swift Conservation web site for information) was finally occupied after being in situ for about seven years. It was fitted on a building in an area where Swifts are present each year and there are usually two other nests within 100 metres of the house. The box is on the east side of the house with the access hole facing down the road to the south. The box is under the eaves and so is not in direct sunshine. The construction is as per normal recommendations but the entrance was fitted at the end purely because it meant that it would be visible to Swifts flying up the road. In the seven years the box had been up there were only a few occasions when it was inspected by Swifts but no Swifts were seen to enter until 2010. Occupation was first noticed in mid July 2010 and at least one chick was seen to fledge that year with a further egg being found later un-hatched. Swifts are hard to observe at nest sites because the adults do not go in and out very often during the day and so a camera is a must. In 2010 a camera was fitted outside the box trained on the nest entrance and this helped to some extent but it was still impossible to tell what was going on inside. With that in mind, in readiness for the 2011 season an infrared camera was fitted inside the box: the sort commonly used in tit nestboxes. Unfortunately the Swifts did not return to the box in 2011.
Interestingly in 2010 the Swifts actually evicted nesting House Sparrows to use the box and in 2012 Sparrows were once again trying to use the box and so in late April the entrance slot was reduced in size to prevent this. To further encourage the use of the box in 2012, a Swift call recording obtained from Swift Conservation was played during early May. This call consists of the screams of Swifts which already have a nest site and is supposed to stimulate other prospective Swifts to investigate. Whether this worked or not it was with great relief that two Swifts were seen to enter the box on 22nd May 2012 and stay in there all morning. Pairing appeared to take place during that time. Prior to that Swifts had been seen in the area from 31st April 2012.
Table 1 shows the observations during the nesting period which produced one fledged nestling. It was a very prolonged period and something may have happened to interrupt the process as there were signs of some disturbance on the 11th June when nesting material was heaved up, and no birds stayed that night which wass the first time that two Swifts had not roosted in the box since the 22nd May. Unfortunately the box wasn’t observed again until the 20th June but the Swifts had returned and they were occupying the box at night. It was a strange year of course weather wise with prolonged cool wet periods and it is easy to speculate that poor conditions had stalled the nesting process. Whatever went on, the routine overnight occupation of the box by two Swifts was observed uninterrupted from the 20th June to the 28th August. It was difficult to see exactly when the egg was laid as the camera angle was not perfect so it was not until the 1st August that the first glimpse of a chick was made. The following 23 days were spent selfishly by the single chick eagerly greeting the adults who returned with foodballs of flies, making a fuss rather like young mammals do on the return of adults with food. Internal cleanliness was good with the adults taking droppings away and the chick reversing kingfisher style to eject faecal material out of the box hole: another confirmation that the end access hole seems to work well. One interesting fact revealed by the camera was that two Swifts used the box each night. Very often it is assumed that the males do not have much contact with the nest site other than bringing food. Assuming that the two in the box were a pair, both male and female had spells brooding the egg. Pair bonding was reinforced by the one sitting on the nest calling loudly if the other one was out collecting food. As soon as the chick had hatched both adults would go out feeding during the day.
| 22/5/2012 | First attempt to enter nest box today. Many entries and two birds were in the box at dusk. Mating appeared to take place. |
| 23/5/2012 | Both birds out all day returning at 21.08 to stay all night |
| 24/5/2012 | Screaming from box at 10:34. Both Swifts in at 21.09 |
| 25/5/2012 | Two birds in nest box at night |
| 26/5/2012 | Two birds in nest box at night |
| 27/5/2012 | Two birds in nest box at 21:00 |
| 28/5/2012 | Two birds in nest box at 08:30 and 22:00 |
| 29/5/2012 | Both birds in and out several times finally entering at 21:15 |
| 30/5/2012 | Two birds in nest box at 21:15 |
| 31/5/2012- 10/6/2012 | Two birds in nest box at night |
| 11/6/2012 | No birds in box overnight |
| 12/6-20/6 | Some sort of disturbance in the box with nesting material being heaved up. Nest was not observed again until 20/6 |
| 20/6/2012 | Two birds screaming from box |
| 21/6/2012 | Two birds in box for night at 18:11 |
| 22/6/2012 | Two birds in box for night at 21:25 |
| 23/6/2012 | Two birds in box for night at 21:30 |
| 24/6/2012 -27/6/2012 | Two birds in box for night |
| 5/7/2012 | Two birds in box for night, one stayed all day |
| 14/7/2012-1/8/2012 | At least one bird sitting all day. Couldn’t see it but an egg had been laid |
| 1/8/2012 | First glimpse of one chick |
| 6/8/2012-20/8/2012 | Chick becoming more feathered, food balls being brought and adults avidly greeted by chick. Wing stretching and press ups by the chick. |
| 20/8/2012 | Chick fully feathered, adults didn’t come back to box at night. Nestling doing press up exercises regularly. |
| 23/8/2012 | No adults returned since 20/8/2012, juvenile climbed out of the box at 18:15, then climbed back in. Finally bailed out at 21:12 and the louse fly jumped off the chick to stay in the box. |
Table. 1. Record of events in the Swift nestbox in 2012.
The Swift Louse Fly Crataerina pallida was observed during the time that Swifts used the box and seemed to show up quite well when the infrared camera was on. When the adult Swifts were in the box for the night the louse flies were quite active during preening. There appeared to be a definite attempt by the louse fly to sit behind the birds head whilst it dealt with its feathers and would dodge from one side to another of the neck as the bird’s head reached around. What was puzzling was why one bird could not deal with the louse fly on its mate as that appeared to be possible as they sat side by side. It is interesting to speculate that perhaps the Swift does not make too much attempt to remove the fly and research has in fact not as yet found any detrimental effect from having the parasites on them.
Recent studies by Walker & Rotherham (2010) on a complex of Swift roosts in a German bridge have found that the mean parasitic load was between one and seven per nest and that louse fly numbers declined throughout the Swift breeding season. Parasite populations were heavily female biased, except for at the initial and final stages of the nestling period. It is in the interests of the fly not to overburden the Swift but they feed every five days with males taking on average 23 mg and females 38 mg of blood on each occasion (Kemper, 1951); this has been calculated as being the equivalent to 5% of an adult Swifts total blood volume.
An interesting feature of this insect is that it does not lay an egg but broods internally before laying a fully formed larva that immediately starts to pupate. When the pupa hatches into an adult it seeks to associate itself with a bird or seeks a nest of young Swifts. Louse flies are of course vertically transmitted ectoparasites only passing on their young to the same family of Swifts and so the relationship is very close. Observing the Swifts in the nest box it was clear that the louse flies stayed on the adult Swifts as they came and went and there didn’t appear to be more than one per adult. It was difficult to be sure of that though as they completely disappeared under feathers but what can be said is that no more than one was ever visible on the adults at any one time.
The literature implies that the pupated louse flies associate themselves with the young chicks as soon as they hatch but some must take up residence on the adults. Doing this they will spend many hours hurtling at up to 70 m.p.h. as the adults search for food and their flat bodies seem well adapted for this. What was most interesting was that the final minutes before the juvenile Swift left the box were observed and when the Swift bailed out for its maiden flight the louse fly was seen to jump off. How that louse fly knew it was time to do that is amazing. After the fledgling had left, the louse fly was clearly visible on the inside wall of the box. The flies would of course not want to go anywhere but would need to deposit the larva to pupate and wait until the following spring when the Swifts return. The other interesting question is how the louse flies that are on the adults know when those adults are not going to return to the box because it is a fact that the adults do desert the young who have to make the long trip to Africa on their own. In my box the juvenile Swift was left for four days on its own before it decided it was time to go.
Swifts now rely heavily on buildings for nest sites in the UK and they are under pressure from home owners as repairs are made to house roofs. Ideally Swifts should be given the same sort of protection that is afforded to bat roosts but it is encouraging that they will use artificial boxes and the hope is that more will be installed by householders in the future. Who would want a world without the scream of Swifts in the summer!
References
Walker, M.D. & Rotherham, I.D. 2010. Characteristics of Crataerina pallida(Diptera: Hippoboscidae) populations; a nest ectoparasite of the common Swift, Apus apus (Aves: Apodidae). Experimental Parasitology,126(4): 451-455.
Kemper, H., 1951. Beobachtungen an Crataerina pallida Latr Und Melophagus ovinus L. (Diptera, Pupipara). Zeitschrift fiir Hygiene (Zoologie)39:225–259.
Swift Conservation web site www.swift-conservation .org
Images
 1 -Two Swifts in the box on the first day
1 -Two Swifts in the box on the first day
 2 – The Louse Fly on an adult
2 – The Louse Fly on an adult
 3 – First Sighting of the Chick
3 – First Sighting of the Chick
 4 – The chick reversing up to the entrance to defecate
4 – The chick reversing up to the entrance to defecate
 5 – A Louse Fly on the chick
5 – A Louse Fly on the chick
 6 – The young chick probably three weeks old
6 – The young chick probably three weeks old
 7 – The first attempt to leave the box failed
7 – The first attempt to leave the box failed
 8 – Crataerina pallida from wikipedia
8 – Crataerina pallida from wikipedia
Worcestershire Record | 33 (Nov 2012) page: 26-27 | Worcestershire Biological Records Centre & Worcestershire Recorders
Dragonflies in Worcestershire 2012
Mike Averill
What a year of weather extremes; from drought in March to floods in July. The summer (Apr-Sep) was very wet with over 150% of annual average rainfall compared to 58% for the same period in 2011. Consequently it was a case of picking the days very carefully for insect watching. The only consolation would be that aquatic insects would not need to search far for wetland habitat.
After a very warm dry start in March when early flowering plants did well for pollination, a wet and cool April brought frosts and this delayed much insect activity. The Common Clubtail Gomphus vulgatissimus didn’t emerge until late May, as much as 14 days later than in 2011 but it wasn’t a bad year for them and they all emerged in a very short two week period making up for lost time.
It has been mentioned in previous years that the Beautiful Demoiselle Calopteryx virgo has been doing well and this was the case again with the species showing in as many numbers and locations as the more commonly encountered Banded Demoiselle Calopteryx splendens. Twenty years ago a walk up the river Teme would reveal few Beautiful demoiselles other than in the minor tributaries but this year they were to be seen alongside the Banded Demoiselle in most places. The species is also being reported from smaller rivers east of the Severn as well.
All dragonflies and all the damselflies except for the demoiselles have a true colour cell in the wing (pterostigma). Of the demoiselles only the females have the equivalent to a pterostigma in that it is a coloured area of cells near the front edge of the wing (Fig. 1.) as opposed to the enlarged single cell as seen in all other dragonflies. Sometimes this is missing even in the females and is rarely recorded but a Banded Demoiselle without one was seen in Kidderminster in late May this year (Fig. 2.).
Immigration of dragonflies from the continent occurred as usual in to southern England but only the Red-veined Darters Sympetrum fonscolombiireached Worcs this time with a few being seen at Grimley Gravel Pits, Pirton Pool, and a few at Kemerton gravel pits.
The Scarce Chaser Libellula fulva continues to expand its range with records occurring at Pendock, Churchill, Hurcott and most surprisingly at Hillditch Pool. All those sites are pools and so are not really the classic river habitat that it is normally associated with this species. Of those sites Hillditch is perhaps the most surprising as there was quite a lot of breeding activity and this site, although small, could be mistaken for a river as for about a hundred metres it does look like a very slow flowing medium sized river. Like most dragonflies the males of the Scarce Chasers are the most noticeable as they adopt display territories but females were seen as well with up to 12 individuals pairing up. Next season will be anticipated with interest to see if the population increases at this site.
The Small Red-eyed Damselfly Erythromma viridulum appeared to be making a relentless push westwards after initially appearing in Essex in 1999, but there seems to be some evidence that it is slowing its progress. It first appeared in Worcestershire in 2006 and has centred its activity at Croome Park. Last year the numbers were in the hundreds possibly thousands but this year they were down to a few hundred. That may be weather related and it could be due to less algal growth, one of its preferred plants, which being more likely to develop in hot weather, is also weather related. The damselfly prefers water milfoil and hornwort but is quite happy on blanket weed. Attempting to estimate damselfly numbers is rather subjective but having the started transect counts at Croome this year has helped make comparisons.
There is not much colour variation in mature dragonflies in the UK but it can be seen in the Aeshnid species, specifically the Southern Hawker Aeshna cyanea. Both the males and females can be seen with all the abdominal colour spots showing as light blue rather than green and the thoracic shoulder stripes appear yellow rather than green (Figs. 3 &4.) It is not mentioned in most identification books and the cause is not fully understood but is thought to be either related to temperature, to stages of development or is a colour form.
Colours will generally go lighter with heat, the point being to enable incoming radiation to be reflected back out. The reverse is the case in cooler weather and may be why the Highland Darter Sympetrum nigrescensis darker being located in Scotland where radiation will be less. This theory is not favoured by observers in Scandinavia where they argue that it should, in that case, be much more commonly seen in the cooler climates.
With ageing, colours normally change in the days immediately after emergence as the individual matures. In the later stages of adult life, individuals will also develop a darker colour and some very old adults like females of the Common Darter Sympetrum striolatum will start to take on the hues of the males.
Many of the examples cited as having blue colouration appear to have recently emerged and it could be that they are just a transitional phase before adopting the normal colours. Again if this is the cause it is strange it isn’t seen more often in a well observed group like dragonflies.
Some damselflies like the Ischnurids, show colour variation in females, the Blue-tailed Damselfly Ischnura elegans has various colour forms and this is called polychromatism (only the females show this) whereas polymorphism is where the males and females show colour differences between each other. Polychromatism isn’t usually encountered in the larger UK dragonflies and so blue forms in the Aeshnids is unusual. We have had a handful of these sightings in Worcestershire all in the Southern Hawker. Both males and females can show the effect. In order to test the temperature and ageing theory a live specimen was collected this summer near Pershore where there were two females hawking a hedgerow. It was fed on mosquitoes in a summer house for four days until it unfortunately broke its neck trying to get out, but in all the time it was there, the colour didn’t change at all (photos). This was despite the temperature ranging from 9 to 27°C. Perhaps this individual can reveal something from its DNA.
The damp cool summer continued in to September and so the flying season was all but over by mid September with just the hardy Common Darters, Migrant and Southern Hawkers Aeshna mixta and Aeshna cyanea making the odd appearance on warmer days.
2012 was a bit disappointing for the last year of recording for the National Atlas, nevertheless the survey time is complete and the full details will appear in the next issue of the Worcestershire Record.
Images

Fig. 1. Banded Demoiselle (f) with pterostigma. Mike Averill

Fig. 2. Banded Demoiselle (f) without pterostigma. Mike Averill

Fig. 3. Southern Hawker (f) blue form. Mike Averill

Fig. 4. Southern hawker (f) blue form side view.Mike Averill
Worcestershire Record | 33 (Nov 2012) page: 9-11 | Worcestershire Biological Records Centre & Worcestershire Recorders
Invertebrate Records from a Worcestershire Garden 2012
Denise Bingham
This year it seemed that our garden in Kidderminster was a good place to be as several unusual and rare species turned up. Guess it goes to show that keen observation of a small area can provide some good finds! Thanks to John Bingham who did some of the identification of my various finds.
Andrena nigrospina, (Thompson, 1872). Hymenoptera: Andrenidae . RDB1(Red Data Book).
This RDB1 solitary bee is well known to local naturalists at its Blackstone Nature Reserve site near to the heathlands of Devil’s Spittleful. (Trevis 2009). A large black bee was seen in the garden on 20 July feeding on Rosa mundi. It was caught and very surprisingly we identified it as Andrena nigrospina. Having made some effort to see this bee at Blackstone over the last few years we could hardly believe it had appeared in the garden. A photograph was sent to Geoff Trevis who confirmed our identification. Our garden is within 0.5km of acidic semi-improved sandy grassland at Hurcott so we can only assume the bee had flown somewhere from there where it may nest to find nectar on our garden plants. This links in with Brett Westwood’s sightings of the bee at Ismere about 2km to the north.
Tiphia femorata, (Fabricus,1775). Hymenoptera:Tiphiidae.
This rather odd-looking wasp which looks more like a rove beetle than a wasp was found on garden flowers on 25 August. The wasp is a parasitoid on scarabaeid beetle larvae (recorded hosts include Aphodius, Rhizotrogusand Anisoplia). The female burrows into the soil to find a larval host in its cell, stings the larva and kneads it with her mandibles. An egg is laid on the larva which hatches to produce a larva which takes about three weeks to consume its host. (BWARS web site). Thanks to Geoff Trevis who confirmed our identification and reported that Andy Jukes has recorded it at Blackstone and Burlish Top.
Sehirus luctuosus , (Mulsant & Rey,1866). Hemiptera: Cydnidae.
After Jane Scott’s report in Worcestershire Record (No32 April 2012, p.27) the hunt was on to find this shieldbug as Forget-me-not was common in our garden. On a sunny day on 8 May I found one on the garden path, thereafter many were seen over the following weeks near Forget-me-not plants. We were spotting them running around on bare soil next to Forget-me-not flowers. Difficult to estimate numbers but certainly dozens must have been present in several areas of the garden. Not to be out done John Bingham searched Forget-me-not plants on the Devil’s Spittleful NR and managed to locate two bugs on 23 May. They do hide under the leaves but in warm weather are quite active and can be seen running over bare ground or on pathways near the host plant.
Volucella inanis, (Linnaeus, 1758). Diptera: Syrphidae
This hoverfly is now well established in the county but still rather scarce in north Worcestershire. It did finally turn up in our garden on 16 August. In the following week several more were seen, both male and female.
Chrysotoxum verralli, (Collin, 1940). Diptera: Syrphidae
I saw several of these attractive hoverflies on 8 July seen taking nectar from various flowers.
Stratiomys potamida, Meigen, (1822). Diptera:Stratiomyidae, Solider-fly.
This attractive solider-fly, the Banded General, is a local species associated with wet places such as pools or carr woodland, occasionally recorded around the county (Stubbs and Drake 2001). So it was not expected to appear in our garden feeding Leucatheum spp. flowers. I recorded it on 19 August and saw another one a few days later. The nearest wet site is Podmore Pool with its carr woodland backwater some 0.5 km distant.
Platyarthrus hoffmannseggii , (Brandt, 1833). Arthopoda: Isopoda
I found a colony of a small white woodlouse on the edge of the garden lawn on 23 June that was identified as Playtharthrus hoffmansegii. The distinctive blind woodlouse is apparently common in Worcestershire but Kidderminster is near the northern limit of this species. A number of Lasius niger ants were nearby and appeared to be associated with the woodlice. The Woodlouse Atlas (Gregory 2009) says it tends towards calcareous soils, perhaps because they warm up quickly.
References / Information
Bantock & Botting, British Bugs; www.britishbugs.org.uk
Gregory, S. 2009. Woodlice and waterlice (Isopoda:Oniscidae & Asellota) in Britain and Ireland. FSC Publications. Preston Montford.
Scott, J. 2012. Interesting bugs at Woodbank, Astley Burf, 2011 Worcestershire Record. 32:27.
Southwood, T. & Leston, D. 1959 Land and Water Bugs of the British Isles. Warne.
Stubbs, A & Drake, M. 2001. British Soliderflies and their Allies. British Entomological and Natural History Society.
The UK Bees, Wasps and Ants Recording Society (BWARS). www.bwars.com
Trevis, G. (2009) Andrena nigrospina. Worcestershire Record. 27:23.
Images

Fig. 1. Andrena nigrospina

Fig. 2. Tiphia femorata

Fig. 3. Sehirus luctuosus

Fig. 4. Volucella inanis

Fig. 5. Chrysotoxum verralli

Fig. 6. Stratiomys potamida

Fig. 7. Platyarthrus hoffmannseggii
Worcestershire Record | 33 (Nov 2012) page: 7-9 | Worcestershire Biological Records Centre & Worcestershire Recorders
Invertebrate Records from Worcestershire
John Bingham
[Correction: original reference to Trox scaber, (Linnaeus, 1767). Coleoptera: Trogidae (Fig. 01. deleted) was in error.]Trachys minutus, (Linnaeus, 1758). Coleoptera: Buprestidae RDB2 and LBAP
This small beetle (3-4mm) was found on 26 August on a small-leaved lime leaf at Shrawley Wood. The tree was next to an open glade area and was in full afternoon sunlight. The female beetle lays eggs on the leaves of deciduous trees, normally sallow, hazel or elm. The larvae eat the green tissue between the upper and lower layer of the leaves, making blotch mines on the edge of the leaf. It may use lime tree leaves at Shrawley, as this is also quoted as a food plant for the larvae. See Web pages: ‘the leaf and stem mines of British flies and other insects’ for image of the leaf mine. Alexander (2003) gives it as an elusive species with leaf mines on willow in ancient semi-natural woodlands. He states that the majority of the records are old and the species has decreased parallel with the decline in active coppice management, woods being too dark and shady for the southern warmth-loving species. The beetle was recorded by Ross Piper on 15/05/2002 also from Shrawley Wood, apparently this is the only site in Worcestershire for this beetle. WBRC (Worcestershire Biological Records Centre). Thanks to Harry Green and John Meiklejohn for supplying information on the records.
Pyrrhidium sanguineum, ( Linnaeus, 1758), Coleoptera: Cerambycidae
Several records of the this longhorn beetle have been found in Wyre Forest but this time a small colony of several dozen was found by Denise Bingham under the bark of a standing dead oak tree. The site was near the Dowles Brook on the Worcestershire side on a tree deliberately ring-barked for deadwood habitat. Good to know these trees do attract rare deadwood species! (no picture).
Platycis minutus, (Fabricius, 1787). Coleoptera: Lycidae, Nationally Scarce B
Discovered on the ‘great bog’ in Wyre Forest by Denise on a rotten wet log on 28 August. Four species of Lycidae (net-winged beetles) occur in Britain. The common name ‘net-winged’ refers to the pattern of raised ridges on the elytra. The larvae develop in large relatively soft moist decaying heartwood, especially beech Fagus and probably ash Fraxinus; mostly in closed-canopy areas of ancient woodland; southern and eastern England. (Alexander 2003).
Interestingly more records came to light for 2012. We recorded another beetle this time on the Cotswolds near Rodborough; Dave Scott found one at Astley and Rosemary Winnall found one at Bliss Gate. Perhaps a good year for this species?
Clytra quadripunctata, (Linnaeus, 1758). Coleoptera: Chrysomelidae
This large beetle is associated with wood ant nests Formica rufa. It appears to be quite common and widespread in the Wyre Forest area but not often seen. Over a dozen records are on WBRC database and all are from the Wyre Forest possibly the only location for this species in Worcestershire. This year I found the beetle at Ribbesford Woods on bracken near to wood ants. In the same area several Scarce 7-spot ladybirds Coccinella magnificawere also noted.
Andrena apicata, (Smith, 1847). Hymenoptera:Andrenidae, Nationally Notable B.
Good numbers of this early flying solitary bee were seen on 22 March along a sandy track on the Devil’s Spittleful/Rifle Range. Both male and females were noted in good numbers and many were resting on gorse stems or tree trunks. It appears to be rare in Worcestershire with only one other record at Bliss Gate, Wyre Forest. Geoff Trevis kindly identified the bee.
Lasiopogon cinctus, (Fabricius, 1781). Diptera:Asilidae, Nationally Scarce.
Several of these flighty robber-flies were seen at Hartlebury Common on 24 April. They were hunting for various small flies over the open heathland. Luckily when a prey item was caught the fly settled on the ground allowing observation and photography. The only previous records were made by Andy Jukes at Devil’s Spittleful and Hartlebury Common, both in 2010. (WBRC records).
Chrysotoxum arcuatum (Linnaeus, 1758). Diptera: Syrphidae
This is smaller than the other similar looking Chrysotoxum species and more common in upland areas, so it is uncommon in Worcestershire. Several were noted just over the county boundary at Longdon Wood, Wyre Forest on 28 August. More than likely it can be found on the Worcestershire side as well.
Grapholita internana, (Guenée, 1845). Lepidoptera:Tortricidae
This small moth was seen in good numbers flying around gorse bushes at Hartlebury Common on 24 April. Apparently, despite suitable habitat, it is quite uncommon in Worcestershire with previous records from the Malverns, Old Hills, Hartlebury Common, and the Worcestershire part of Kinver Edge. (Tony Simpson per. comm.)
Neottiglossa pusilla, (Gmelin, 1789). Hemiptera:Pentatomidae, National Local.
This small brown shieldbug 4-5mm long appears to be near the edge of its range being more common further east in Britain. On 5 June I found two at Shrawley Wood resting on a grass stem along a sunny ride. Later on 28 June just over the county boundary in Shropshire Denise found another specimen on a grassy ride at Malpass Wood, Wyre Forest. Probably an overlooked species, the larvae are said to feed on Poa grass species. (Evans and Edmondson 2005). There are only a few records from Worcestershire, one in 2006 by John Partridge at Hawkbatch, Wyre Forest. It may prefer more acidic grasslands and possibly sandy soils limiting its distribution in the county perhaps? Sweeping taller grasses with a net might find it. Rosemary Winnall also reported a discovery on 23 June 2012 from the Shropshire side of the Dowles Brook.
Corizus hyoscyami, (Linnaeus, 1758). Hemiptera: Rhopalidae.
Now becoming quite widespread with records by Denise from Wyre Forest at Longdon Wood, Shropshire, on 28 August. I found it at Blackstone Nature Reserve near Bewdley on arable fields on 31 August. (No picture).
Pholidoptera griseoaptera, (De Geer, 1773). Orthoptera: Dark Bush Crickets.
The abundance of this species at Shrawley Wood was quite notable in August and September in 2012. Nearly every sunny patch was crawling with dozens of crickets many of which looked larger than normal. Yet a few miles to the north in Wyre Forest they remain an uncommon insect more restricted to the River Severn area and a few locations around the forest. I have seen the occasional insect along the Dowles Brook valley but as yet they do not appear to be along other forest rides. Perhaps just a matter of time?
References / Information
The leaf and stem mines of British flies and other insects. http://www.ukflymines.co.uk/Beetles/Trachys_minutus.php
Southwood, T & Leston, D. 1959. Land and Water Bugs of the British Isles. Warne.
Bantock & Botting, British Bugs; www.britishbugs.org.uk
Hawkins, R. 2003. Shieldbugs of Surrey. Surrey Wildlife Trust
Alexander, K.N. A. 2003. Provisional Atlas of the Cantharoidea and Buprestoidae (Coleoptera) of Britain and Ireland. Biological Records Center
The UK Bees, Wasps and Ants Recording Society (BWARS) www.bwars.com
Evans, M. & Edmondson, R. 2005. A photographic guide to the Shieldbugs and Squashbugs of the British Isles. WGUK in association with WildGuideUK
Images
Fig. 01. Deleted (was an error)

Fig. 02. Trachys minutus. John Bingham

Fig. 03. Platycis minutus. John Bingham

Fig. 04. Clytra quadripunctata. John Bingham

Fig. 05. Andrena apicata Female. John Bingham

Fig. 06. Andrena apicata Male. John Bingham

Fig. 07. Lasiopogon cinctus. John Bingham

Fig. 08. Chrysotoxum arcuatum. John Bingham

Fig. 09. Grapholita internana. John Bingham

Fig. 10. Neottiglossa pusilla. John Bingham

Fig. 11. Pholidoptera griseoaptera Dark Bush Cricket. John Bingham
Worcestershire Record | 33 (Nov 2012) page: 50-53 | Worcestershire Biological Records Centre & Worcestershire Recorders
Waxcap and other fungi near Stourbridge – an Update
John Bingham
In April 2011 Rosemary Winnall reported in Worcestershire Record (Winnall 2011) the discovery in 2009 of a waxcap grassland, or rather a series of waxcap garden lawns at Broome near Hagley, Worcestershire. The owner David Taft was very obliging and allowed further access in the autumn of 2011 when Rosemary asked several mycologists to help with the survey of the fungal mycota of David’s lawns. Little did we know what would develop from this waxcap survey. Rosemary has asked me to report the findings.
The four lawns date from about the time the house was built around 1906 and appear to have been landscaped as they are flat but on slightly different levels. The largest is about 0.1ha in area. The soil is derived from the sandstone typical of the Kidderminster area and would naturally support acid grassland or heath. The lawns are dominated by mosses with some lichen patches suggesting semi-improved grassland such as NVC U1 (Festuca ovina-Agrostis capillaris-Rumex acetosella grassland), but generally more characteristic of MG6 (Lolium perenne – Cynosurus cristatusgrassland), albeit with a very moss dominated sward. Botanically the lawns were not particularly rich supporting mainly rosette forming species such as Cat’s-ear Hypochaeris radicata. No fertilisers or chemicals had been used for many years, if ever?
I made my first visit on the 15th November 2011 together with Rosemary and Denise Bingham. Rosemary had already recorded good numbers of Hygrocybe calyptriformis (Pink Waxcap) the previous week. Waxcaps were present in good numbers, and recording was carried out. After searching the first lawn I moved to the second larger lawn and after about 10 minutes I spotted a small fungus I was familiar with: Squamanita paradoxa(Powdercap Strangler). I could hardly believe it. At first three were found then after some excitement by all concerned Rosemary found some more specimens making at total of nine fruiting bodies. We believe this is the first county record for the species.
Squamanita pardoxa is listed as Vulnerable in Red Data (RDB) List edition 1 and Near Threatened in RDB List edition 2. It appears to occur at 15 sites in the UK and I personally have been responsible for finding two sites back in 2005 (Bingham & Bingham 2005). Denise and I had added another two sites from Shropshire in 2011 (Titterstone and Brown Clee Hills), that is excluding the site at Broome. Globally the fungus is rare with most records coming from Norway and Sweden. What makes S. paradoxa particularly interesting is the fact it is a parasite on other fungi, within the Cystodermagenus and in particular C. amianthinun (Earthy Powdercap) that appears to be the host for all UK specimens. It takes over its host and is a gall or ‘cecidiocarp’ first described in 1948 but not fully understood until 1965 (Redhead et al.1984) when it was described further. S. paradoxa is a member of the Tricholomataceae family with 10 species in total world-wide.
It grows with the stem of the Cystoderma and replaces the upper stem and cap of the orange coloured Cystoderma with its own greyish cap. Exactly how is still not understood but Gareth Griffith is undertaking research at Aberystwyth University (Matheny & Griffith 2010). I forwarded a photo of the Broome find to Gareth and immediately came a request for a specimen for his DNA research at Aberystwyth. Several specimens were sent and are now in culture with Cystoderma to see how the ceidicarp develops with the Cystoderma host.
A return visit was made to Broome on the 17 September with David Antrobus who was identifying Entoloma fungi, Mark Lawley to look at bryophytes and Brett Westwood who had an idea for a BBC Radio 4 item on grassland fungi. All this was much to the surprise and interest of David Taft and his gardener David Elder, who were both very helpful and welcoming to our strange antics. In due course Brett recorded a feature with Rosemary and me on grassland fungi, duly broadcast on the TheLiving Worldprogramme (http://www.bbc.co.uk/search/?q=Powdercap strangler or http://www.bbc.co.uk/nature/16029977) with my photograph of S.paradoxa on the BBC Nature webpage.
In Fig. 1. note the orange colour of the lower stipe which is the Earthy Powdercap Cystoderma amianthinun forming a stocking-like ring above which is the grey Powdercap Strangler Squamanita paradoxa stipe and cap.
Of course we did record the waxcaps with (total for all years) 14 Hygrocybespecies, nine Clavariod, one Geoglossaceae and three Entolomas. (See Table 1).
This is a very respectable list given that a score of 10-11 Hygrocybespecies, 5-6 Clavariod, 4 or more Geoglossium and 8-9 Entolomas are considered to make a site of national importance. (Nitare 1988). David Boertmann in his book on Hygrocybe has a table showing a score of between 11-15 Hygrocybe as being of regional importance and with 16-20 species being of national importance. (Boertmann 1996).
Of course there will be more to discover, certainly Entolomas were under recorded due to the dry weather and a late flush in 2011. More recording would certainly be of interest at such a rich site.
| Name of fungus | Associated Organism | Medium | Ecosystem | Frequency | Date |
| AGARICALES | |||||
| Agaricus augustus | Gramineae | soil grass | Grassland lawn | Rare | 15/11/2011 |
| Arrhenia retiruga | Moss | soil moss | Grassland lawn | Rare | 17/11/2011 |
| Auriscalpium vulgare | Pinus | cone | Grassland lawn | Rare | 17/11/2011 |
| Calocybe carnea | Gramineae | soil grass | Grassland lawn | Rare | 15/11/2011 |
| Clitocybe fragrans | Gramineae | soil grass | Grassland lawn | Occasional | 15/11/2011 |
| Clitocybe rivulosa | Gramineae | soil grass | Grassland lawn | Rare | 15/11/2011 |
| Conocybe apala | Gramineae | soil grass | Grassland lawn | Rare | 15/11/2011 |
| Cystoderma amianthinum | Gramineae | soil grass | Grassland lawn | Frequent | 15/11/2011 |
| Dermoloma cuneifolium | Gramineae | soil grass | Grassland lawn | Rare | 15/11/2011 |
| Entoloma infula | Gramineae | soil grass | Grassland lawn | Occasional | 17/11/2011 |
| Entoloma jubatum | Gramineae | soil grass | Grassland lawn | Occasional | 17/11/2011 |
| Galerina clavata | Gramineae | soil grass | Grassland lawn | Occasional | 17/11/2011 |
| Galerina vittiformis | Gramineae | soil grass | Grassland lawn | Occasional | 17/11/2011 |
| Galerna pumila | Gramineae | soil grass | Grassland lawn | Frequent | 15/11/2011 |
| Hygrocybe calyptriformis | Gramineae | soil grass | Grassland lawn | Occasional | 15/11/2011 |
| Hygrocybe ceracea | Gramineae | soil grass | Grassland lawn | Frequent | 15/11/2011 |
| Hygrocybe chlorophana | Gramineae | soil grass | Grassland lawn | Rare | 15/11/2011 |
| Hygrocybe coccinea | Gramineae | soil grass | Grassland lawn | Occasional | 15/11/2011 |
| Hygrocybe flavipes | Gramineae | soil grass | Grassland lawn | Rare | 15/11/2011 |
| Hygrocybe irrigata (uguinosus) | Gramineae | soil grass | Grassland lawn | Rare | 15/11/2011 |
| Hygrocybe laeta var. laeta | Gramineae | soil grass | Grassland lawn | Rare | 15/11/2011 |
| Hygrocybe miniata (strangula) | Gramineae | soil grass | Grassland lawn | Rare | 15/11/2011 |
| Hygrocybe pratensis | Gramineae | soil grass | Grassland lawn | Frequent | 15/11/2011 |
| Hygrocybe psittacina | Gramineae | soil grass | Grassland lawn | Occasional | 15/11/2011 |
| Hygrocybe punicea | Gramineae | soil grass | Grassland lawn | Rare | 15/11/2011 |
| Hygrocybe reidii | Gramineae | soil grass | Grassland lawn | Rare | 15/11/2011 |
| Hygrocybe russocoriacea | Gramineae | soil grass | Grassland lawn | Rare | 15/11/2011 |
| Hygrocybe virginea | Gramineae | soil grass | Grassland lawn | Frequent | 15/11/2011 |
| Hygrophoropsis aurantiacum | Pinus | soil grass | Garden | Occasional | 17/11/2011 |
| Inocybe pusio | Pinus | soil bare | Garden | Occasional | 15/11/2011 |
| Inocybe sindonia | Pinus | bare soil | Grassland lawn | Rare | 17/11/2011 |
| Laccaria laccata | Pinus | soil grass | Garden | Occasional | 15/11/2011 |
| Lactarius deliciosus | Pinus | soil needle | Grassland lawn | Occasional | 15/11/2011 |
| Lactarius glyciosmus | Pinus | soil grass | Grassland lawn | Occasional | 15/11/2011 |
| Mycena aetites | Gramineae | soil grass | Grassland lawn | Frequent | 15/11/2011 |
| Mycena avenacea | Gramineae | soil grass | Grassland lawn | Occasional | 17/11/2011 |
| Mycena clavularis | Gramineae | cone | Grassland lawn | Rare | 17/11/2011 |
| Mycena flavoalba | Gramineae | soil grass | Grassland lawn | Occasional | 17/11/2011 |
| Mycena pura | Gramineae | soil grass | Grassland lawn | Rare | 15/11/2011 |
| Panaeolina foenisecii | Gramineae | soil grass | Grassland lawn | Occasional | 15/11/2011 |
| Panaeolus olivaceus | Gramineae | soil grass | Grassland lawn | Rare | 17/11/2011 |
| Paxillus involutus | Angiosperm | soil | Garden | Rare | 17/11/2011 |
| Rickenella fibula | Gramineae | soil grass | Grassland lawn | Occasional | 15/11/2011 |
| Rickenella swartzii | Gramineae | soil grass | Grassland lawn | Occasional | 15/11/2011 |
| Russula gracillima | Betula | soil grass | Grassland lawn | Rare | 15/11/2011 |
| Russula nigricans | Betula | soil grass | Garden | Rare | 17/11/2011 |
| Squamanita paradoxa | Gramineae | soil moss | Grassland lawn | Rare | 15/11/2011 |
| Stropharia caerulea | Gramineae | soil grass | Grassland lawn | Occasional | 17/11/2011 |
| Stropharia pseudocyanea | Gramineae | soil grass | Grassland lawn | Rare | 17/11/2011 |
| Suillus luteus | Pinus | soil needle | Grassland lawn | Occasional | 15/11/2011 |
| APHYLLOPHORALES | |||||
| Bjerkandera adusta | Angiosperm | stump | Garden | Rare | 17/11/2011 |
| Clarvaria acuta | Gramineae | soil grass | Grassland lawn | Rare | 15/11/2011 |
| Clarvaria fragilis | Gramineae | soil grass | Grassland lawn | Occasional | 15/11/2011 |
| Clavaria argillacea | Gramineae | soil moss | Grassland lawn | Rare | 15/11/2011 |
| Clavulinopsis corniculata | Gramineae | soil moss | Grassland lawn | Occasional | 15/11/2011 |
| Clavulinopsis fusiformis | Gramineae | soil grass | Grassland lawn | Occasional | 15/11/2011 |
| Clavulinopsis helvola | Gramineae | soil grass | Grassland lawn | Frequent | 15/11/2011 |
| Clavulinopsis laeticolor | Gramineae | soil grass | Grassland lawn | Occasional | 15/11/2011 |
| Clavulinopsis luteoalba | Gramineae | soil grass | Grassland lawn | Occasional | 15/11/2011 |
| Geoglossum fallax | Gramineae | soil moss | Grassland lawn | Rare | 08/11/2010 |
| Hypomyces aurantius | Polypore | fruit body | Grassland lawn | Rare | 15/11/2011 |
| Phellinus pomaceus | Prunus | branch | Garden | Rare | 17/11/2011 |
| Polyporus durus | Angiosperm | stump | Garden | Rare | 17/11/2011 |
| Ramariopsis kunzei | Gramineae | soil grass | Grassland lawn | Rare | 30/11/2009 |
| HETEROBASIDOMYCETES | |||||
| Auricularia auricula-judae | Sambucus | stem | garden | Rare | 17/11/2011 |
| ASCOMYCETES | |||||
| Peziza badia | Angiosperm | soil bare | garden | Rare | 15/11/2011 |
| Rhyisma acerinum | Acer | leaf | garden | Frequent | 17/11/2011 |
| Trochila ilicina | Ilex | leaf | garden | Frequent | 17/11/2011 |
| MYXOMYCETES | |||||
| Sebacina incrustans | Gramineae | soil grass | Garden | Rare | 15/11/2011 |
Table 1 Fungi recorded at The Croft, Broome, Worcestershire, November 2011 by John Bingham, Denise Bingham, Rosemary Winnall, David Antrobus. Site SO897790, garden lawn, mossy, unfertilized for 50 years.
In addition to the fungi we have Mark Lawley’s list of associated bryophytes.
The lawns consisted mainly of moss but with no notable species. Mark recorded the following 39 bryophytes from the garden. Nearly all the moss in the lawns is Rhytididadelphus squarrosus, with much less Atrichum undulatum, Brachythecium albicans, B. rutabulum, Hypnum cupressiforme, H. jutlandicum, Plagiomnium rostratum, P. undulatum, Polytrichum juniperinum, P. piliferum, and Lophocolea bidentata. Hypnum jutlandicumand the two Polytrichum species were growing only in sandy (or sandier) soil in the lawn beside the drive. There was also a noticeable amount of Cladonia lichen there too.
Acknowledgement
Many thanks to David Taft for his kindness and access permission.
References
Bingham, J. & Bingham, D. 2005. Squamanita paradoxa in Shropshire. Field Mycology 6(1):11–12
Boertmann, D. 1996. The Genus Hygrocybe. Fungi of Northern Europe. Danish Mycological Society.
Mathey, P M. & Griffith, G. W. 2010. Mycoparasitism between Squamanita paradoxa and Cystoderma amianthinum (Cystodermateae, Agaricales). Published online.
Nitare, J. 1988. Jordtunger, en svampgrupp pa tillbakagang I naturliga fodermarker. Svensk. Bot. Tidskr. 82:341-368.
Redhead, S.A., Ammirati, J.F., Walker, G.R., Norvell, L.L. & Puccio, M.B. 1994. Squamanita contortipes, the Rosetta Stone of a mycoparasitic agaric genus. Can. J. Bot. 72:1812–1824.
Winnall, R. 2011. Waxcap Fungi Near Stourbridge. Worcestershire Record30:35-36.
Image

Fig. 1. Squamanita paradoxa. John Bingham
Worcestershire Record | 33 (Nov 2012) page: 42-43 | Worcestershire Biological Records Centre & Worcestershire Recorders
Bechstein’s Bat – a study on the population size, foraging range and roosting ecology at Grafton Wood, Worcestershire, 2012. A preliminary summary of findings
Johnny Birks, Eric Palmer and James Hitchcock
This article follows on from The National Bechstein’s Survey and Beyond in Worcestershire Record (Sedgeley et al 2012), which outlined the efforts to identify the presence of Bechstein’s bat in Worcestershire, and the subsequent successful bid for funding from the Peoples’ Trust for Endangered Species (PTES) for further studies. The funding enabled us to carry out a radio tracking project aimed at increasing our knowledge of the roosting and foraging habitats of Bechstein’s Bat in a managed woodland. Worcestershire Wildlife Trust initiated the work, in partnership with Johnny Birks and Eric Palmer, following the record of a lactating female Bechstein’s Bat at Grafton Wood which indicated there was probably a colony of the bats living in, or around, the wood. Since Grafton Wood became a nature reserve in 1997, its management has been driven mainly with invertebrates in mind and their need for open space and high light levels. This management has been very successful in encouraging diversity and increasing butterfly numbers and breeding birds. However, past studies have shown (Schofield & Morris, 2000) that Bechstein’s bats favour foraging under closed canopy woodland with a strong understorey (Harris & Yalden 2008) which is well represented in Grafton Wood (Fig. 1.), alongside the actively managed rides and coppice plots. So, it was felt a detailed study to aid understanding of how the bats use the wood was much needed and would provide information for the management plan and ensure that bats were considered alongside other species in the future.
The project depended largely upon a very successful collaboration with the Worcestershire Bat Group whose members committed a huge amount of time and energy helping with catching (using harp traps and mist nets) and radio-tracking Bechstein’s bats at Grafton Wood through the rain-soaked summer ot 2012. We are indebted to them for their massive contribution and their remarkable ability to stay cheerful through the wettest and muddiest summer in 100 years.
Well, what did the project find out? At the time of writing the detailed analysis of data is still taking place, but, we can share some preliminary findings.
Despite the best disruptive attempts of the awful weather, tagging and radio-tracking was carried out, as planned, across three main survey periods: May, June and end August/beginning of September – chosen to avoid catching bats when they would be heavily pregnant or feeding young.
Our early season catch was almost entirely of underweight bats of all species, with additional problems due to a higher than average tag failure rate and of course the weather. Despite this we were able to fit one tag in May and start the process of chasing bats around in the dark. By the end of the season we had fitted a total of eight tags, four less than was hoped and had identified eleven day-roost trees – five in Grafton Wood and six outside. Tree species used were Willow(3), Oak(4), Ash(3) and Silver Birch(1). None of the roosts were in old woodpecker holes, one in a natural rot hole and the other (a lone male bat) in a crack. Through the identification of roosts we were able to carry out timed roost emergence counts which gave us a maximum count of 50 bats which emerged from a woodpecker hole in a Crack Willow pollard.
Of the roosts outside the wood the furthest away was just over 1.5km south. This kind of distance has been recorded in other studies, but was still something of a surprise to us, especially given that the area to the south of the wood happens to be the least wooded area around Grafton Wood.
Overall, we found a bias in bat activity towards the south of the wood, both in terms of roosting and foraging within the wood and the roosts outside of it. Roosts were often near water – the Piddle Brook or a farm pond – which could be a coincidence or it could highlight a need for flight corridors.
Tracking foraging bats revealed that they favour discrete areas of closed-canopy woodland with a strong understorey, which confirmed much of what we had read about the bats in previous studies. However, our late-season animals also foraged around field edges, plantation woodland and along watercourses.
The radio-tags allowed us to confirm that the colony frequently switched roosts, often en masse, which may have been a result of the inclement weather, but may highlight other needs: the bats may move regularly to prevent the build up of waste and parasites, which may increase the risk of disease. Frequent roost switching does bring home the importance of the availability of a wide variety of potential roost trees, in a variety of settings, with woodpecker holes (and other cavities) in them.
The final report will aim to characterise the roost trees, define and characterise foraging areas and perhaps shed some light on what features of the landscape around the Wood really are the most important habitat for commuting and foraging Bechstein’s Bats.
One thing that we can say at this stage is that despite the challenges presented by the weather and the demands of night-time radio-tracking faced so bravely by our band of volunteers, we have hugely increased our knowledge and understanding of Bechstein’s Bat in Grafton Wood and its environs. And it appears that we have a healthy sized breeding colony. As with all scientific field studies we have only just begun to scratch the surface and there is still much more to learn and more work to be done. We hope to follow up this study with the introduction of 100 roosting boxes – woodcrete Schwegler 2FNs – into Grafton Wood which, if used, will allow us to monitor the colony size and, if we can organise ringing the bats, we will hopefully be able to gather data on other important population parameters.
These are exciting times! A full update will follow after the report is published
Notes
Grafton Wood Nature Reserve is jointly owned and managed by Worcestershire Wildlife Trust and Butterfly Conservation.
Harp traps (Fig. 2.) have vertical nylon strings – fishing line type stuff – that is strung vertically in two slightly offset rows. They are sited in areas where bats are likely to be foraging and are used in conjunction with an acoustic lure, which plays bat social calls. The lure elicits a territorial response in the bats and they fly toward the sound and then into the trap. The strings are set so they “absorb” the bat, causing its wings to fold, the bat then falls down into a pouch which has a two layers of plastic, one clear and one cream, which are set up so the bat can fall in but not fly or crawl out. The bat crawls into the space between the plastic as it is rather like being in a roost for them. The bats are then easily extracted by hand – much easier than mist nets so can be done by less experienced people.
Worcestershire Wildlife Trust gratefully acknowledges the financial support given to the project by the Peoples’ Trust for Endangered Species (PTES).
References
Harris, S.& Yelden, D.W. 2008 Mammals of the British Isles: Handbook. 4th Edition. Southampton: The Mammal Society.
Schofield, H. & Morris, C. 2000. Ranging behaviour and habitat preferences of female Bechstein’s bat, Myotis bechsteinii (Kuhl, 1818), in summer.Report held by The Vincent Wildlife Trust.
Sedgeley, J., Hitchcock, J. & Birks, J. 2012. The National Bechstein’s Bat survey and beyond. Worcestershire Record. 32:24-25.
Images

1. Excellent habitat for Bechstein’s Bat with mistnet.

2. Harp trap set in Grafton Wood.
Worcestershire Record | 33 (Nov 2012) page: 13-19 | Worcestershire Biological Records Centre & Worcestershire Recorders
Coleoptera of note in the Kidderminster area, Winter 2011-2012
Alan Brown
Well, there was no let up in my survey recording last winter, all done within two miles of Kidderminster. I started off recording ladybirds and then I noticed certain beetles were still active during the winter months so I recorded those as well. Others I recorded hibernating and come the spring of 2012 I turned to some of our lesser known species, Silphidae, etc. Then in late spring I went back to saproxylic beetles and also began recording some interesting weevil species for the first time. I was quite amazed at how many different species I was recording compared with last year’s finds. My time searching suitable habitats was severely curtailed by the bad weather but I persevered and was rewarded beyond my expectations. To be able to observe these species active at night was really quite fascinating. My only disappointment is the fact that one of the most important habitats, namely hollowed out oak trees are almost non existent in the Kidderminster area: I saw only two. Nonetheless the results of the survey shows there is still a vast array of species to be found here. All the species listed were recorded at night and most have digital photos to match. The approximate body length is given for each species.

Fig. 01. Sphaeriestes castaneus (Salpingidae: local. 3 mm): 2.12.2011. This was the first winter active species that I came across. A predatory species that I only found on Scots Pine. This species appears to hunt alone but is very active and detected on branches looking for prey. Last individual was seen on January 21st.

Fig. 02. Rabocerus gabrieli (Salpingidae: notable B. 4 mm): 10.12.2011. Another predatory species. These were active throughout the winter, seen pairing up in early February and then disappeared soon after. I would sometimes find lone individuals on other types of tree, but usually came across this species on Silver Birch in groups up to a dozen situated near small bark beetle galleries. Last one seen on Feb 8th.

Fig. 03. Phloiophilus edwardsii (Phloiophilidae: notable B. 3 mm): 10.12.2011. A fungus beetle. I found these active throughout the winter months on decayed branches of oak trees, linked to the fungus, Peniophora quercina.Last seen on Feb 24th this species was found at three sites in good numbers around Kidderminster.

Fig. 04. Aplocnemus impressus (Melyridae :notable B. 5 mm) : 20.12.2011. The adult feed on pollen but the larvae are predatory feeding on various bark beetle larvae. Much more metallic than the photo suggests, I found this species hibernating usually in niches on birch trees but sometimes on Pine also.

Fig. 05. Rhizophagus nitidulus (Monotomidae: notable B. 4 mm): 21.1.2012. Another predatory species, I first saw active in numbers very early in the year on decaying Silver Birch trees linked to a colony of Xyloterus lineatus. This beetle is very good at slipping into small cavities. Occasionally I saw this species throughout the spring.

Fig. 06. Dorytomus ictor (Curculionidae: notable B. 5 mm): 8.1.2012. One of the catkin weevils, this species can usually be found active at night on Grey Poplar trees. I usually found this species on the trunks of mature trees very early in the year and others I found in the state of hibernation under Pine bark close by. This species can be seen throughout the year and appears to be doing well in Kidderminster.

Fig. 07. Anthribus nebulosus (Anthribidae: notable B. 3 mm): 22.1.2012. Unlike most other weevils this is one of two weevil species which are predatory, feeding on various scale insects, larvae parasitising ovipositing female scale insects. It is also restricted to Pine trees. I found a single specimen hibernating in a niche on the trunk of a mature Scots Pine. Very difficult to find and very small.

Fig. 08. Anthribus fasciatus (Anthribidae: notable A. 4 mm): 8.3.2012. This is the other predatory weevil which also predates various scale insects restricted to deciduous trees. I found four of these hibernating on an open roadside verge. Two were found in niches on a mature Lime and two others on a nearby Cherry tree. A quite colourful species.

Fig. 09. Hadrobregmus denticollis (Anobiidae: notable B. 6 mm): 8.3.2012. I found one of these behind decaying bark on the same old Cherry tree on the roadside verge. The beetle and larvae feed on dead wood and this species has also been linked to Hawthorn trees.

Fig, 10. Caenopsis fissirostris (Curculionidae:notable B. 6 mm): 7.4.2012. I saw two of these actively investigating some decaying bark near the base of a mature Beech tree at Hurcott wood. Most records for this species come from West Wales so it was a real surprise to see them in the Kidderminster area. I’ve not seen anymore since.

Fig. 11. Acalles ptinoides (Curculionidae:notable B. 3 mm): 7.4.2012. A small species associated with decaying wood on very old Chestnut stumps, I first recorded these in good numbers at Hurcott Wood and also in woodland alongside the Devil’s Spittleful Nature Reserve (NR). They are active throughout the spring and summer.

Fig. 12. Ptinus sexpunctatus (Ptinidae:notable B. 4 mm): 24.4.2012. A spider beetle usually associated with dead wood on deciduous trees, I find these occasionally on decaying Lime trunks and also on an old Cherry tree on a roadside verge.

Fig. 13. Aclypea opaca (Silphidae:notable A. 12 mm): 3.5.2012. I found a single specimen of the sugar beet beetle on the roadside verge opposite some arable fields. This herbivorous species is well known for its taste for sugar beet but also feeds on cereal plants and various seeding grasses. A beautiful species.

Fig. 14. Silpha tristis (Silphidae: local. 17 mm): 27.8.2011. The snail hunter. This is a true predatory species that also feeds on slugs and is rarely found in carrion traps. Also seems to prefer open sandy grassland areas. I found these in good numbers at Springfield Park but also on roadside verges and temporary arable meadows.

Fig. 15. Megatoma undata (Dermestidae:notable B. 6 mm): 23.5.2012. This species is usually found on dead wood feeding on dead insect remains. I found this specimen in open parkland in a niche on a healthy Ash tree next to a pile of decaying willow logs.

Fig. 16. Trichosirocalus barnevillei (Curculionidae:notable B. 2.5 mm):3.5.2012 A herb feeding weevil, I found a good number of these on Narrow Leaved Plantain on sandy, roadside grass verges and temporary arable meadows.
Strophosoma faber (Curculionidae:notable B. 6 mm): 3.5.2012.(no picture). A seed feeding weevil which is also found in sandy, dry habitats. I found these in good numbers on roadside grass verges on Narrow Leaved Plantain and Yarrow.

Fig. 17. Mesosa nebulosa (Cerambycidae RDB 3. 15 mm): 26.5.2012. This Longhorn beetle is an extremely difficult species to find due to its natural camouflage. It is usually found after falling from decaying oak branches in the uppermost reaches of mature oak trees. So I was lucky to find two of these on a decaying oak branch barely five feet from the ground. The secret in this case was to search a stunted, isolated old Oak tree. It was about fifteen foot tall with a wide canopy shading the decaying branches beneath. I found this one at Devil’s Spittleful NR.

Fig. 18. Mycetophagus piceus (Mycetophagidae:local. 4 mm): 27.5.2012. A nocturnal beetle found in various decaying oak situations relating to fungus. I found these on a very old oak bough although no fungus was seen. Quite a colourful species.

Fig. 19. Abdera quadrifasciata (Abderiidae:noteble A. 4 mm): 16.6.2012. I found this species also on decaying oak branches with crust fungi. Active at night on woodland fringe, Devil’s Spittleful NR.

Fig. 20. Scaphisoma boleti (Staphylionidae:notable B. 2 mm): 30.6.2012 [no picture]. Another species linked to decaying wood with fungus, I found two of these on white-rot fungus on a decaying oak log in open situation. Active at night at Devil’s Spittleful NR.

Fig. 21. Ernobius mollis (Anobiidae:local. 5 mm): 22.6.2012. This species is strictly linked to Pine. I found large numbers on a dead, standing Pine tree, I also found them on dead bark on healthy trees. Hurcott Wood.

Fig. 22. Symbiotes latus (Endomychidae:notable B. 5 mm.): 15.7.2012. A fungus beetle linked to fungus on dead wood, I found four of these on decaying heart wood on an oak with white-rot fungus, active at night. I also found these in wood detritus in the bottom of a hollowed-out oak. Confirmed by Paul Whitehead.

Fig. 23. Pediacus depressus (Cucujidae:notable A. 4 mm): 15.7.2012. Similar to Pediacus.dermestoides, but usually the head and pronotum of this species are bright red and the tooth on the pronotal hind-angles is more blunt. A predatory species I found one active at night on a dead standing Pine investigating a beetle gallery. Devil’s Spittleful NR

Fig. 24. Xyleborus dispar (Scolytidae:notable B. 3 mm): 21.7.2012. The European shot-hole borer. Until recently this species was listed as RDB 3, however this species now increasing its range. I found two of these looking over a recently cut, mature oak trunk at night. Its usual preference is for decaying oak related to fungus.

Fig. 25. Platypus cylindrus (Platypodidae:notable B. 5 mm): 21.7.2012. The Oak Pinhole-Borer has also become much more widespread. I saw plenty of these and the small telltale piles of white shavings on the same recently cut oak trunk.

Fig. 26. Odonteus armiger (Bolboceratidae:notable A. 10 mm): 21.7.2012. A scarab beetle which is very seldom seen, I found one female which is rather less spectacular than the male, on a sandy trail on heathland. This species is usually linked to subterranean fungi and also linked to dung. The female has the ridge running across the pronotum which identifies it. On the heath cattle have been grazing there for most of the year. Devil’s Spittleful NR
Acknowledgements
Thanks to John Meiklejohn whose identifications have been really appreciated and also to Paul Whitehead for some of the trickier identifications.
Images
Fig. 01. Sphaeriestes castaneus (Salpingidae: local. 3 mm): 2.12.2011.
Fig. 02. Rabocerus gabrieli (Salpingidae: notable B. 4 mm): 10.12.2011.
Fig. 03. Phloiophilus edwardsii (Phloiophilidae: notable B. 3 mm): 10.12.2011.
Fig. 04. Aplocnemus impressus (Melyridae :notable B. 5 mm) : 20.12.2011.
Fig. 05. Rhizophagus nitidulus (Monotomidae: notable B. 4 mm) :21.1.2012.
Fig. 06. Dorytomus ictor (Curculionidae: notable B. 5 mm): 8.1.2012.
Fig. 07. Anthribus nebulosus (Anthribidae: notable B. 3 mm): 22.1.2012.
Fig. 08. Anthribus fasciatus (Anthribidae: notable A. 4 mm): 8.3.2012.
Fig. 09. Hadrobregmus denticollis (Anobiidae: notable B. 6 mm): 8.3.2012.
Fig. 10. Caenopsis fissirostris (Curculionidae:notable B. 6 mm): 7.4.2012.
Fig. 11. Acalles ptinoides (Curculionidae:notable B. 3 mm): 7.4.2012.
Fig. 12. Ptinus sexpunctatus (Ptinidae:notable B. 4 mm): 24.4.2012.
Fig. 13. Aclypea opaca (Silphidae:notable A. 12 mm): 3.5.2012.
Fig. 14. Silpha tristis (Silphidae: local. 17 mm): 27.8.2011.
Fig. 15. Megatoma undata (Dermestidae:notable B. 6 mm): 23.5.2012.
Fig. 16. Trichosirocalus barnevillei (Curculionidae:notable B. 2.5 mm): 3.5.2012.
Fig. 17. Mesosa nebulosa (Cerambycidae RDB 3. 15 mm): 26.5.2012.
Fig. 18. Mycetophagus piceus (Mycetophagidae:local. 4 mm): 27.5.2012
Fig. 19. Abdera quadrifasciata (Abderiidae:noteble A. 4 mm): 16.6.2012.
Fig. 20. Scaphisoma boleti (Staphylionidae:notable B. 2 mm): 30.6.2012.
Fig. 21. Ernobius mollis (Anobiidae:local. 5 mm): 22.6.2012.
Fig. 22. Symbiotes latus (Endomychidae:notable B. 5 mm.): 15.7.2012.
Fig. 23. Pediacus depressus (Cucujidae:notable A. 4 mm): 15.7.2012.
Fig. 24. Xyleborus dispar (Scolytidae:notable B. 3 mm): 21.7.2012.
Fig. 25. Platypus cylindrus (Platypodidae:notable B. 5 mm): 21.7.2012.
Fig. 26. Odonteus armiger (Bolboceratidae:notable A. 10 mm): 21.7.2012.
Worcestershire Record | 33 (Nov 2012) page: 11 | Worcestershire Biological Records Centre & Worcestershire Recorders
Haeterius ferrugineus rediscovered after 92 years absence
Alan Brown
On June 10th, 2012, a single specimen of the ant-associated Hister Beetle Haeterius ferrugineus was found at night in the nest of the slave maker ant Formica sanguinea in the Kidderminster area. This small (1.5-2.0 mm long) Hister Beetle, which is classed as being extremely rare, was last recorded on the Isle of Wight in 1920.
Description: Very small, about 2mm long, oval, bright orange-red in appearance with beautifully sculptured appendages and pronotal edges (Fig.1.).
Habitat description: Generally in the ant nests of Formica sanguinea and Formica fusca. In this case under a piece of oak wood in an open, sandy grassland terrain on a slightly south-facing slope. The beetle itself was observed moving in and out of a small section of white-rot fungus attached to the underside of the wood close to the ants’ larval chamber. A second ant specimen taken from the nest was identified as Lasius brunneus (the Tree Ant) but this may have been an accidental occurrence and will need further investigation before confirmation can be ascertained. The site is largely undisturbed and sun-exposed with a warm micro-climate which is perfect for the ants which is there in abundance.
History: Notes from Donisthorpe (1927).
“ … Haeterius ferrugineus , whose normal hosts are Formica fusca and Formica sanguinea, with fusca as slaves, was first captured in this country by E.W. Johnson,who found it with F. fusca and A. (C.) flavus at Hampstead in 1848, and subsequent years. In 1862 Power took it with F. sanguinea at Weybridge, and Douglas and Stott found it with the same ant near Croydon. Since then, so far as I know, it did not occur again until 1909 when Bedwell found it sparingly with F. fusca at Box Hill, where I subsequently took it with him. A single specimen which Stott found in an ants’ nest at Luccombe Chine, Isle of Wight, in 1920, is the only other record known to me. The host species was unfortunately not noted.”
Habits: This species is much more common on the continent than it is with us and is also found with far more species of ants. According to Donisthorpe (1927) it has been kept in captivity by Wasmann, Janet and others. Wasmann kept specimens alive in these nests for between 2-4 years and noticed their copulation, treatment by hosts, etc. The larvae are still unknown. The ants frequently lick the beetle, carry it about and play with it, sometimes very roughly. The beetle itself feeds on dead and wounded ants, ant larvae, pupae, and mites etc. Both Wasmann and Viehmeyer once saw the beetle being fed by its hosts although this was a very exceptional instance.
A second sighting of Haeterius ferrugineus in Worcestershire?
Two months after the initial find, on the night of 15th August 2012 I was busy doing a night hunt for Silpha beetles in the same area of grassland where I had seen the original Haeterius ferrugineus beetle. I ended up back at the same piece of wood and decided out of curiosity to have another look. The Formica sanguinea nest was larger than I had remembered it, with a large pile of pupa in the centre. For a few seconds I saw nothing but ants, but then from beneath the pupae I caught sight of the familiar orange-red glint of a Hister Beetle. Then from under more pupae another appeared and another, four all told. I couldn’t believe my luck. I waited until both Hister Beetles and ants had withdrawn deeper into the ground and then carefully re-laid the wood and left them to it. The site itself I have decided to keep secret but there is no reason not to believe that this species may exist in other sandy grassland sites around Kidderminster and perhaps also around the Wyre Forest where large colonies of the ant have been recorded. Could the beetle itself be under-recorded? Possibly, as all subterranean species are difficult to find and one relies to some degree of good fortune. Perhaps by laying out pieces of wood in suitable locations the species could be found more often, as the ant involved does seem to like building nests against decaying wood. I have also found the ant under Limestone slabs too, so possibly suggesting another way of finding the beetle without causing too much disruption to the ants nest.
Acknowledgements: Special thanks should go to Cristoph Benisch who kindly allowed me to use a photo from his excellent Coleoptera website www.kerbtier.de. Also thanks to Rosemary Winnall for supplying the literature from Donisthorpe (1927).
Editor’s Note: Genus name formerly spelt Hetaerius. The species is extremely rare or unrecorded: ‘Very local in S England (S Hampshire, Isle of Wight, Surrey and Middlesex); Rare’. (Duff 2012).
References
Benisch, Christoph. 2007-2012 www.kerbtier.de. The beetle fauna of Germany.
Donisthorpe, H. St. J. K. 1927 The guests of British ants, their habits and life-histories. Routledge, London.
Duff, A.G. 2012. Beetles of Britain and Ireland. Volume 1. Duff:West Runton, Norfolk.
Image

Fig. 1. Haeterius ferrugineus Picture copyright Christoph Benisch www.kerbtier.de.
Worcestershire Record | 33 (Nov 2012) page: 12 | Worcestershire Biological Records Centre & Worcestershire Recorders
Vine Weevil Otiorhynchus armadillo in Kidderminster
Alan Brown.
There’s an Armadillo loose in Worcestershire …
On the 26th of April 2012 a single specimen of the invasive Vine Weevil Otiorhynchus armadillo was found on Ivy at Springfield Park, Kidderminster. Close by, a large colony was found in the gardens of a new housing estate overlooking Puxton Marsh. Quite how long they have been established here is unknown but in South West London where they were first discovered in 1998 the species is now reaching pest proportions and it has officially become the most common weevil there.
In Kidderminster the species seems to have a distinct preference for Spindle Tree leaves, Rosacea plants and Ivy. Other plant species thought to be at risk are Cherry Laurel, Ornamental Willow and Viburnum. It is without doubt a very distinct weevil, the females reaching 15mm in length. The species seems to be mainly nocturnal and the larvae feed on various plant roots whilst the adults feed on the leaves. I have seen it in three separate locations now,: Springfield Park, Crossley Park (feeding on Dog rose and Brambles) and on the Kidderminster to Hagley road (found on Primrose). Although wingless this species seems to have no trouble increasing its range, either that or they have been here for some time. The females can lay up to 1500 eggs with both adults and larvae able to survive our colder winters. It has also been seen in Cardiff, Edinburgh and Warwickshire so it looks as though this invasive species is here to stay.
Acknowledgements
Thanks to Paul Whitehead for identifying the species and to John Meiklejohn for his help.
Reference
Lane, Steve, 2009.Otiorhynchus armadillo (Rossi) in Coventry, Warwickshire (VC 38). Beetle News 1 (3):3.
Image

Fig.1. Vine Weevil Otiorhynchus armadillo. Picture Alan Brown
Worcestershire Record | 33 (Nov 2012) page: 20-22 | Worcestershire Biological Records Centre & Worcestershire Recorders
Clitostethus arcuatus (Rossi, 1794) (Col., Coccinellidae) breeding in the Kidderminster area of Worcestershire: overwintering strategies and breeding biology
Alan Brown & P.F. Whitehead
AB: 54 Findon Street, Kidderminster, Worcestershire DY10 1PU. abb5412@yahoo.co.uk
PFW: Paul. F. Whitehead, Moor Leys, Little Comberton, Pershore, Worcestershire WR10 3EH. paul@thewhiteheads.eu
Introduction
Clitostethus arcuatus (Rossi, 1794) (Fig. 6.) was for long one of the rarer British coccinellids; up to now there have been no published Worcestershire records although it has occurred elsewhere in the English midlands. In Oxfordshire it was recorded in 1915, 1979 and 1980 (Mills, 1981; Shirt, 1987). The only other record is an 1872 one from Shenton Hall near Market Bosworth in Leicestershire (Fowler, 1887-1891). In October 2011 AB found C. arcuatus at Kidderminster and subsequently went on to make further significant discoveries which form the subject of this paper. As a result C. arcuatus has now been found at four sites in Kidderminster: Springfield Park, Crossley Park, a town garden and Hurcott Wood.
The Kidderminster sites
Springfield Park (SO8378)
An adult C. arcuatus found overwintering on the underside of a leaf of a holly Ilex aquifolium L. at Springfield Park on 6th October 2011 constitutes the first known Worcestershire record. During October a further eight examples were found under holly leaves always on the sheltered side of the tree facing towards overarching mixed woodland (Fig. 1). The beetles remained on the holly foliage until a warm spell on 12th March 2012 and showed considerable fidelity to it. When a falling oak bough dislodged an adult on 2nd February 2012 it returned to the same holly branch within 48 hours. It was observed at night that the holly leaves remained dry whilst the foliage of other plants became covered in condensation. During their time on holly the beetles survived air temperatures of -9oC.
Mills (1981) recorded C. arcuatus associated with whiteflies (Hemiptera:Aleyrodidae) on the ornamental shrub Viburnum tinus L. in Oxfordshire and a watch was kept both for whiteflies and for any developing populations of foliicolous mites in Springfield Park which might have provided an additional food resource. This proved unproductive but during March whitefly Aleyrodes proletella (L.) began to appear on both Wood Avens Geum urbanum L. and Greater Celandine Chelidonium majus L.) only three metres away from the C. arcuatus overwintering sites. The departure of the beetles from the holly took place by the third week of March as whiteflies began to appear and only one beetle remained on the holly on 1st April 2012. Despite the proximity of whitefly host-plants no C. arcuatuswere found on them and it is presumed that they were elsewhere or overlooked; not difficult for a beetle only 1.5 mm or so in length.
Crossley Park (SO8277)
This is a built-up retail park in urban Kidderminster. The site was first visited on 4th July 2012 when an adult C. arcuatus was found amongst a proliferation of whitefly A. proletella on Chelidonium majus at the base of a Grey Alder Alnus incana (L.) Moench (Fig. 2). This site was monitored daily. On 30th July 2012 two adult C. arcuatus and five larvae were observed amongst the whitefly (Fig. 3) with the first pupa noted on 21st August 2012 (Fig. 4).
Kidderminster town (SO8376)
On 4th July 2012 a single C. arcuatus was found under a leaf of Chelidonium majus at a site in the town centre (Fig. 5). These plants were destroyed shortly afterwards.
Hurcott Wood (SO8577)
On 28th November 2011 a single C.arcuatus was found on the underside of a holly leaf in this block of ancient woodland. The holly was situated at the edge of the woodland and although there was dense ivy nearby no C. arcuatus were found on it. It is therefore presumed that the use of holly by the beetle is preferential. However, a single C. arcuatus was found on ivy Hedera helix L. at Springfield Park on 8th February 2012.
There were further developments during the summer of 2012 when three adult, one larval and two pupal C. arcuatus was found amongst whitefly Aleyrodes lonicerae Koch, 1857 on honeysuckle Lonicera periclymenum L. scrambling up an oak tree close by.
Discussion
These are the first breeding records of C. arcuatus in Worcestershire and the first evidence of it breeding on Chelidonium majus in Britain as a predator of Aleyrodes spp.. Chelidonium is cited by Evans (2007) as a recognised host-plant genus for Aleyrodes proletella (L.). Of interest in Worcestershire is the use of holly for overwintering, ideally close to the host plants, and the fidelity demonstrated for that plant. This is not the first occasion that holly has been cited as an overwintering plant of C. arcuatusin Britain. Welch (1999) refers to a specimen beaten from holly under oak at Bookham Common, Surrey on 29th February 1993. Welch also cites specimens from sylvicultural contexts in plantations of Scots Pine Pinus sylvestris L. in Hampshire and Norfolk in June 1995.
The Kidderminster sites occupy a linear ribbon no more than three kilometres in length, amongst which are other woodlands and beyond which are various satellite woodlands almost certainly occupied by C. arcuatus. It is clear that C. arcuatus is now well-established as a whitefly predator in the urban, suburban, semi-natural and ancient wooded landscapes of Kidderminster and probably elsewhere in the region. Neither of us has yet found C. arcuatus amongst A. proletella on cultivated brassicas implying that the relationship of breeding sites to successful wintering sites and microclimate may all be significant. In particular the winter climate with particular reference to humidity may prove to be determining factors for the species success.
Although C. arcuatus has demonstrated an ability to accept urban areas and may therefore now be regarded as a facultative synanthrope in much the same way as Rhyzobius chrysomeloides (Herbst, 1792) (Whitehead, 2008), another southern coccinellid, it remains scarce. We recommend that it should now be accorded Nationally Scarce status rather than its original Red Data Book-listed status. It is, however, a species of conservation significance wherever it occurs at its north-western range edge. Springfield Park is managed by Wyre Forest District Council and they should be made generally aware of their interesting resident; Mr Paul Allen the Wyre Forest District Council Countryside Officer has already been informed of the biological significance of Chelidonium and has excluded it from clearance schemes at Springfield Park.
These findings interlace with those of Springate & Arnold (2011) who recently found C. arcuatus breeding on Wild Cabbage Brassica oleracea L. and honeysuckle Lonicera periclymenum L. in Kent and on Welsh Poppy Meconopsis cambrica (L.) in Greater Manchester. Clitostethus arcuatus is a thermophilous species widely distributed in central and southern Europe. Coccinellids can be sensitive indicators of ameliorated climate and microclimate (Thomas, 2010; Whitehead, 2007) and all of these observations point to a recently expanded and invigorated British population of C. arcuatus.
References
Evans, G.A., 2007. The whiteflies (Hemiptera:Aleyrodidae) of the World and their host plants and natural enemies. http://keys.lucidcentral.org/keys/v3/whitefly/PDF_PwP%20ETC/world-whitefly-catalog-Evans.pdf
Fowler, W.W., 1887-1891. The Coleoptera of the British Islands 1-5.London, L. Reeve & Co.
Mills, N.J., 1981. Essential and alternative foods for British Coccinellidae (Coleoptera). Entomologist’s Gazette 32:197-202.
Shirt, D.B. (ed.), 1987. British Red Data Books 2. Insects. Peterborough, Nature Conservancy Council.
Springate S. & Arnold S.E.J., 2011. New vice-county records of Clitostethus arcuatus (Rossi) (Col., Coccinellidae) and a new association with wild cabbage. British Journal of Entomology and Natural History 24:224-225.
Thomas, J., 2010. Scymnus interruptus (Goeze) (Coccinellidae) in Cumbria. The Coleopterist 19:33-34.
Welch, R.C., 1999. Clitostethus arcuatus (Rossi) (Col., Coccinellidae) from malaise traps in Northamptonshire, Norfolk and Hampshire. Entomologist’s Record and Journal of Variation 111:133-134.
Whitehead, P.F., 2007. Invertebrates on Mexican Orange Choisya ternataHumboldt in English gardens with observations on Rhyzobius chrysomeloides (Herbst, 1792) (Col., Coccinellidae) and other coccinellids. Entomologist’s Monthly Magazine 143:7-10.
Whitehead, P.F., 2008. Rhyzobius chrysomeloides (Herbst, 1792) (Col., Coccinellidae) new to north-west England. Entomologist’s Monthly Magazine144:68.
Images

Fig. 1. Springfield Park Kidderminster December 2011. Overwintering site of Clitostethus arcuatus in holly at left. Picture Alan Brown.

Fig. 2. Crossley Park Kidderminster August 2012. Site of Clitostethus arcuatus on Chelidonium majus at base of Grey Alder. Picture Alan Brown.

Fig. 3. Crossley Park Kidderminster 30 July 2012. Larva of Clitostethus arcuatus on Chelidonium majus. Picture Alan Brown.

Fig. 4. Crossley Park Kidderminster 21 August 2012. Pupa of Clitostethus arcuatus on Chelidonium majus. Note the total absence of whitefly. Picture Alan Brown.

Fig. 5. Kidderminster town garden 4 July 2012. Adult Clitostethus arcuatusfound on Chelidonium majus. Picture Alan Brown.

Fig. 6. Clitostethus arcuatus overwintering on a Holly leaf at Springfield Park. Picture Alan Brown.
Worcestershire Record | 33 (Nov 2012) page: 91-94 | Worcestershire Biological Records Centre & Worcestershire Recorders
Botanical Notes, Ipsley Alders, Redditch, 4th August 2012
J.J. Day
Ipsley Alders’ ecological interests were first recognised in the mid 1960s when Fred Fincher came across the site during his Ecological Survey for the Redditch Development Corporation. It has been reasonably well covered by survey work since, with fairly intensive botanical surveys in most subsequent decades.
Its principal interests lie in its soils, which include fen peat and the associated plant communities. Habitats and communities include open water, swamp, fen, wet grassland, unimproved mesotrophic grassland, alder carr and plantation woodland, all of which were sampled during the course of the field meeting. The flora is diverse and in the period 1966-2011 267 species were recorded.
The current survey recorded a total of 195 species of which 166 were of species already known from the site. This represents a 62% rediscovery rate. This is reasonable, given the extent of the survey and the time of year. Perhaps only 25-35% of the area was covered in the current survey and it largely avoided the peripheral woodland zones.
| Ipsley Alders | No of Species | |
| pre-2012 | 267 | |
| 4th August 2012 | 195 | |
| Common species | 166 | 62% |
| Species not refound | 101 | |
| New species 2012 | 29 | 15% (day list) |
| Total | 296 |
Table 1. Species numbers at Ipsley Alders
The jewel in Ipsley’s crown is its diverse calcareous fen community. The majority of the wetland is now dominated by a M22 Juncus subnodulosus-Cirsium palustre fen-meadow community. This is by a considerable extent the largest such fen community in Worcestershire. It has spread noticeably during the course of the past 30 years. Other scarce species of the fen, which occur here in some abundance, include Fen Bedstraw, Galium uliginosum and Brown Bent, Agrostis canina. The latter species had surprisingly not been recorded at Ipsley before. I suspect it was likely present in small numbers but overlooked. Within the fen there are extensive stands of Hemp Agrimony, Eupatoria cannabinum, which probably represents a community or sub-community not circumscribed within the NVC (National Vegetational Classification). Unfortunately for the entomologists, the peak flowering period of this rich nectar source was well over.
As Ipsley is a well recorded since the 1960s, it is worth comparing this survey’s results with past records. This enables insights into the ecological processes on site. Given the extent of the survey, any gains in species are potentially more significant than losses. For instance, amongst the 101 species not rediscovered, there is a vernal group of 24 species, which would have been missed in August.
| Habitat | pre-2012 only | 2012 only |
| Grassland | 32 | 3 |
| Ruderal | 9 | |
| Wetland | 4 | 3 |
| Open Water | 15 | 4 |
| Woodland | 41 | 19 |
| Total | 101 | 29 |
Table 2. Habitats of Species either novel to the site or not rediscovered
| Dispersal Agent | No. | % | Alien | Native |
| Wind | 7 | 24% | 2 | 5 |
| Bird food -berries | 9 | 31% | 8 | 1 |
| Animal bodies incl. human | 10 | 34% | 1 | 9 |
| Animal husbandry | 2 | 7% | 0 | 2 |
| Deliberate introduction | 1 | 3% | 1 | 0 |
| Total | 29 | 12 | 17 |
Table 3. Status and dispersal mechanisms of the species new to the site
Of the 29 new species to the site, 12 (40%) are either alien to Great Britain or are not native locally. The high proportion of alien additions is not unusual in habitats close to urban centres. Most of the additions into native habitats, as occurs here, tend to be either water plants or woodland species.
Grassland plants represent a large proportion of those not relocated. Even allowing for seasonality and grazing regime, the number and quality of the grassland species not rediscovered e.g. Sawwort Serratula tinctoria, Devils Bit Scabious Succisia pratensis and Sneezewort Achillea ptarmica suggests a possible deterioration in the grassland resource. Whilst some of the losses may be due to the spread of fen communities, the species loss is marked enough to indicate that a more thorough investigation of the situation should be undertaken.
The wetland/fen species assemblage seems fairly stable. The proportion of species acquired compared to those not rediscovered suggests a fairly stable assemblage. The acquisitions may indicate a strengthening of this resource.
The proportion of pond losses is very high. The pond was not surveyed with a grapnel so the losses may not be quite as dramatic as suggested. Despite this there have been significant losses but also moderate gains. Species turnover rate in ponds is often high. Despite this, the species variation here is much higher than I would anticipate for a pool of this size with a restricted and mainly unimproved catchment. Once again the figures suggest that a more thorough investigation of the losses here is warranted.
Woodland was the least surveyed habitat on the day in terms of time and area, so the high number of not found species is considered not significant. This is in marked contrast to a high acquisition rate of new species which should be considered a “real” phenomenon. Most of this group are genuine immigrants. Many of the species are plants of secondary woodlands and their acquisition here can be considered to be natural species accumulation as the peripheral woodlands (mainly 30-40 years old) age. Secondary woodland species tend by definition to be good dispersers. The majority take a ride on animals, including human footwear and clothing.
Species accumulation is however not confined to secondary woodland sites. I have witnessed high acquisition rates of woodland species in urban and urban fringe woods in Worcestershire on a number of studied woods. The phenomenon seems to relate to urban landscape management in the second half of the 20th century. The explosion in horticulture plus urban landscaping, particularly associated with new builds, over this period has brought considerable numbers of woodland species, very many of them alien to the British Isles into our built environments. Each generation of landscape architects has produced a new wave of fashionable species. Redditch exemplifies this phenomenon in extremis. The landscape architects of the Development Corporation inadvertently created a vast ecological experiment with the millions of alien woodland plants they introduced into a lowland farmed landscape. The accumulation of woodland species at Ipsley Alders is a part of the legacy. The end point of this massive inoculation is yet to be determined; we still lie close to the beginning of the experimental time line.
A common feature of this woodland species accumulation is the high number of berry producing plants involved. The vast majority of the dispersal here is by birds. Nine of the new species (31%) are berry producers and mostly alien. Berries for birds may be popular but it is having a knock on effect on our native woodlands. Those close to urban centres are most at risk but it is quite possible that in a couple of hundred years that the species assemblage of all Worcestershire woodlands may be quite different. I suspect that cotoneasters will be found in most Worcestershire woodlands before too long.
The most surprising addition was Thin-spiked Wood Sedge, Carex strigosawhich is almost confined to ancient woodland sites in the county. It was growing in an area of secondary woodland by a path and judging from its size was a relatively recent colonist. There are a couple of locations in ancient woods in the Redditch area but there appears no natural mechanism for dispersal to Ipsley Alders. With no sign of recent disturbance (long buried seed) my best suggestion is a conservation workparty who had picked up seed on boots or tools at a Wildlife Trust ancient woodland reserve to be deposited here inadvertently.
The grassland acquisitions are likely to relate to animal husbandry. Yellow Rattle, Rhinanthus minor and Meadow Barley, Hordeum secalinum have most probably entered the site as hay, stock feed. Indeed in the case of the large heavy seeded Yellow Rattle it was the most significant dispersal agent in traditional farming.
Only one species, Greater Spearwort, Ranunculus lingua was considered a deliberate introduction. It is in my experience always a deliberate introduction. Some folk seem unable to throw aquatic species into the compost when clearing out ponds, always seemingly to prefer to give them a chance in the wild. It’s amazing just how quickly newly dug pools even in remote areas can acquire alien water plants.
Worcestershire Record | 33 (Nov 2012) page: 91-94 | Worcestershire Biological Records Centre & Worcestershire Recorders
Worcestershire Record | 33 (Nov 2012) page: 89-91 | Worcestershire Biological Records Centre & Worcestershire Recorders
Botanical notes and records, Slade Wood, Goom’s Hill, Abbots Morton,7thJuly 2012
J.J. Day
Slade is large and predominately ancient semi-natural woodland. It is mainly situated on the heavy clays of the Mercian Mudstones, with some gravels in down slope lenses. The southern parts lie along the calcareous Rhaetic scarp slope. Consequently, soil conditions vary from calcareous to mildly acidic and from freely draining to waterlogged. This has created the conditions for the development of a rich flora, with a total of 241 species recorded from the site up until 2011.
During the field meeting, a total of 168 species were recorded. Of these, 161 were found within Slade Wood and of these 11 species and one subspecies were new. This is about a 62% refind rate, which is a fairly good return given that perhaps only a third of the woodland area was covered during the day. Given the level of coverage, it is more pertinent to examine the novel records rather than potential losses.
Six of the gains were woodland plants and four of these are ancient woodland indicator species: a Tufted Hair-grass, Deschampsia parviflora;Wood Spurge; Euphorbia amygdaloides ssp amygdaloides, Beautiful St John’s Wort, Hypericum pulchrum and Butterfly Orchid, Platanthera chlorantha. This suggests that this large site has not yet been exhaustively surveyed.
Three of the gains were alien species. The accumulation of alien taxa has been a general trend through Worcestershire’s woodlands over the last 40 years. The rate appears to be increasing and seems in large part to be related to human population density. Here, one species, the cultivated water-lily is no doubt a deliberate introduction. The Horse Chestnut sapling appeared to be natural spread but the origins of the Variegated Yellow Archangel is uncertain. The latter species was dominating an area of about 500sqm on the southern ridge. Its spread suggests an aggressive invader on the site.
The pond in the wood is about 20 years old. It has experienced a high turnover in aquatic species. Since 2001, it has apparently lost two native pondweeds, Potamogeton natans and P berchtoldii, gained a cultivated water-lily and the native Water-plantain, and retained the aliens, Curly Waterweed (dominant) and White-water lily.
The most curious find of the day was a small colony of Herb Robert Geranium robertianum of a form green in all its parts, including its petals. It appeared to be a plant totally devoid of anthocyanin pigments, which is suggestive of a possible gene mutation (Fig.1.).
| Slade Wood 07/07/2012 | Compiled by J.J.Day |
| records in bold italics new for site | |
| Acer campestre | Field Maple |
| Aesculus hippocastanum | Horse-chestnut |
| Agrimonia eupatoria | Agrimony |
| Agrostis canina sens.str. | Brown Bent |
| Agrostis capillaris | Common Bent |
| Agrostis stolonifera | Creeping Bent |
| Ajuga reptans | Bugle |
| Alisma plantago-aquatica | Water-plantain |
| Alliaria petiolata | Garlic Mustard |
| Alopecurus pratensis | Meadow Foxtail |
| Anemone nemorosa | Wood Anemone |
| Angelica sylvestris | Wild Angelica |
| Anthriscus sylvestris | Cow Parsley |
| Arctium minus ssp. minus | Lesser Burdock |
| Arrhenatherum elatius | False Oat-grass |
| Arum maculatum | Lords-and-ladies |
| Athyrium filix-femina | Lady Fern |
| Betonica officinalis | Betony |
| Betula pendula | Silver Birch |
| Brachypodium sylvaticum | False-brome |
| Bromopsis ramosa | Hairy Brome |
| Calamagrostis epigejos | Wood Small-reed |
| Callitriche stagnalis sens.str. | Common Water-starwort |
| Cardamine flexuosa | Wavy Bitter-cress |
| Carex acutiformis | Lesser Pond-sedge |
| Carex flacca | Glaucous Sedge |
| Carex otrubae | False Fox-sedge |
| Carex pendula | Pendulus Sedge |
| Carex remota | Remote Sedge |
| Carex sylvatica | Wood-sedge |
| Cerastium fontanum ssp. vulgare | Common Mouse-ear |
| Circaea lutetiana | Enchanter’s-nightshade |
| Cirsium arvense | Creeping Thistle |
| Cirsium palustre | Marsh Thistle |
| Cirsium vulgare | Spear Thistle |
| Cornus sanguinea | Dogwood |
| Corylus avellana | Hazel |
| Crataegus laevigata | Midland Hawthorn |
| Crataegus monogyna | Hawthorn |
| Cynosurus cristatus | Crested Dog’s-tail |
| Dactylis glomerata | Cock’s-foot |
| Dactylorhiza fuchsii | Common Spotted-orchid |
| Deschampsia cespitosa ssp. parviflora | Tufted Hair-grass |
| Dryopteris dilatata | Broad Buckler-fern |
| Dryopteris filix-mas | Common Male Fern |
| Dryopteris x complexa | Hybrid Male & Scaly Fern |
| Epilobium hirsutum | Great Willowherb |
| Epilobium montanum | Broad-leaved Willowherb |
| Euphorbia amygdaloides ssp. amygdaloides | Wood Spurge |
| Fagus sylvatica | Beech |
| Festuca rubra sens.str. | Red Fescue |
| Filipendula ulmaria | Meadowsweet |
| Fragaria vesca | Wild Strawberry |
| Fraxinus excelsior | Ash |
| Galeopsis tetrahit agg. | Common Hemp-nettle |
| Galium aparine | Cleavers |
| Galium odoratum | Woodruff |
| Galium palustre | Common Marsh-bedstraw |
| Geranium robertianum | Herb-robert |
| Geum urbanum | Herb Bennet |
| Glechoma hederacea | Ground-ivy |
| Glyceria fluitans | Floating Sweet-grass |
| Hedera helix ssp. helix | Common Ivy |
| Heracleum sphondylium | Hogweed |
| Holcus lanatus | Yorkshire-fog |
| Holcus mollis | Creeping Soft-grass |
| Hyacinthoides non-scripta | Bluebell |
| Hypericum hirsutum | Hairy St. John’s-wort |
| Hypericum pulchrum | Slender St. John’s-wort |
| Hypericum tetrapterum | Square-stalked St. John’s-wort |
| Ilex aquifolium | Holly |
| Iris pseudacorus | Yellow Iris |
| Juncus articulatus | Jointed Rush |
| Juncus conglomeratus | Compact Rush |
| Juncus effusus | Soft Rush |
| Lagarosiphon major | Curly Waterweed |
| Lamiastrum galeobdolon ssp. argentatum | Variegated Yellow Archangel |
| Lapsana communis | Nipplewort |
| Lemna minor | Common Duckweed |
| Ligustrum vulgare | Wild Privet |
| Lolium perenne | Perennial Rye-grass |
| Lonicera periclymenum | Honeysuckle |
| Lotus pedunculatus | Large Bird’s-foot-trefoil |
| Luzula multiflora ssp. multiflora | Heath Wood-rush |
| Luzula pilosa | Hairy Wood-rush |
| Lysimachia nummularia | Creeping-Jenny |
| Malus pumila | Apple |
| Melica uniflora | Wood Melick |
| Mentha arvensis | Corn Mint |
| Mercurialis perennis | Dog’s Mercury |
| Milium effusum | Wood Millet |
| Myosotis scorpioides | Water Forget-me-not |
| Nymphaea alba ssp. alba | White Water-lily |
| Nymphaea marliacea | Water-lily |
| Oxalis acetosella | Wood-sorrel |
| Phleum pratense sens.str. | Timothy |
| Pinus sylvestris | Scots Pine |
| Plantago major ssp. major | Greater Plantain |
| Platanthera chlorantha | Greater Butterfly-orchid |
| Poa annua | Annual Meadow-grass |
| Poa nemoralis | Wood Meadow-grass |
| Poa trivialis | Rough Meadow-grass |
| Polygonum aviculare sens.str. | Knotgrass |
| Polystichum aculeatum | Hard Shield-fern |
| Polystichum setiferum | Soft Shield-fern |
| Populus tremula | Aspen |
| Potamogeton natans | Broad-leaved Pondweed |
| Potentilla erecta ssp. erecta | Tormentil |
| Potentilla reptans | Creeping Cinquefoil |
| Potentilla sterilis | Barren Strawberry |
| Primula vulgaris | Primrose |
| Prunella vulgaris | Selfheal |
| Prunus avium | Wild Cherry |
| Prunus domestica | Wild Plum |
| Prunus spinosa s.l. | Blackthorn |
| Pteridium aquilinum | Bracken |
| Quercus robur | Pedunculate Oak |
| Quercus x rosacea | Hybrid Oak |
| Ranunculus acris | Meadow Buttercup |
| Ranunculus flammula ssp. flammula | Lesser Spearwort |
| Ranunculus repens | Creeping Buttercup |
| Rhamnus cathartica | Buckthorn |
| Rosa arvensis | Field Rose |
| Rosa canina agg. | Dog Rose |
| Rubus fruticosus agg. | Bramble |
| Rumex obtusifolius | Broad-leaved Dock |
| Rumex sanguineus | Wood Dock |
| Salix caprea | Goat Willow |
| Salix cinerea ssp. oleifolia | Grey Willow |
| Sambucus nigra | Elder |
| Sanicula europaea | Sanicle |
| Schedonorus giganteus | Giant Fescue |
| Scrophularia nodosa | Common Figwort |
| Senecio aquaticus | Marsh Ragwort |
| Senecio jacobaea | Common Ragwort |
| Silene flos-cuculi | Ragged Robin |
| Stachys sylvatica | Hedge Woundwort |
| Stellaria holostea | Greater Stitchwort |
| Succisa pratensis | Devil’s-bit Scabious |
| Tamus communis | Black Bryony |
| Taraxacum sp. | Dandelion |
| Teucrium scorodonia | Wood Sage |
| Tilia cordata | Small-leaved Lime |
| Tilia platyphyllos | Large-leaved Lime |
| Tilia x vulgaris | Lime |
| Trifolium repens | White Clover |
| Triticum aestivum | Bread Wheat |
| Ulex europaeus | Gorse |
| Ulmus glabra | Wych Elm |
| Urtica dioica | Common Nettle |
| Veronica beccabunga | Brooklime |
| Veronica chamaedrys | Germander Speedwell |
| Veronica montana | Wood Speedwell |
| Veronica officinalis | Heath Speedwell |
| Veronica serpyllifolia ssp. serpyllifolia | Thyme-leaved Speedwell |
| Viburnum lantana | Wayfaring-tree |
| Viburnum opulus | Guelder-rose |
| Vicia sativa ssp. segetalis | Common Vetch |
| Vicia sepium | Bush Vetch |
| Viola riviniana | Common Dog-violet |
| Species from adjoining Grasslands | |
| Epilobium ciliatum | American Willowherb |
| Geranium dissectum | Cut-leaved Crane’s-bill |
| Juncus inflexus | Hard Rush |
| Lathyrus nissolia | Grass Vetchling |
| Persicaria maculosa | Redshank |
| Rumex crispus | Curled Dock |
| Stellaria graminea | Lesser Stitchwort |
| Vicia tetrasperma | Smooth Tare |
Image
 Fig. 1. Herb Robert Geranium robertianum green in all parts. Rosemary Winnall.
Fig. 1. Herb Robert Geranium robertianum green in all parts. Rosemary Winnall.
Worcestershire Record | 33 (Nov 2012) page: 86-88 | Worcestershire Biological Records Centre & Worcestershire Recorders
Botanical notes and records: The Red House Farm, Pillows Green, Eldersfield, 9th June 2012
J.J. Day
The Red House site consists of the farmhouse and yard plus grounds and gardens, a small meadow and a strip of arable land. There are many introductions and formal parts but much of the area is allowed to regenerate naturally. This presented a conundrum for botanical recorders: which species were worth noting in such a setting?
The gardens are semi-formal but there are wild areas and zones where the vegetation is allowed to self sow, particularly around the former yard. This has created a mixed assemblage of native and alien species exhibiting various degrees of naturalisation. The small colony of Yellow Figwort, Scrophularia vernalis growing in a shady corner, which the owner reported as origin unknown and new is a good record for Worcestershire. It has only been seen in the county once before at Hewell Grange and last noticed there during the First World War.
Old spikes of the Ivy Broomrape, Orobanche hederacea were located by the southeast corner of the farmhouse. Botanists have had no problem recording this, which has persisted here since 1995, although it is very likely an inadvertent hortal introduction. The uncommon native species Small Toadflax Chaenorhinum minus (c30 plants) and Knotted Hedge parsley, Torilis nodosa (one plant) were both found in the courtyard. All the botanists seemed amenable to noting these although both are likely inadvertent introductions. In the case of the Small Toadflax this was likely as seeds with the limestone chippings on which it grew.
All seemed happy to record the Yellow Nonea Nonea lutea which appeared to be behaving as a typical garden ruderal with no obvious introduced source. Consensus broke down around the biggest group – those species which had no doubt been introduced but had in some way become established or not. Those with populations established and self regenerating mainly by seed and widely beyond the point of introduction were mainly conceded as established. Those regenerating vegetatively were generally not accepted, unless spreading in “wild” areas. Recording aliens is problematic.
The Wild Liquorice, Astragalus glycophyllus growing luxuriantly in a flower bed was noted on the basis of a conserved gene pool, having been sourced from the local wild population on nearby Gadbury Bank.
To the north of the farm yard is a small meadow supporting an unimproved grassland flora. The highlight here is a substantial population of the scarce county plant, Corky-fruited Water-dropwort Oenanthe pimpinelloides. In addition to the diverse native flora, there are several showy and established introductions Eastern Gladiolus, Gladiolus communis ssp. byzantinus, Fritillary, Fritillaria meleagris and Salsify, Tragopogon porrifolius.
Bert Reid and John Day also surveyed the lane and green lane from the Gloucestershire border to Marsh Brook. The most significant find was Grass Vetchling, Lathyrus nissolia in the Green Burial Ground to the north of Red House.
Brett Westwood and Ann Fells found Great Brome, Anisthera diandra on arable margins west of Red House.
The long list of plants for Red House is all the plants noted on the day. It does not infer that the plant was considered established (notes were attached to all records entered in the database).
| Red House Eldersfield 09/06/2012 | Compiled by J.J.Day |
| Red House Farm and Meadow Eldersfield | |
| Acer campestre | Field Maple |
| Acer platanoides | Norway Maple |
| Achillea millefolium | Yarrow |
| Aethusa cynapium ssp. cynapium | Fool’s Parsley |
| Agrostemma githago | Corncockle |
| Agrostis stolonifera | Creeping Bent |
| Ajuga reptans | Bugle |
| Alisma plantago-aquatica | Water-plantain |
| Alliaria petiolata | Garlic Mustard |
| Alopecurus pratensis | Meadow Foxtail |
| Anagallis arvensis ssp. arvensis | Scarlet Pimpernel |
| Anisantha sterilis | Barren Brome |
| Anthoxanthum odoratum | Sweet Vernal Grass |
| Anthriscus sylvestris | Cow Parsley |
| Aquilegia vulgaris | Columbine |
| Arctium lappa | Greater Burdock |
| Arrhenatherum elatius | False Oat-grass |
| Artemisia vulgaris | Mugwort |
| Arum maculatum | Lords-and-ladies |
| Aspelenium scolopendrium | Hart’s-tongue |
| Astragalus glycyphyllos | Wild Liquorice |
| Atriplex patula | Common Orache |
| Bellis perennis | Daisy |
| Brachypodium sylvaticum | False-brome |
| Briza maxima | Greater Quaking Grass |
| Bromus commutatus | Meadow Brome |
| Bromus hordeaceus ssp. hordeaceus | Soft Brome |
| Bromus racemosus | Smooth Brome |
| Caltha palustris | Marsh Marigold |
| Calystegia silvatica | Large Bindweed |
| Capsella bursa-pastoris | Shepherd’s-purse |
| Cardamine hirsuta | Hairy Bitter-cress |
| Carex pendula | Pendulus Sedge |
| Centaurea montana | Perennial Cornflower |
| Centranthus ruber | Red Valerian |
| Cerastium fontanum ssp. vulgare | Common Mouse-ear |
| Ceratophyllum demersum | Rigid Hornwort |
| Chaenorhinum minus | Small Toadflax |
| Cichorium intybus | Chicory |
| Cirsium vulgare | Spear Thistle |
| Conium maculatum | Hemlock |
| Corylus avellana | Hazel |
| Crataegus monogyna | Hawthorn |
| Crepis vesicaria | Beaked Hawk’s-beard |
| Cymbalaria muralis | Ivy-leaved Toadflax |
| Cynosurus cristatus | Crested Dog’s-tail |
| Dactylis glomerata | Cock’s-foot |
| Daucus carota ssp. carota | Wild Carrot |
| Digitalis purpurea | Foxglove |
| Dipsacus fullonum | Wild Teasel |
| Dryopteris filix-mas | Common Male Fern |
| Echium vulgare | Viper’s Bugloss |
| Elytrigia repens | Common Couch |
| Epilobium montanum | Broad-leaved Willowherb |
| Epilobium parviflorum | Hoary Willowherb |
| Erigeron karvinskianus | Mexican Fleabane |
| Eupatorium cannabinum | Hemp-agrimony |
| Euphorbia helioscopia | Sun Spurge |
| Euphorbia peplus | Petty Spurge |
| Foeniculum vulgare | Fennel |
| Fraxinus excelsior | Ash |
| Fritillaria meleagris | Fritillary |
| Galium aparine | Cleavers |
| Galium verum | Lady’s Bedstraw |
| Geranium dissectum | Cut-leaved Crane’s-bill |
| Geranium lucidum | Shining Crane’s-bill |
| Geranium phaeum | Dusky Crane’s-bill |
| Geranium pratense | Meadow Crane’s-bill |
| Geranium pyrenaicum | Hedgerow Crane’s-bill |
| Geranium robertianum | Herb-robert |
| Geranium sanguineum | Bloody Crane’s-bill |
| Geranium x magnificum | Purple Crane’s-bill |
| Geranium x oxonianum | Druce’s Crane’s-bill |
| Geum urbanum | Herb Bennet |
| Gladiolus communis ssp. byzantinus | Eastern Gladiolus |
| Glechoma hederacea | Ground-ivy |
| Hedera helix ssp. helix | Common Ivy |
| Hedera ‘Hibernica’ | Irish Ivy |
| Heracleum mantegazzianum | Giant Hogweed |
| Heracleum sphondylium | Hogweed |
| Hesperis matronalis | Dame’s Violet |
| Hieracium scotostictum | Dappled Hawkweed |
| Holcus lanatus | Yorkshire-fog |
| Hordeum distichon sens. str. | Barley |
| Hordeum secalinum | Meadow Barley |
| Inula conyzae | Ploughman’s-spikenard |
| Iris foetidissima | Stinking Iris |
| Lamium album | White Dead-nettle |
| Lamium maculatum | Spotted Dead-nettle |
| Lapsana communis | Nipplewort |
| Lemna minor | Common Duckweed |
| Lemna minuta | Least Duckweed |
| Lemna trisulca | Ivy-leaved Duckweed |
| Leucanthemum vulgare | Oxeye Daisy |
| Linaria purpurea | Purple Toadflax |
| Lunaria annua | Honesty |
| Lysimachia nummularia | Creeping-Jenny |
| Malus pumila | Apple |
| Meconopsis cambrica | Welsh Poppy |
| Medicago lupulina | Black Medick |
| Melissa officinalis | Balm |
| Milium effusum | Wood Millet |
| Muscari armeniacum | Garden Grape-hyacinth |
| Myosotis arvensis | Field Forget-me-not |
| Myosotis sylvatica | Wood Forget-me-not |
| Nigella damascena | Love-in-a-mist |
| Nonea lutea | Yellow Nonea |
| Oenanthe pimpinelloides | Corky-fruited Water-dropwort |
| Onopordum acanthium | Cotton Thistle |
| Orobanche hederae | Ivy Broomrape |
| Osmunda regalis | Royal Fern |
| Papaver lecoqii | Yellow-juiced Poppy |
| Papaver somniferum | Opium Poppy |
| Pentaglottis sempervirens | Green Alkanet |
| Phleum bertolonii | Smaller Cat’s-tail |
| Phleum pratense sens.str. | Timothy |
| Phuopsis stylosa | Caucasian Crosswort |
| Plantago lanceolata | Ribwort Plantain |
| Plantago major ssp. major | Greater Plantain |
| Poa angustifolia | Narrow-leaved Meadow-grass |
| Poa annua | Annual Meadow-grass |
| Poa trivialis | Rough Meadow-grass |
| Polystichum setiferum | Soft Shield-fern |
| Potentilla reptans | Creeping Cinquefoil |
| Potentilla sterilis | Barren Strawberry |
| Primula veris | Cowslip |
| Prunella vulgaris | Selfheal |
| Prunus spinosa s.l. | Blackthorn |
| Pulmonaria officinalis | Lungwort |
| Quercus robur | Pedunculate Oak |
| Ranunculus acris | Meadow Buttercup |
| Ranunculus repens | Creeping Buttercup |
| Rhinanthus minor | Yellow-rattle |
| Rosa canina agg. | Dog Rose |
| Rubus fruticosus agg. | Bramble |
| Rumex acetosa ssp. acetosa | Common Sorrel |
| Rumex crispus ssp. crispus | Curled Dock |
| Rumex obtusifolius | Broad-leaved Dock |
| Rumex sanguineus | Wood Dock |
| Sagina apetala sens. lat. | Annual Pearlwort |
| Sambucus nigra | Elder |
| Scorzoneroides autumnalis | Autumnal Hawkbit |
| Scrophularia vernalis | Yellow Figwort |
| Sedum acre | Biting Stonecrop |
| Sedum album | White Stonecrop |
| Sedum spurium | Caucasian-stonecrop |
| Senecio jacobaea | Common Ragwort |
| Senecio vulgaris var. vulgaris | Groundsel |
| Silene flos-cuculi | Ragged Robin |
| Sinapis arvensis | Charlock |
| Sisymbrium officinale | Hedge Mustard |
| Smyrnium perfoliatum | Perfoliate Alexanders |
| Solanum dulcamara | Bittersweet |
| Sonchus asper | Prickly Sow-thistle |
| Sonchus oleraceus | Smooth Sow-thistle |
| Stachys sylvatica | Hedge Woundwort |
| Stellaria graminea | Lesser Stitchwort |
| Stellaria media | Common Chickweed |
| Tamus communis | Black Bryony |
| Tanacetum parthenium | Feverfew |
| Taraxacum sp. | Dandelion agg. |
| Taxus baccata | Yew |
| Torilis nodosa | Knotted Hedge-parsley |
| Tragopogon porrifolius | Salsify |
| Tragopogon pratensis | Goat’s-beard |
| Trifolium dubium | Lesser Trefoil |
| Trifolium pratense | Red Clover |
| Trifolium repens | White Clover |
| Trisetum flavescens | Yellow Oat-grass |
| Triticum aestivum | Bread Wheat |
| Ulmus procera | English Elm |
| Urtica dioica | Common Nettle |
| Veronica arvensis | Wall Speedwell |
| Veronica chamaedrys | Germander Speedwell |
| Veronica persica | Common Field-speedwell |
| Veronica serpyllifolia ssp. serpyllifolia | Thyme-leaved Speedwell |
| Vicia faba | Broad Bean |
| Vicia hirsuta | Hairy Tare |
| Vicia sativa ssp. segetalis | Common Vetch |
| Vicia tetrasperma | Smooth Tare |
| Viola odorata | Sweet Violet |
| Viola reichenbachiana | Early Dog-violet |
| Vulpia bromoides | Squirrel-tail Fescue |
| Additional Species | |
| Red House Lane Eldersfield | |
| Alnus glutinosa | Alder |
| Alopecurus geniculatus | Marsh Foxtail |
| Alopecurus myosuroides | Black-grass |
| Atriplex prostrata sens.str. | Spear-leaved Orache |
| Bromopsis ramosa | Hairy Brome |
| Bryonia dioica | White Bryony |
| Calystegia sepium ssp. sepium | Great Bindweed |
| Cardamine flexuosa | Wavy Bitter-cress |
| Carex otrubae | False Fox-sedge |
| Carex remota | Remote Sedge |
| Carex spicata | Spiked Sedge |
| Centaurea nigra agg. | Common Knapweed |
| Chaerophyllum temulum | Rough Chervil |
| Chamerion angustifolium | Rosebay Willowherb |
| Chenopodium polyspermum | Many-seeded Goosefoot |
| Cirsium arvense | Creeping Thistle |
| Clematis vitalba | Traveller’s Joy |
| Convolvulus arvensis | Field Bindweed |
| Cornus sanguinea | Dogwood |
| Cornus sericea | Red-osier Dogwood |
| Epilobium hirsutum | Great Willowherb |
| Equisetum arvense | Field Horsetail |
| Euonymus europaeus | Spindle |
| Festuca rubra sens.str. | Red Fescue |
| Fumaria officinalis | Common Fumitory |
| Humulus lupulus | Hop |
| Hyacinthoides non-scripta | Bluebell |
| Hypericum hirsutum | Hairy St. John’s-wort |
| Iris pseudacorus | Yellow Iris |
| Juncus bufonius sens.str. | Toad Rush |
| Lamiastrum galeobdolon ssp. argentatum | Variagated Yellow Archangel |
| Lathyrus nissolia | Grass Vetchling |
| Lepidium coronopus | Swine-cress |
| Ligustrum vulgare | Wild Privet |
| Lolium perenne | Perennial Rye-grass |
| Lotus corniculatus | Common Bird’s-foot-trefoil |
| Mentha aquatica | Water Mint |
| Mercurialis perennis | Dog’s Mercury |
| Oxalis corniculata | Procumbent Yellow-sorrel |
| Poa pratensis sens.lat. | Smooth Meadow-grass |
| Polygonum aviculare sens.str. | Knotgrass |
| Populus x canadensis | Hybrid Black Poplar |
| Potentilla anserina | Silverweed |
| Rosa arvensis | Field Rose |
| Rubus caesius | Dewberry |
| Rubus ulmifolius | a bramble |
| Salix alba | White Willow |
| Salix cinerea ssp. oleifolia | Grey Willow |
| Salix fragilis | Crack Willow |
| Schedonorus arundinaceus | Tall Fescue |
| Schedonorus pratensis | Meadow Fescue |
| Scrophularia auriculata | Water Figwort |
| Scrophularia nodosa | Common Figwort |
| Silene dioica | Red Campion |
| Sison amomum | Stone Parsley |
| Symphoricarpos x chenaultii | Pink Snowberry |
| Torilis japonica | Upright Hedge-parsley |
| Ulmus glabra | Wych Elm |
| Vicia sepium | Bush Vetch |
| Viscum album | Mistletoe |
Worcestershire Record | 33 (Nov 2012) page: 86-88 | Worcestershire Biological Records Centre & Worcestershire Recorders
Worcestershire Record | 33 (Nov 2012) page: 22 | Worcestershire Biological Records Centre & Worcestershire Recorders
New Worcestershire Galls in 2012
J.J. Day
The sawfly Pontania triandrae produces bean galls on the Almond Willow Salix triandra. On 18th September 2012 a bush of Salix triandra was found in wet woodland along the Acton Brook at SO855667 supporting about 20 galls. This appears to be the first record for Worcestershire (Fig. 1.).
Corn Parsley Petroselinum segetum is a scarce Worcestershire plant with only about 20 colonies known. On 12th July 2012 whilst surveying the colony, first located by Brett Westwood in 1991, on the southern embankment of the A38 Droitwich bypass at SO898639, I found several plants, whose inflorescences were apparently galled and infested with mites. I carried the survey eastwards and onto the northern cutting at SO901638, where I located both more Petroselinum segetum (a new hectad record) and more infected inflorescences (Fig. 2.).
There are no named gall causers for Petroselinum segetum in British Plant Galls (Redfern & Shirley 2011) so scanned images and a description were sent to Margaret Redfern. Petroselinum segetum is apparently a new British host species for galls (Margaret Redfern pers comm). The naming of gall causing mites is problematic but the most likely species involved is Aceria peucedani which is the only mite known to gall members of the Apiaceae in Britain.
Reference.
Redfern, M & Shirley, P. 2011. P. British Plant Galls. Field Studies Council, Preston Montford.
Images
 Fig. 1. Gall Pontania triandrae, Acton Brook, SO855667, Worcestershire 18/09/2012. J J Day
Fig. 1. Gall Pontania triandrae, Acton Brook, SO855667, Worcestershire 18/09/2012. J J Day
 Fig. 2. Gall on Petroselinum segetum inflorescences probably Aceria peucedani, A38 Droitwich bypass cutting on north side SO901638, Worcestershire 17/07/2012. J J Day
Fig. 2. Gall on Petroselinum segetum inflorescences probably Aceria peucedani, A38 Droitwich bypass cutting on north side SO901638, Worcestershire 17/07/2012. J J Day
Worcestershire Record | 33 (Nov 2012) page: 23 | Worcestershire Biological Records Centre & Worcestershire Recorders
Vasates quadripedes Maple Bladder Gall Mite in Worcestershire
J.J. Day (John Day)
The gall mite Vasetes quadripedes is native to North America. It is parasitic on the leaves North American maples including Silver Maple Acer saccharinum; Sugar Maple Acer saccharum; Red Maple, Acer rubrum. The mite causes red pustules up to 5mm high normally on the upperside of the leaves (Figs. 1 & 2). They are well established on the leaves by June, when much of the leaf surface may be infested. They become black later in the season.
The first British record for the gall was in London (Wurzell 2002) but is now known to be spreading across the country (Dr.M.Redfern pers.comm.). The only host known in Britain is the Silver Maple Acer saccharinum (Redfern & Shirley, 2011). The Silver Maple is a native of Eastern North America from Ontario to Florida. It was first introduced into Britain in 1725 (Sell & Murrell, 2009). It is commonly planted, in parks and gardens as an ornamental tree.
Worcestershire’s first record for the gall came in August 2008 when it was noted by J.J.Day from a park in Droitwich. There were no further records until June 2011 when three new sites were located, by J J Day (2) & G H Green (1), from widely spaced areas of the county (SO83, SO84, SO96). Two more sites were also located in the same year in SO85 & SP06; the latter site is in VC38 Warwickshire (Table 1.)
Both young and mature trees have been noted as hosts in Worcestershire. The gall is unmistakeable and Silver Maples anywhere in the county should be checked as potential hosts.
Table 1. Worcestershire Records of Vasates quadripedes
| Site | Grid Reference | Date | Recorder | Host | Vice County |
| Eldersfield – Linkend Road | SO81743077 | Jun-11 | J J Day | Young tree <10years | vc37 |
| Droitwich – St Peters Park | SO902629 | Jun-11 | J J Day | Large tree | vc37 |
| Redditch – Matchborough | SP071664 | Jul-11 | J J Day | Tree 30-40 years | vc38 |
| Worcester – County Hall | SO876542 | Oct-11 | J J Day | Tree 30-40 years | vc37 |
| Droitwich – Celvestune Way Park | SO886624 | Aug-08 | J J Day | Large tree | vc37 |
| Earls Croome – Garden Centre | SO863421 | Jun-11 | G H Green | Young tree | vc37 |
References
Redfern, M & Shirley, P. 2011. P. British Plant Galls. Field Studies Council, Preston Montford.
Sell, P. & Murrell, Gina. 2009. Flora of Great Britain & Ireland Vol. 3.
Wurzell, B. 2002. The maple bladder gall of the gall mite Vasates quadripedes new to Britain Cecidology.17(1):31-35.
Images
 Fig. 1. Vasates quadripedes on Acer saccharinum 14th June 2011, Earls Croome, Worcestershire SO863421. G.H.Green
Fig. 1. Vasates quadripedes on Acer saccharinum 14th June 2011, Earls Croome, Worcestershire SO863421. G.H.Green
 Fig. 2. Vasates quadripedes on Acer saccharinum 11th June 2012, Eldersfield, Worcestershire, SO81743077. G.H.Green
Fig. 2. Vasates quadripedes on Acer saccharinum 11th June 2012, Eldersfield, Worcestershire, SO81743077. G.H.Green
Worcestershire Record | 33 (Nov 2012) page: 22 | Worcestershire Biological Records Centre & Worcestershire Recorders
Fungal parasites in Oak Bushcrickets Meconema thalassinum
Gary Farmer
On 4th September 2012 I collected two female Oak Bushcrickets Meconema thalassinum from Grafton Wood. They were found about five meters apart on a sunny ride, each located on the underside of a leaf about a metre from the ground. On inspection they were both in perfect condition but totally immobile. Suspecting an internal parasite to be responsible I placed them in a collecting box for further observation. On returning home I found that a fungus had erupted from both specimens making them almost unidentifiable (Fig. 1.). It appears they had been parasitised by a fungus. I sent details to the Orthoptera Recording Scheme and received the following response from Helen Roy of CEH “It looks like an entomopthoralean fungus – a species specific fungus – the way it has adhered the insect to the leaf surface is very characteristic. For identification the conidia (spores) need to be assessed but I will forward to a European expert for further comment. Great find!”
I’ve seen rows of hoverflies and dungflies on grassheads in the past and suspected fungal parasites but had not realised that the fungus had actually glued them in place!
Image
 1. Oak bush-cricket attcked by fungus. Gary Farmer
1. Oak bush-cricket attcked by fungus. Gary Farmer
Worcestershire Record | 33 (Nov 2012) page: 19 | Worcestershire Biological Records Centre & Worcestershire Recorders
Is the Rosemary Rainbow Beetle Chrysolina americana at large in Worcestershire?
Gary Farmer
During a visit to Snowshill Lavender near Broadway my son found several metallic, striped beetles. I had seen this species once before (in Solihull) and so could immediately identify them as Rosemary Rainbow Beetles Chrysolina americana.
This species is beautiful but feeds on the leaves of Rosemary, Lavender, and related plants and it appears that some species in the Lamiaceae family can also be hosts.
The beetle grows to around 8mm long and is metallic green with purple stripes and so is very distinctive.
The beetle is a native of southern Europe and was first found living out of doors in the UK in Surrey in 1994 and by 1998 established colonies of the beetle had been discovered near London’s Waterloo Station and near Reading, Berkshire. By the end of 2005 the beetle had become widespread throughout London and surrounding areas. Rosemary Beetle is now widespread in England and has been found in parts of Scotland, Wales, Northern Ireland and the Irish Republic.
I sent a record off to Gloucestershire BRC and received the following response:
“The Rosemary Beetle seems to be cropping up quite often this year; a lady reported some from the middle of Cheltenham recently. They are beautiful but not the gardener’s friend, as they really do eat Rosemary, often stripping whole bushes of leaves. Unfortunately for Cotswold Lavender, they also eat Lavender leaves.… it’s awkward to treat them as any insecticides are highly likely to affect bees. I notice that Mayfield Lavender Farm in Surrey are giving out jars for people to collect the beetles as they look around the fields, rather than risk spraying.…”
The Royal Horticultural Society is keeping track of the spread of rosemary beetle at the moment: http://www.rhs.org.uk/Science/Plant-pests/Rosemary-beetle.
Having found this species to the north east and south of the county I do wonder if it is lurking somewhere in Worcestershire maybe in a garden near you. This is certainly one to watch for in the future.
Images
 Fig. 1. Rosemary Beetle Chrysolina americana 15.07.12 Snowshill Lavender. Gary Farmer
Fig. 1. Rosemary Beetle Chrysolina americana 15.07.12 Snowshill Lavender. Gary Farmer
 Fig. 2. Rosemary Beetle Chrysolina americana 24.08.08 Malvern Park, Solihull. Gary Farmer
Fig. 2. Rosemary Beetle Chrysolina americana 24.08.08 Malvern Park, Solihull. Gary Farmer
Worcestershire Record | 33 (Nov 2012) page: 24 | Worcestershire Biological Records Centre & Worcestershire Recorders
Lesne’s Earwig Forficula lesnei. Scarce in Worcestershire or just overlooked?
Gary Farmer
Lesne’s Earwig is an uncommon species nationally. The soon to be updated Grasshoppers, Crickets and Allied Insects in Britain and Ireland (Haes & Harding 1997) notes that it is Nationally Scarce (B) and is ‘recorded from scattered locations in Southern England and the Gower’. At that time there were no records for Worcestershire but I subsequently found it at Windmill Hill (Worcestershire Wildlife Trust Nature Reserve) and along the river Avon at Twyford Garden Centre (Evesham Country Park). Twelve years on I revisited the two sites to look for this small earwig and found it again at both places. Its chosen habitat at these sites is Old-man’s-beard Clematis vitalba growing over or adjacent to old Hawthorn hedges. NBN Gateway now shows several records for our area; a cluster around Evesham, a single record from Pershore and a single record further north, from Highley. So are they waiting to be found at other localities in Worcestershire?
Turn your brollies up-side-down and give an Old-man’s-beard a good hard shake (just make sure it’s not attached to an old man!). You are pretty much guaranteed to find Common Earwigs Forficula auricularia so get your records in for those as well. If you find a smaller (around 8mm), ginger/brown earwig with no protruding wings amongst the larger chestnut/brown individuals check the cerci (pincers). If you have a male the identification is straight forward (see images).
References
Farmer, G. 2001. The earwigs of Worcestershire Worcestershire Record11:32
Haes, E.C.M. & Harding, P.T.. 1997. Atlas of grasshoppers,crickets and allied insects in Britain and Ireland. The Stationery Office, London
Images
 1. Lesne’s Earwig Forficula lesnei male 10.09.12 Windmill Hill. Gary Farmer
1. Lesne’s Earwig Forficula lesnei male 10.09.12 Windmill Hill. Gary Farmer
 2. Lesne’s Earwig Forficula lesnei female 10.09.12 Windmill Hill. Gary Farmer
2. Lesne’s Earwig Forficula lesnei female 10.09.12 Windmill Hill. Gary Farmer
 3. Common Earwig Forficula auricularia male cerci
3. Common Earwig Forficula auricularia male cerci

Worcestershire Record | 33 (Nov 2012) page: 67-68 | Worcestershire Biological Records Centre & Worcestershire Recorders
Sorbus domestica, The Woodward Family Connection in Worcestershire
Hermione Gerry
The True Service Tree of Worcestershire, also called the ‘Whitty Pear’ or ‘The Sorb Tree’.
The Woodward Family Connection, Upper Arley, Worcestershire.
Hermione Gerry née Woodward. October 2012
It is well-documented that the ‘Whitty Pear’ has been growing in the Wyre Forest, Worcestershire since 1678 when a single specimen was first mentioned as a native tree by Edmund Pitt. Since then various mentions of its existence there have been made. This specimen was known to be very old even from the time when it was discovered. As a child I was told that it was thought that it may have been introduced to this country by the Romans although this was not proven and is, I understand, doubted by some experts. What was certain was that it was very rare, with the only example growing in the wild being that in the Wyre Forest.
Its rarity was plainly known by a local vagrant who, it is said, had a grievance with the local land-owner and, presumably in a moment of pique, decided to set fire to and destroy this unique specimen. There is a suggestion that a local magistrate, who may or may not have been the Landlord, was very fond of the tree and used to hold his ‘court’ beside it. The vagrant had apparently been found guilty of poaching: and this is why he burnt the tree down. What the grievance was, I was never told, so the poaching story may or may not be true, however it seems clear that he did a ‘good’ job and the tree was destroyed. This was in 1862, 150 years ago. Now this could have been the end of the story, but fortunately it isn’t.
The Whitty Pear is not easy to propagate from cuttings, however it is possible to grow it from seeds (pips). To the best of my knowledge and from what I was told by my Father, the late Sir Chad Woodward, seed from the original specimen had luckily been grown at Arley Castle. My family had moved to Upper Arley in 1852, when my great Grandfather, Robert Woodward, bought the Arley Castle Estate from Lord Mountnorris. It is known that the Tree was alive in 1856, and only destroyed in 1862, ten years after the Woodwards moved to Arley and so it is very likely that the living descendants of the original specimen, had been germinated, or raised, by members of my family. This is what I was always led to believe. However, as Lord Mountnorris started the Arley Arboretum in circa 1820, it is also possible I imagine, that seeds had been germinated before my family moved to Arley.
My Father’s family had a great interest and love of trees and shrubs and took delight in continuing the development of the Arley Arboretum. His elder brother, Robert Woodward Jnr, who should have inherited the Estate from his Father had he not been killed in the First World War, had without doubt germinated Whitty Pear seeds from descendants of the original, including the one planted in the Wyre Forest by my Grandmother. Not only was he interested and most knowledgeable about trees and shrubs in general, but also meticulous in keeping a record of the trees in the grounds of Arley Castle. I am lucky enough to have this record which is entitled ‘Catalogue of trees in – Arboretum, Lawn, Naboth, Flower garden & Park at Arley Castle’.
There is an entry made in the Catalogue by my Uncle in December 1903 which reads, and I quote:
“A strike or seedling from the original tree in the Wyre Forest, which was described by Phil: Trans of 1678 by Mr Pitt. Then considered an old tree. Alive in 1856. Destroyed by fire in 1862, kindled by a vandal who knew that it was a tree of interest. He was afterwards Transported for setting fire to some farm buildings.
See (i) Lightfoots Flora Sestica page 257 (ii) Loudon’s Arboretum Brittanicum (iii) The Botany of Worcs. By Edwin Lees pub: 1867 (iv) Nash’s Worcestershire.
Sir Walter Blount and Sir Edward Winnington both tried to propagate it but apparently without success.”
After my Uncle was killed, in France on 9th May 1915 it was decided to plant a tree in his memory, and what better tree to plant than an offspring, raised by him, of the original Whitty Pear. The place selected to plant this sapling was the site of the original tree, in the Wyre Forest, and so on 30th March 1916 it was planted by my Grandmother, Mrs Robert Woodward (Mary Jane) on that site. It is still growing there, not far from the Forestry Commission Visitor Centre.
My Grandfather, Robert Woodward, died in 1919 after which my Father, being the second son, inherited the Arley Estate, but moved out of the Castle which was let out as a girl’s school, and into Arley Cottage. This is where I grew up. We have, in the Arley Cottage garden, a Whitty Pear, another direct descendant of the original Tree, planted by my Father in around 1930. This tree regularly flowers and bears fruit.
Since moving back into Arley Cottage in 2005, we have germinated several seedlings from ‘pips’ from this tree. We have not tried to take cuttings. Some of the seedlings have been given away but we still have three saplings. One of these has been planted out in our garden and has already reached around eight feet tall, but has not yet flowered. The other two are in pots, one of which, it is planned, will be planted back into the Wyre Forest in March 2013, almost exactly 97 years after my Grandmother planted the existing specimen. It is currently over a metre tall, and growing fast!
Editor’s note
Information on the Whitty Pear has appeared in many natural history and other publications. In 2009 I wrote a summary ‘time-line’ of its history (Green 2009) and included a list of the most useful publication which is copied below. Following Hermione Gerry’s article we plan to write a more extended version in the (hopefully) not-to-distant future.
Reference
Green, H. 2009. The Old Sorb Tree, Whitty Pear or The True Service Tree, Sorbus domestica L, Previously Sorbus pyriformis. Wyre Forest Study Group Review, 10:69-73. Also see http://www.wyreforest.net/wp-content/uploads/2012/04/The-Old-Sorb-Tree.pdf
Short bibliography.
Claxton, Frances 1999 the Whitty Pear Sorbus domestica L. A Natural Pioneer Millennium Award Project – Work in Progress. Worcestershire Record 7:28-30.
Hampton, Marc 1996. Sorbus domestica L. – comparative morphology and habitats. BSBI News No 73 pages 32-37.
Hampton, M. & Kay, Q. O. N. 1995. Sorbus domestica L., new to Wales and the British Isles. Watsonia. 20:379-384.
Hastings, Charles. 1834. Illustrations of the Natural History of Worcestershire. London: published by Sherwood, Gilbert & Piper; Lees, Worcester, at the request of the Worcestershire Natural History Society.
Jeffrey, F. Ronald. 1916. The Wyre Forest Sorb Tree. Transactions of the Worcestershire Naturalists’ Club. Volume 6, part 3, pages 250-257. This article is a useful account of the history of the tree.
Jennings, Fred. 2005. The sorb tree of Wyre – the True Service or Whitty Pear (Sorbus domestica). Wyre Forest Study Group Review 6:53-56.
Jones, Mary M. 1980. The Lookers-out of Worcestershire. The Worcestershire Naturalists’ Club. Chapter 7: The fate of the old sorb tree and its descendants: Pages 69-80. The best modern summary of the history of the tree up to 1980.
Kausch-Blecken von Schmeling, W. 2000. Der Speierling Sorbus domestica L. In German. A book on history, distribution, cultivation, timber and use of the fruit.
Lees, Edwin 1867. The botany of Worcestershire. Worcestershire Naturalists Club
Tomkinson, K & Everett, E. 1984. Selections from Nash’s Worcestershire. . Kenneth Tomkinson Ltd. From Collections for the history of Worcestershire by Treadway Russell Nash written between 1781 -1782 in two large volumes.
Worcestershire Record | 33 (Nov 2012) page: 67-68 | Worcestershire Biological Records Centre & Worcestershire Recorders
Worcestershire Record | 33 (Nov 2012) page: 5-6 | Worcestershire Biological Records Centre & Worcestershire Recorders
Ash dieback caused by the fungus Hymenoscyphus pseudoalbidus(Helotiales: Helotiaceae)(Chalara fraxinea): up to 90% of ash trees Fraxinus excelsior expected to die, and prospects for Tiddesley Wood, Pershore
David M. Green davidmxgreen@gmail.com
Up to 90% of ash trees Fraxinus excelsior in Britain are expected to die within the next 10 years from the pathogenic fungus Hymenoscyphus pseudoalbidus which causes the chronic disease “ash dieback” (not to be confused with the previously known “die back” of the crown of ash trees from physical conditions). There are now almost 300 confirmed cases at separate sites of infected trees, the majority found in mature woodland (Department for Environment, Food and Rural Affairs (Defra) as reported in http://www.bbc.co.uk/news/science-environment-20626053 6th December 2012).
Hymenoscyphus pseudoalbidus – Chalara fraxinea is the asexual (anamorphic) stage of the same species – was ravaging ash Fraxinus excelsior in Poland in 1992, thereafter various parts of Europe. The first record in Britain was February 2012 when it was found in a consignment of infected trees sent from a nursery in the Netherlands to a nursery in Buckinghamshire, England. (http://www.forestry.gov.uk/chalara 2012-Nov-06).
Observing in Denmark that ash plants were dying in nurseries, in 2009 Britain’s Horticultural Trades Association proposed to Defra that imports of ash plants to the UK should be banned. But the Forestry Commission of the UK considered the disease was a fungus already endemic to the UK, namely Hymenoscyphus albidus which is not pathogenic, but it was supposed environmental conditions led it to be damaging. (http://www.guardian.co.uk/environment/2012/oct/29/government-ignored-ash-dieback-warnings 6th November 2012). So at that time the pathogen was not correctly identified. Basic knowledge was developing. The following quote from the Wikipedia article Hymenoscyphus pseudoalbidusseems to surmise the taxonomic background conveniently (http://en.wikipedia.org/wiki/Hymenoscyphus_pseudoalbidus 2012-Nov-06):
“The fungus Hymenoscyphus pseudoalbidus was first identified and described in 2006 under the name Chalara fraxinea. [5] In 2009, based on morphological and DNA sequence comparisons, Chalara fraxinea was suggested to be the asexual stage (anamorph) of the ascomycete fungus Hymenoscyphus albidus.[5] However, Hymenoscyphus albidus has been known from Europe since 1851 and is not regarded as pathogenic.[6] In 2010, using molecular genetic methods, the sexual stage (teleomorph) of the fungus was recognized as a new species and named Hymenoscyphus pseudoalbidus.[5] Although Hymenoscyphus pseudoalbidus is said to be “morphologically virtually identical” to Hymenoscyphus albidus, there are substantial genetic differences between the two species.[7]
“The fungus grows during summer on ash petioles in the litter.[6] The ascospores are produced in asci and are transmitted by wind; this might explain the rapid spread of the fungus.[6] It is not known why it has emerged or become so aggressive.[8] but researchers are investigating the theory that the fungus originated in Asia, where ash trees are immune to the disease”.
However, there is the argument that it was wrong for to allow continuing import of plants into the UK from areas of Europe where a disease was newly ravaging ash, as knowledge of the pathogen should not have been assumed satisfactory enough to conclude the pathogen was already endemic to Britain where ash was not being ravaged. Spores of the fungus are wind-spread; but import into Britain of spore carrying material hastens or increases the odds of transmission.
Wildlife network failure
It is unsatisfactory that despite a new disease of ash was evidently ravaging parts of Europe nothing significant was communicated generally within Britain so numerous relevant people seem never to have heard of it until recently. The network of conservation organisations in the UK or organisational structure failed to communicate or take note of this threat so nature reserves were managed with the assumption ash trees would continue. Now we face the prospect of woodland reserves containing about 50% ash, such as Tiddesley Wood, near Pershore, that have been managed accordingly, are set to lose up to 40% of their trees. The woodland management was designed for certain conditions which will become different. Ash is particularly important on Bredon Hill where work is done to retain old ash trees. The rare violet click beetle Limoniscus violaceus(Coleoptera: Elateridae) breeds in old ash trees. These sites are only two local examples. Obviously knowledge of events in continental Europe indicating what was likely or might happen in the UK should have been discussed & communicated widely. Other tree diseases & pests are threatening, but this disease of ash is especially obviously virulent, the consequences to woods & landscape changes of the countryside especially consequential. Even if many trees ultimately fail to die, the prospect cannot be ignored. The devastating Dutch elm disease of the elm Ulmus showed what might suddenly happen to a tree species in the UK.
Images

Fig. 01. Tiddesley Wood, south end, looking south: the compartment has been recently felled leaving ash as almost the only trees, so this compartment may be rendered almost treeless (except for coppice growth). 3rd December 2012. David M. Green

Fig. 02. North-east Tiddesley Wood: dense ash, trunks appearing pale in photograph. This is a compartment with trees too dense too grow at the normal rate. The fungus might be expected to thin them out considerably if not eliminating them. 3rd December 2012. David M. Green

Fig. 03. Tiddesley Wood, middle, looking west of main ride: a mixture of oak, ash & silver birch over a vigorous coppice growth much of ash. The ash coppice might be expected to die first, then the remaining ash in following years. The canopy is already very open. The lack of competition of ash coppice might be expected to favour maximum rate of growth for the oaks thereafter. 18th September. David M. Green

Fig. 04. North-east Tiddesley Wood: an example of one of the many approximately 50-60 year old ash grown from coppice from when most of the wood was clear felled or killed with herbicide in the 1950-1960s. 3rdDecember 2012. David M. Green

Fig. 05. Bredon Hill: near Doctors Wood, Little Comberton: an ash with branches lopped to prolong its life. Will the higher altitude mean Bredon Hill stays above the disease for some years? Older trees are more resistant to the disease, tend more to being chronically affected than saplings that die fast. 15th September 2012. David M. Green
Worcestershire Record | 33 (Nov 2012) page: 29-30 | Worcestershire Biological Records Centre & Worcestershire Recorders
A cocoon of Hypera rumicis (L., 1758). The golden net ball
Harry Green
On Sunday 1st July 2012 a group of us visited the Vale Landscape Heritage Trust Reserve on the River Avon flood plain near Lower Moor in the Vale of Evesham. At one point I noticed a tiny golden net ball (c4 mm diameter) attached to a leaf of Gypsywort Lycopus europaeus growing on the edge of a ditch (Fig. 1 & 2.). Nobody present could identify it so I carefully picked the leaf and placed it in a tube for further investigation. Next day a weevil had emerged from the golden ball and was sitting next to it on the leaf (Fig. 3.). I sent pictures to Paul Whitehead who responded as follows:
“Your cocoon is certainly that of a Hypera and from the beetle image you have sent to me I am reasonably confident that the species is Hypera rumicis (L., 1758). The nomenclature of eririnomorph** Hypera is presently somewhat unstable. Some continental workers place Hypera alternans, adspersa, pollux and various other names in synonymy with H. conmaculata(Herbst, 1795) and it remains to be seen whether the forthcoming volume 8 of the Palaearctic Catalogue will follow this. British collections of Hyperamay then require reassessment. In my experience Hypera rumicis sensu stricto is locally distributed in the English midlands on floodplains and in wetlands and I have seen it in coastal wetlands in Macedonia.”
“** Hypera are subdivided into various subgenera. Most Hypera you will encounter, most of the dry-loving ones, are in the subgenus Hypera. One or two are in the subgenus Dapalinus and Hypera zoilus, a large species that may now be declining, is in Antidonus. Hypera rumicis and Hypera conmaculata are in the subgenus Eririnomorphus hence ‘Eririnomorph’.”
I vaguely remembered seeing a similar picture of a golden net ball in the past but could not remember when. Rosemary Winnall reminded me that John Partridge had circulated a picture taken on 14th June 2011 which remained unidentified at the time (Fig. 4.). However, following these clues readers will be able to find similar pictures via an internet search. See for example http://www.flickr.com/photos/23111015@N04/2744112277/
http://www.commanster.eu/commanster/Insects/Beetles/SpBeetles/Hypera.rumicis.html
Images

Fig. 1. Hypera rumicis coccoon on Gypsywort. Harry Green

Fig. 2. Hypera rumicis coccoon on Gypsywort with weevil inside. Harry Green

Fig. 3. Hypera rumicis newly hatched from coccoon in Figs. 1 & 2. Harry Green

Fig. 4. Golden ball cocoon 14June 2011.
Worcestershire Record | 33 (Nov 2012) page: 41-42 | Worcestershire Biological Records Centre & Worcestershire Recorders
Climbing Land Caddis Enoicyla pusilla in Wyre Forest
Harry Green
On the 20th April 2012 members of the Wyre Forest Study Group (WFSG) were led by Brett Westwood and others to the Dowles Brook near Dowles Manor in search of Lampreys. Unfortunately run-off from recent heavy rain had caused considerable turbidity and this and raised water levels in the brook made a search almost impossible. However, the Group stopped for lunch at a collection of wet logs (Figs. 1 & 2.) on wet ground (SO772764), and soon discovered many (at least 50) Land Caddis larvae (Fig. 3.) actively climbing up the logs and several apparently browsing on a patch of black slime-mould (Figs. 4 & 5).
Several WFSG re-visited the site again on 28th April 2012 following yet more wet weather. Rosemary Winnall wrote:
“We revisited the logs this morning at 9.30am and counted 30 land caddis as before up to 6 inches off the ground. We measured several and they were between 7 and 9 mm in length. Someone had rolled one of the logs as you can see, but we left it as there were several caddis up on the side. There were two on that patch of black slime mould. Interestingly Mike Averill went to photograph the logs with land caddis yesterday afternoon and found none! He and Will Watson set off down there again at 4.30p.m. this afternoon after a day without rain to see if they could find them – we look forward to hearing what they found!”
Mike Averil wrote: “Interestingly there were only a handful showing by 4.30 p.m.. Will and I managed to get some pictures but they were as numerous as in the morning. None on our favourite slime mould patch”.
Land caddis larvae usually live within leaf litter in woodlands. Green & Westwood (2005) wrote: “Although not aquatic, Land Caddis larvae require a relative humidity of near 100%, although they can survive in drier conditions for a short time. They lack gill filaments and respiration is through the skin. If they are soaked by heavy rain they will climb up vegetation, for example low brambles, and hang themselves out to dry, dropping down again when relative humidity falls below 70%. This behaviour probably explains the origin of Hickin’s larvae invading a tent. We have occasionally seen many larvae together on the surface of mosses and liverworts growing on a stream bank following rain, and we have an entomological friend who passed a sweep net through low bramble in Wyre Forest and was astonished to collect many Land Caddis larvae”.
This note reports what is probably another example of land caddis larvae climbing to escape from very wet ground conditions. AS far as we are aware there are no reports of Land Caddis larvae eating fungi or slime moulds. Even in this instance we cannot be sure they were actually consuming the slime mould although their mouthparts appeared to be moving when the larvae were static on the mould.
Reference
Green, G. H. & Westwood, B. 2005. In search of the land caddis. British Wildlife 17(1):21-26.
Images

Fig. 1 Land caddis larvae climbed these logs in wet conditions. Rosemary Winnall

Fig. 2. Land caddis larvae climbed these logs in wet conditions. Rosemary Winnall

Fig. 3. Land caddis Enoicyla pusilla larva. John Bingham

Fig. 4. Land caddis Enoicyla pusilla larva on slime mould. Harry Green

Fig. 5. Land caddis Enoicyla pusilla larva on slime mould. John Bingham
Worcestershire Record | 33 (Nov 2012) page: 73-76 | Worcestershire Biological Records Centre & Worcestershire Recorders
Field Recording Days 2012. Worcestershire Recorders and Worcestershire Biological Records Centre
Harry Green
Three special field recording days were arranged for the summer of 2012:
Saturday 9th June 2012. The Red House and environs, Pillows Green Lane, near Staunton SO793301. A site with a good variety of habitats including new and old herb-rich grasslands, garden and orchard, and part of an ancient green lane. By kind permission of owner Keith Turner.
Saturday 7th July 2012. Slade Wood, Goom’s Hill, Abbots Morton, central grid reference SP023537. An ancient broad-leaved woodland. By kind permission of the owner, Lady Walker.
Saturday 4th August 2012 Ipsley Alders Nature Reserve in Redditch, central grid reference SP078676. Marshland, grassland, scrub, stream and woodland. By kind permission of Worcestershire Wildlife Trust.
The most striking feature of the 2012 recording days was cold wet weather. For the visit to The Red House the weather was comparatively good, merely cloudy and wet underfoot. Slade Wood was incredibly wet underfoot after days of very heavy rain: it also rained during the visit but there were occasional short bright periods when the sun shone very briefly. It was cool and quite dry at Ipsley Alders although wet underfoot. The poor weather may have reduced attendance as between 20 and 25 people booked for each visit but, in date order, only 16, 15 and 14 attended. Several of our regular recorders could not attend so we are shorter than usual on Coleoptera and Diptera records, and on invertebrate records generally.
Nevertheless there were highlights. The Red House turned out to be a very good site for bumblebees with nine species recorded including the two Worcestershire rarities Bombus humilis and Bombus ruderarius indicating the importance of extensive flowery grasslands, We had hoped to return later in the year for the bad weather prevented another visit (Figs. 1-4). Slade Wood was so wet that recording was difficult. Nevertheless a good number of moths (including leaf miners) were found largely because county moth recorder Tony Simpson was with us, and an abundance of frogs and toads. We hope for a return visit to do justice to this interesting wood (Figs. 5-9). Ipsley Alders Marsh is a well-known site for both Long-winged Conocephalus fuscus (discolor) and Short-winged Coneheads Conocephalus dorsalis. We found the first but not the second which is only known from two Worcestershire sites. However the development through nymphal stages of both these two species was slow in summer 2012 and adult C. dorsalis appeared several weeks later than usual and were later found at Ipsley Alders by Gary Farmer. Finding the curious bug Ledra aurita and Eyed Ladybird Anatis ocellata were interesting bonuses (Figs.10-13).
The lists that follow contain all the records received by WBRC by December 2012. Outstanding records are still very welcome and should be sent to Simon Wood, WBRC Manager, at records@wbrc.org.uk or to the address on the back of Worcestershire Record. The aim of these recording days is to obtain a snapshot of the species on the site for the visit day. The lists may be more interesting to people in one hundred years time than now!
The separate lists of Vascular Plants were compiled by John Day and Bert Reid. All other records extracted from the WBRC database by Simon Wood.
Recorders initials used in the lists:
| Alan Lawson | AL |
| Tony Simpson | AS |
| Brett Westwood | BW |
| Denzil Devos | DD |
| Dave Scott | DS |
| Gary Farmer | GAF |
| Harry Green | GHG |
| Geoff Trevis | GHT |
| John Bingham | JB |
| John Day | JJD |
| Jane Scott | JS |
| John Meiklejohn | JWM |
| Mike Averill | MA |
| Martin Skirrow | MS |
| Rosemary Winnall | RAW |
Images
 Fig. 01. The Red House. Old meadow. Harry Green
Fig. 01. The Red House. Old meadow. Harry Green
 Fig. 02. The Red House. Newly planted meadow. Harry Green
Fig. 02. The Red House. Newly planted meadow. Harry Green
 Fig. 03. The Red House. Bombus hypnorum. Harry Green
Fig. 03. The Red House. Bombus hypnorum. Harry Green
 Fig. 04. The Red House. Bombus ruderarius. Brett Westwood.
Fig. 04. The Red House. Bombus ruderarius. Brett Westwood.
 Fig. 05. Slade Wood showing wet woodland ride conditions. Harry Green
Fig. 05. Slade Wood showing wet woodland ride conditions. Harry Green
 Fig. 06. Slade Wood. John Day and Bert Reid by pond in brief sunshine. Harry Green
Fig. 06. Slade Wood. John Day and Bert Reid by pond in brief sunshine. Harry Green
 Fig. 07. Slade Wood. Recorders amongst wet vegetation. Harry Green
Fig. 07. Slade Wood. Recorders amongst wet vegetation. Harry Green
 Fig. 08. Slade Wood. John & Denise Bingham in heavy rain. Harry Green
Fig. 08. Slade Wood. John & Denise Bingham in heavy rain. Harry Green
 Fig. 09. Slade Wood. Volucella bombylans on wet leaf. Harry Green
Fig. 09. Slade Wood. Volucella bombylans on wet leaf. Harry Green
 Fig. 10. Ipsley Alders .Wet meadow. Harry Green
Fig. 10. Ipsley Alders .Wet meadow. Harry Green
 Fig. 11. Ipsley Alders sign board & recorders near pool
Fig. 11. Ipsley Alders sign board & recorders near pool
 Fig. 12. Ipsley Alders. Ledra aurita. Harry Green
Fig. 12. Ipsley Alders. Ledra aurita. Harry Green
 Fig. 13. Ipsley Alders. Eyed Ladybird Anatis ocellata. Harry Green
Fig. 13. Ipsley Alders. Eyed Ladybird Anatis ocellata. Harry Green
Worcestershire Record | 33 (Nov 2012) page: 39-40 | Worcestershire Biological Records Centre & Worcestershire Recorders
Sightings of adult Land Caddis Enoicyla pusilla and a mating pair, Worcestershire, October 2012
Harry Green and Brett Westwood.
On 8th October 2011 members of a Wyre Forest Study Group (WFSG) outing including one of the authors (BW) in Rock Coppice discovered several male land caddis Enoicyla pusilla in oak-leaf litter (Fig. 02.). The males were typically restless, but flying short distances between leaves or other low vegetation no more than 30cm above the ground. As they seemed to be localised, we searched in the moss and leaf litter and soon found a wingless female, crawling through the dead leaves (Fig. 01). The weather was overcast, rather damp with little or no sunshine.
On 13th October 2012, the authors while on a WFSG field meeting again found two male Eniocyla pusilla on leaf-litter on the slopes of a dingle at Hawkbatch Valleys. On gently raking away the topmost leaves, they uncovered two females at the same spot, looking rather like large springtails but easily identified by their long legs and constantly waving antennae. The males were apparently reluctant to leave the area and, as in Rock Coppice, and were hard to find elsewhere, which suggests that they may have been assembling in response to cues from the females. Being flightless, it is possible that pheromones are used by the female to lure in the winged males (Fig.s 3, 4, & 5).
Land Caddis near Astley Mill
During the morning of 18th October 2012 one of the authors (HG), together with Dave & Jane Scott, and Mike Averill visited a small woodland plantation (larch, poplar) near Astley Mill which was for sale by auction. Just across the road from the wood is an enormous old oak pollard grid referenceSO7819 6737 (Fig. 05.) situated on a wooded bank (oak, hazel) which we decided to record for the ancient tree register (6.85m chest height circumference). HG noticed a good layer of interesting oak and hazel leaf litter beneath the tree and searched therein for land caddis larval cases (Fig. 06.). After scrabbling around for a few minutes one of us suddenly realised there were 20-30 male land caddis flying about. HG said “look for females!” A task for which we are ill-equipped! (poor eyesight, poor ambient light). However, Jane Scott noticed one male dip down and – amazingly – joined what looked like a female. The two were caught in a specimen tube confirming the fact. We tipped them back onto a litter and after the male had skittered about for a few seconds they mated! (Figs. 7 & 8). Other males appeared in the immediate vicinity and one other seemed to also mate with the female who then started to crawl into hiding amongst the leaf litter leaving several males skittering about. Had she switched off pheromone attractants?
The excitement of observing this behaviour, hitherto un-recorded in the wild in Britain (as far as we know) was almost too much! We all attempted to take photographs but most were out of focus because of poor light and camera shake. Mike Averill obtained the best ones.
We visited the site again on 23rd October 2012 and male land caddis still about but no females were found. These observations and others add confirmation that adult land caddis are active only in October.
Our land caddis records hold only one other from monad SO7867 in which Astley Mill is situated. This was by M. C. Clark (1984) who found larval cases in plant litter on 15th February 1980 under mat of grass and other vegetation in marshy ground, in itself an unusual habitat. However, Malcolm Clark was a mycologist who spent a great deal of time on his knees searching for micro-fungi and made others finds in similar places. Further searches are required in the area to add to records of Land Caddis distribution.
Reference
Clark, M.C. 1984. Proceedings of Birmingham Natural History Society. 25 (2):108.
Images
 Fig.01. Female Land Caddis, Rock Coppice. Rosemary Winnall
Fig.01. Female Land Caddis, Rock Coppice. Rosemary Winnall
 Fig.02. Male Land Caddis Rock Coppice. Rosemary Winnall
Fig.02. Male Land Caddis Rock Coppice. Rosemary Winnall
 Fig.03. Male Land Caddis, Hawkbatch. John Bingham
Fig.03. Male Land Caddis, Hawkbatch. John Bingham
 Fig.04. Male Land Caddis, Hawkbatch. John Bingham
Fig.04. Male Land Caddis, Hawkbatch. John Bingham
 Fig.05. Female Land Caddis, Hawkbatch. John Bingam.
Fig.05. Female Land Caddis, Hawkbatch. John Bingam.
 Fig.06. Ancient oak beneath which male and female Land Caddis were found. Harry Green
Fig.06. Ancient oak beneath which male and female Land Caddis were found. Harry Green
 Fig.07. Leaf litter beneath ancient oak where Land Caddis were found. Harry Green
Fig.07. Leaf litter beneath ancient oak where Land Caddis were found. Harry Green
 Fig.08. A pair of mating Land Caddis beneath ancient oak. Mike Averill
Fig.08. A pair of mating Land Caddis beneath ancient oak. Mike Averill
 Fig.09. A pair of mating Land Caddis beneath ancient oak. Harry Green
Fig.09. A pair of mating Land Caddis beneath ancient oak. Harry Green
Worcestershire Record | 33 (Nov 2012) page: 44-48 | Worcestershire Biological Records Centre & Worcestershire Recorders
White-Clawed Crayfish Population within the Wyre Forest – 2012 Update
Ann Hill and Graham Hill
Background
A baseline white-clawed crayfish survey of Bell Brook and Forest Lodge Stream, both within the Wyre Forest, was undertaken in 2010. Following the 2010 survey a monitoring programme was set-up to record the white-clawed crayfish in Wyre and in 2011 the first monitoring survey of the two watercourses was undertaken. In addition, surveys were also undertaken of Kingswood Stream and Longdon Stream which were also found to support white-clawed crayfish. During 2012 all four watercourses were surveyed as part of the monitoring programme and this article gives a summary of the survey findings.
The aims of the study are: to build up a record of white-clawed crayfish in watercourses across the Wyre forest; to detect any change in population range, abundance and dynamics; to trigger a response if any changes are undesirable; and to contribute to the knowledge of white-clawed crayfish population within the Wyre Forest, Worcestershire and the UK.
The monitoring targets are: a) Crayfish Population Range: White-clawed crayfish should be present in all four watercourses; and non-native crayfish should be absent from all four watercourses; b) Crayfish Population Abundance: There should be no statistically significant reduction in the number of white-clawed crayfish in the watercourse over three or more years in succession; the relative abundance of white-clawed crayfish in the watercourse should remain relatively constant from year to year; c) Crayfish Population Dynamics: Juvenile crayfish should be present in the watercourse; the proportion of juveniles (<25 mm carapace length) from a healthy population should be about 40%; and male and female white-clawed crayfish should be present in a watercourse.
Method
The 2012 survey replicated (where possible) the same team of surveyors, the same number and location of sample patches (fixed-area sampling) and the same amount of survey effort as surveys undertaken in previous years. The survey work was undertaken by two licensed surveyors and several assistants between 20 August and 18 September 2012 following published guidelines and best practice (Peay 2002).
Site
The site and the location of the four watercourses are shown in Fig. 01.
Results
Crayfish Habitat
Bell Brook retained excellent in-stream and bankside habitat. Water clarity was excellent and there was no significant change in water chemistry from the previous year.
Forest Lodge Stream water channel was difficult to access in places due to high water levels caused by heavy rain in the previous 24 hours. Water clarity was excellent and there was no significant change in water chemistry from the previous year.
Kingswood Stream had excellent in-stream and bankside habitat. The in-stream duck pond has been removed. Water clarity was very poor during both the daytime and nocturnal surveys and the habitat had a distinct smell of fertiliser. Water temperature was significantly lower in 2012 compared with measurements taken in 2011. There was no significant change in water pH with the previous year. However, Kingswood Stream had the highest recording of dissolved solids in the water of all four watercourses and levels had increased on 2011 measurements, although not significantly.
Longdon Stream had excellent in-stream and bankside habitat. The water was turbid on both survey visits and water clarity was poor. Longdon Stream had significantly higher water temperature and a significantly lower water pH and concentration of dissolved solids in the water in 2012 compared with measurements taken in 2011. Longdon Stream had the second highest recording of dissolved solids in water of all four watercourses but levels were lower than in 2011.
Crayfish Population Range
White-clawed crayfish were present in all four watercourses in 2012, Fig. 02. No non-native crayfish were found in any of the watercourses surveyed.
Crayfish Population Abundance
Bell Brook had a statistically significant increase in standard daytime manual observations in 2012 compared with observations in 2010 and 2011 (Kruskal-Wallis test, P-value 0.01), Fig. 03. There was no significant increase in night-time torching observations (Kruskal-Wallis test, total observations P-value 0.125).
Forest Lodge Stream observations across the three recording years have been very variable because of the influence of flow conditions, Fig. 04. The increase of crayfish observation in 2011 was the result of drought conditions concentrating the white-clawed crayfish into a smaller area and which also provided surveyors with greater access to the stream bed for recording. However, in 2012 the opposite was true and the high water levels limited surveyor access to the stream bed for recording and thereby reduced the survey efficiency. In addition, standard survey effort in 2011 was replaced by a rescue operation. There was a non-significant increase in standard daytime manual observations compared with observations in 2010 and 2011 (Kruskal-Wallis test, P-value 0.737). However, this result has to be tempered by the fact that there no standard survey in 2011.
There was a statistically significant decrease in night-time torching observations in 2012 compared with 2011 observations (Kruskal-Wallis test, P-value 0.042). Nonetheless, when compared with observations from when the monitoring study began, there was a non-significant decrease in night-time torching observations in 2012 compared with observations made in 2010 and 2011 (Kruskal-Wallis test, P-value 0.148).
Kingswood Stream had a non-significant decrease in both standard daytime manual observations (Kruskal-Wallis test, P-value 0.494) and in night-time torching observations (Kruskal-Wallis test, P-value 0.246) in 2012 compared with observations made in 2011, Fig. 05.
Longdon Stream had a non-significant decrease in night-time torching observations (Kruskal-Wallis test, P-value 0.658), Fig. 06. White-clawed crayfish were undetected using the standard daytime manual observations in 2012 in Longdon Stream.
Relative abundance of crayfish was calculated as the number of crayfish per ten refuges searched (Catch per Unit Effort, CPUE) or fifteen-minutes timed night-view, Fig. 07 and 08. The CPUE in Bell Brook increased and the population remained in the very high category. Forest Lodge Stream increased in CPUE using the standard method but decreased using the fifteen-minutes timed night-view method: overall the Forest Lodge crayfish population remained in the low to moderate population abundance category. Kingswood Stream had a decrease in CPUE but remained in the moderate population abundance category. Longdon Stream had a decrease in CPUE and had a negative change in population abundance category from low abundance to none or an undetected population.
Crayfish Population Dynamics
Juvenile white-clawed crayfish (<25mm carapace length) were caught and measured in Bell Brook, Forest Lodge Stream and Kingswood Stream, Table 1. No juvenile white-clawed crayfish (or adults) were caught in Longdon Stream. Juveniles were recorded in Bell Brook, Forest Lodge Stream and Longdon Stream using the night-time torching method, Table 2.
| Standard manual survey | ||||||||||
| Bell Brook | Forest Lodge Stream | Kingswood Stream | Longdon Stream | |||||||
| 2010 | 2011 | 2012 | 2010 | 2011 | 2012 | 2011 | 2012 | 2011 | 2012 | |
| Adult | 3 | 14 | 28 | 1 | 25 | 1 | 4 | 2 | 1 | 0 |
| Juvenile | 5 | 13 | 19 | 0 | 21 | 3 | 7 | 8 | 0 | 0 |
Table 1. . Comparison of adult and juvenile white-clawed crayfish caught and measured in four watercourses of the Wyre Forest during three years.
| Night viewing survey | ||||||||||
| Bell Brook | Forest Lodge Stream | Kingswood Stream | Longdon Stream | |||||||
| 2010 | 2011 | 2012 | 2010 | 2011 | 2012 | 2011 | 2012 | 2011 | 2012 | |
| Adult | 187 | 305 | 351 | 21 | 66 | 5 | 11 | 1 | 5 | 5 |
| Juvenile | 179 | 291 | 288 | 16 | 20 | 2 | 8 | 0 | 3 | 1 |
Table 2.. Comparison of adult and juvenile records recorded in four watercourses of the Wyre Forest during three years using night-view survey method.
There was no significant difference between the white-clawed crayfish carapace length of individuals caught and measured over the three years in Bell Brook, Forest Lodge Stream and Kingswood Stream (Bell Brook: Kruskal-Wallis test, sig. = 0.592, p = n.s., n = 2; Forest Lodge Stream: Kruskal-Wallis test, sig. = 0.793, p = n.s., n = 2; Kingswood Stream: Kruskal-Wallis test, sig. = 0.178, p = n.s., n = 1), Fig. 09. Neither was there any significant difference between year (2010, 2011 and 2012), watercourse (Bell Brook, Forest Lodge Stream, Kingswood Stream and Longdon Stream) and white-clawed crayfish carapace length (Two-way Anova (with replication) year P-value = .337; watercourse P-value = .278; year*watercourse P-value = .420).
Following two relatively stable years the crayfish population in Bell Brook showed an increase in average carapace size, indicating the possibility of an older population (skewness -0.161), Fig. 10. Fewer individuals were caught and measured in Kingswood Stream but those measured exhibited the same increase in average carapace length suggesting that Kingswood Stream may also have an ageing population (skewness -1.451), Fig. 11 There were too few observations to comment on population status in Forest Lodge Steam (Fig. 12) and Longdon Stream.
All crayfish that were caught were sexed, Table 3. Female and male white-clawed crayfish were recorded in Bell Brook and Kingswood Stream in 2012. However, only female white-clawed crayfish were recorded in Forest Lodge Stream and Longdon Stream.
| Bell Brook | Forest Lodge Stream | Kingswood Stream | Longdon Stream | |||||||
| 2010 | 2011 | 2012 | 2010 | 2011 | 2012 | 2011 | 2012 | 2011 | 2012 | |
| Male | 2 | 19 | 18 | 1 | 29 | 0 | 3 | 4 | 0 | |
| Female | 8 | 33 | 12 | 0 | 14 | 2 | 4 | 4 | 2 | |
Table 3. Comparison of male and female records recorded in four watercourses of the Wyre Forest during three years using standard survey method.
Discussion
There was no change in the geographical range of the white-clawed crayfish in Wyre Forest although there was an overall reduction in white-clawed crayfish observations across the Wyre Forest.
Bell Brook continued to support a very high population of white-clawed crayfish and there was a statistically significant increase in standard daytime manual observations in 2012 when compared with the previous two years. There was a mix of adults, both male and female, and juveniles indicating a healthy population structure although it may be slightly ageing. The results were likely due to favourable environmental conditions in the past twelve months and/or underlying natural population cycles (currently unknown).
Forest Lodge Stream has a small catchment that responds quickly to rainfall but with little storage capacity and as a consequence the white-clawed crayfish population is vulnerable to the peculiarities of the weather. The white-clawed crayfish population was recovering from the drought conditions of 2011 and had a low to moderate population abundance. Juveniles made up at least 40% of the white-clawed crayfish population so whilst no male crayfish were caught and recorded the varied size range indicated that Forest Lodge Stream continued to have a healthy but small population of white-clawed crayfish.
There was no significant change in the white-clawed crayfish population in Kingswood Stream and the presence of juveniles and a varied size range of adults (both male and female) was indicative of a breeding population. Catch per Unit Effort decreased although Kingswood Stream continued to have a moderate population abundance category for white-clawed crayfish. The in-stream duck pond has been removed during the past twelve months. Kingswood Stream had the highest recording of dissolved solids in the water of all four watercourses and levels had increased on 2011 measurements. Water quality remained an issue for the survival of the Kingswood Stream white-clawed crayfish population.
There was negative change in the population condition of white-clawed crayfish in Longdon Stream. No white-clawed crayfish were detected during the standard daytime survey and only six crayfish were recorded at night-time (eight were recorded in 2011). The two crayfish that were caught and measured were both female. No juveniles were recorded. The overall conclusion is that the breeding success and population structure is poor and that there is a problem with recruitment. Water clarity was poor with the second highest recording of dissolved solids in water of all four watercourses. Water quality is likely to be the critical issue for the survival of this population of white-clawed crayfish.
In conclusion, after three years of study there is still only limited information about the way the white-clawed crayfish of the Wyre Forest vary in abundance and spatially over time. The trends will need to be compared over many years and information on long-term variation will only gradually be obtained from continuing the monitoring of crayfish populations in Wyre Forest.
References
Hill, A., 2010. Atlantic Stream Crayfish in Wyre. In: Winnall, R. (Ed.) Wyre Forest Study Group Review 2010.
Hill, A., 2011. Crayfish of Wyre Forest – an update. In: Winnall, R. (Ed.) Wyre Forest Study Group Review 2011.
Peay, S., 2002. A Standardised Survey and Monitoring Protocol for the White-clawed Crayfish in the UK. Life in UK Rivers LIF 02-11-37. EC LIFE Programme, DG Env.D.1., Brussels, Belgium.
Peay, S., 2003. Monitoring the White-clawed Crayfish Austropotamobius pallipes. Conserving Natura 2000 Rivers Monitoring Series No. 1, English Nature, Peterborough.
Acknowledgements
We are grateful to the Wyre Forest Study Group and Forestry Commission who provided local knowledge and access details, Natural England and private landowners for access permissions and the volunteers, especially Mike Averill, Jane Scott and Rosemary Winnall, who assisted with the survey work.
Images
 Fig. 01. The location of the crayfish monitoring site in the Wyre Forest showing the four sample watercourses.
Fig. 01. The location of the crayfish monitoring site in the Wyre Forest showing the four sample watercourses.
 Fig. 02. Observations of white-clawed crayfish within four watercourses in the Wyre Forest, Worcestershire between 2010 and 2012.
Fig. 02. Observations of white-clawed crayfish within four watercourses in the Wyre Forest, Worcestershire between 2010 and 2012.
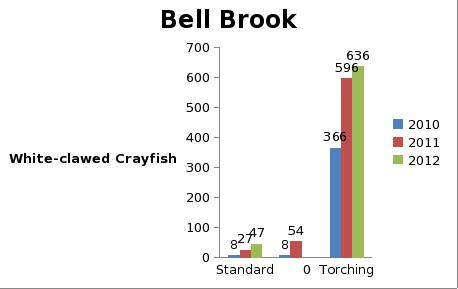 Fig. 03. Observations of white-clawed crayfish within Bell Brook, Wyre Forest between 2010 and 2012.
Fig. 03. Observations of white-clawed crayfish within Bell Brook, Wyre Forest between 2010 and 2012.
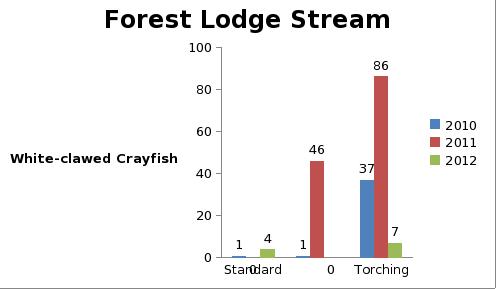 Fig. 04. Observations of white-clawed crayfish within Forest Lodge Stream, Wyre Forest between 2010 and 2012
Fig. 04. Observations of white-clawed crayfish within Forest Lodge Stream, Wyre Forest between 2010 and 2012
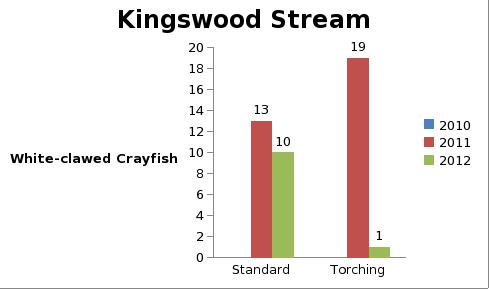 Fig. 05. Observations of white-clawed crayfish within Kingswood Stream, Wyre Forest between 2010 and 2012
Fig. 05. Observations of white-clawed crayfish within Kingswood Stream, Wyre Forest between 2010 and 2012
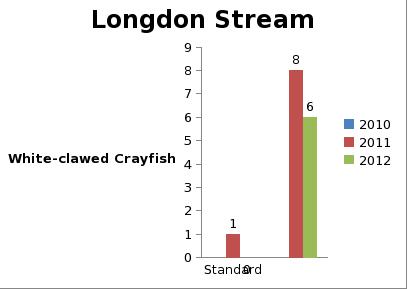 Fig. 06. Observations of white-clawed crayfish within Longdon Stream, Wyre Forest between 2010 and 2012.
Fig. 06. Observations of white-clawed crayfish within Longdon Stream, Wyre Forest between 2010 and 2012.
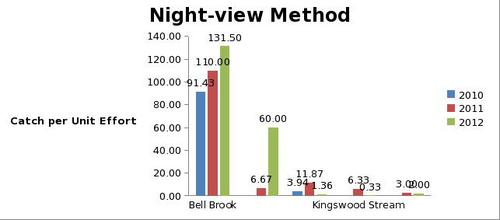 Fig. 07-1. Comparison of relative abundance of white-clawed crayfish recorded per unit effort searched (ten refugia) between 2010, 2011 and 2012 in four streams within the Wyre Forest.
Fig. 07-1. Comparison of relative abundance of white-clawed crayfish recorded per unit effort searched (ten refugia) between 2010, 2011 and 2012 in four streams within the Wyre Forest.
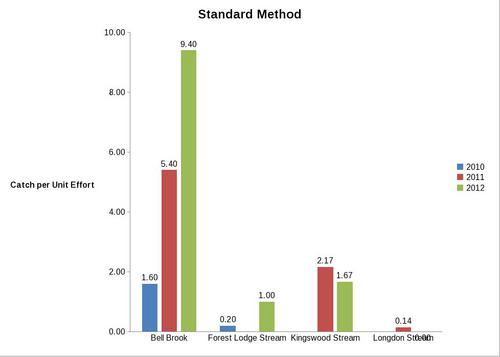 Fig. 07-2. Comparison of relative abundance of white-clawed crayfish recorded per unit effort searched (ten refugia) between 2010, 2011 and 2012 in four streams within the Wyre Forest.
Fig. 07-2. Comparison of relative abundance of white-clawed crayfish recorded per unit effort searched (ten refugia) between 2010, 2011 and 2012 in four streams within the Wyre Forest.
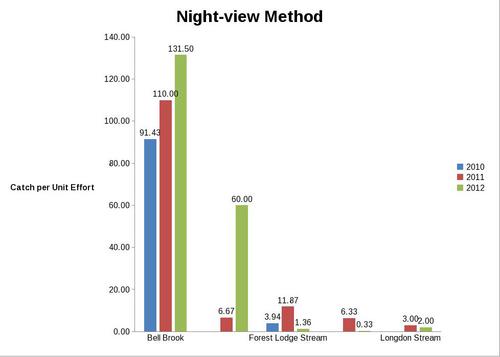 Fig. 08. Comparison of relative abundance of white-clawed crayfish recorded per unit effort searched (fifteen-minute timed search) between 2010, 2011 and 2012 in four watercourses within the Wyre Forest.
Fig. 08. Comparison of relative abundance of white-clawed crayfish recorded per unit effort searched (fifteen-minute timed search) between 2010, 2011 and 2012 in four watercourses within the Wyre Forest.
 Fig.09: Comparison of white-clawed crayfish carapace length of individuals caught and measured over the three years (2010, 2011 and 2012) in Bell Brook, Forest Lodge Stream, Kingswood Stream and Longdon Stream, Wyre Forest.
Fig.09: Comparison of white-clawed crayfish carapace length of individuals caught and measured over the three years (2010, 2011 and 2012) in Bell Brook, Forest Lodge Stream, Kingswood Stream and Longdon Stream, Wyre Forest.
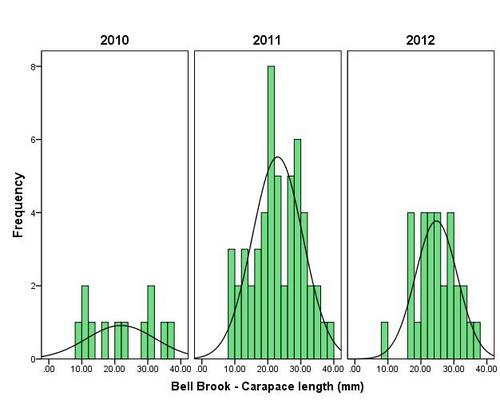 Fig. 10: Histogram showing size distribution (carapace length mm) of white-clawed crayfish population in Bell Brook, Wyre Forest in three survey years
Fig. 10: Histogram showing size distribution (carapace length mm) of white-clawed crayfish population in Bell Brook, Wyre Forest in three survey years
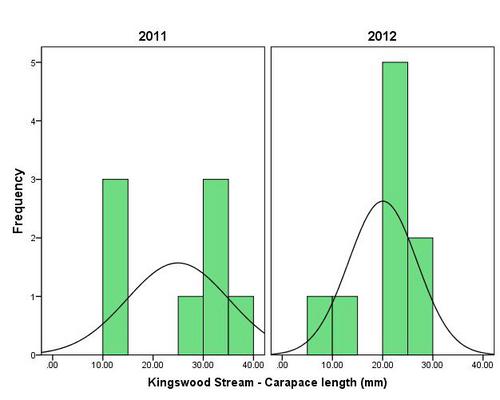 Fig. 11: Histogram showing size distribution (carapace length mm) of white-clawed crayfish population in Kingswood Stream, Wyre Forest in three survey years
Fig. 11: Histogram showing size distribution (carapace length mm) of white-clawed crayfish population in Kingswood Stream, Wyre Forest in three survey years
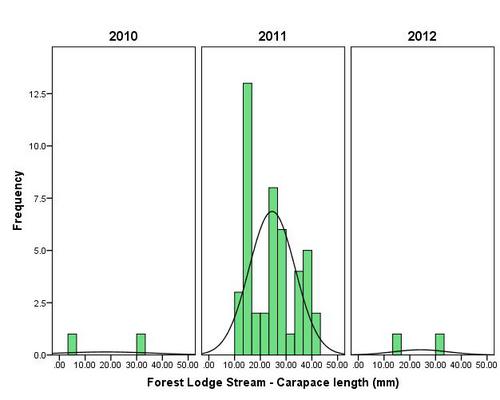 Fig.12. Histogram showing size distribution (carapace length mm) of white-clawed crayfish population in Forest Lodge Stream, Wyre Forest in two survey years
Fig.12. Histogram showing size distribution (carapace length mm) of white-clawed crayfish population in Forest Lodge Stream, Wyre Forest in two survey years
Worcestershire Record | 33 (Nov 2012) page: 53 | Worcestershire Biological Records Centre & Worcestershire Recorders
Worcestershire Bryophyte Group 2012
Ann Hill
This has been a busy year for Worcestershire bryophyte recording. During 2012 Worcestershire Bryophyte Group had several successful field recording days at five Worcestershire sites. The group recorded a total of 283 bryophytes (105 species: 11 liverworts and 94 mosses) in a variety of habitats. Five pleurocarpous mosses, Brachythecium rutabulum, Hypnum cupressiforme var. cupressiforme, Kindbergia praelonga and Rhynchostegium confertum and the liverwort Lophocolea bidentata, were ubiquitous and were recorded at every site visited. Another seven species, Amblystegium serpens var. serpens, Barbula unguiculata, Dicranoweisia cirrata, Fissidens taxifolius var. taxifolius, Orthotrichum affine, O. diaphanum and Rhytidiadelphus squarrosus, were recorded in four out of the five sites visited. A summary of the five sites visited and their bryophyte interest is given below.
The Knapp and Papermill (visited 18/02/12). We recorded 42 bryophytes (7 liverworts and 35 mosses) which, considering the atrocious weather conditions, and shortened recording day, was very good. Five bryophytes were a new record for the site: Acaulon muticum was recorded on an ant hill in Papermill Meadow (there are only four county records to date); Brachytheciastrum velutinum was found growing on soil covered rock along the main track; Dicranum scoparium was noted on the ground in the woodland; Didymodon fallax was growing on disturbed ground beside the track; and Pohlia wahlenbergii var. wahlenbergii was recorded in Papermill Meadow. We have now visited The Knapp and Papermill on four occasions and the total number of bryophytes recorded is 95 (17 liverworts and 78 mosses) which is an amazing record for one site and proves the value of re-visiting an area.
Northwick Marshes Site of Special Scientific Interest and Northwick Lido (visited 11/03/12). This was a joint field trip with the Border Bryologists. It was a glorious day for both weather and bryophytes and we recorded 65 species (4 liverworts and 61 mosses). The list included many bryophytes that we do not normally collect because we of the expertise of Mark Lawley and also because we were recording in a different habitat to our usual excursions. No rarities were found but Bryum violaceum, Cinclidotus fontinaloides, Ditrichum heteromallum, Entosthodon fascicularis, Hennediella stanfordensis, Orthotrichum sprucei and Scleropodium cespitans were interesting and valuable records for VC37.
Monkwood (visited 21/04/12). We recorded 31 bryophytes (5 liverworts and 26 mosses). This was not a large list but an interesting record. Species of note were Cryphaea heteromalla, Dicranella staphylina, Dicranum tauricum, Orthotrichum stramineum, Ulota phyllantha and Radula complanata. All are common species that are undoubtedly under recorded in the county.
The Devil’s Spittleful and Rifle Range (visited 22/09/12). We recorded 28 bryophytes (1 liverwort and 27 mosses) of which the majority were typical heathland mosses; overall the site was disappointing for bryophytes. The most interesting record was of Pseudoephemerum nitidum, an ephemeral moss that was recorded on the site of a bonfire. Pseudoephemerum nitidum is of frequent to common abundance throughout England but we have only five records to date in VC37 so this was an excellent find. In addition, the moss Campylopus fragilis formed dense tufts on crag ledges associated with the Devil’s Spittleful. There are only two records of Campylopus fragilis in VC37: one in the Wyre Forest and one at Broad Down, Malvern. One of our commonest and most cosmopolitan mosses, Ceratodon purpureus, was widely distributed across the site on acidic, well-drained sandy soil. Due to the absence of the usually abundant capsules, we confused this common moss with a number of acrocarpous mosses with triangular leaves and recurved margins before we correctly identified it.
Droitwich Community Woods (visited 13/10/12). We recorded 32 bryophytes (5 liverworts and 27 mosses) and the species list was one of the most interesting to date. The notable species was Syntrichia papillosa which was found growing on the bark of an ash. This species is infrequent throughout Britain and Ireland, but may be spreading. It is particularly associated with urban street trees and is often abundant on tree trunks in western towns. Syntrichia papillosa has only been recorded twice before in VC37: both occasions were in 2004 during the British Bryological Society meeting to the county.
For anyone wishing to discover a bit more about bryophytes, the following field days are planned and people are welcome to join us (although please email me ann@hillsecology.co.uk prior to the day to advise that you will be joining us).
Ravenshill Wood Nature Reserve, Alfrick – Saturday 24th November 2012
Worcestershire Wildlife Trust, Smite – Saturday 26th January 2013
Malvern Hills Castlemorton Common – Joint day with Border Bryologists – Sunday 17th March 2013
An Invitation to YOU
We are a small informal group (this year between five and eleven active participants) that visit Worcestershire sites to record and learn about bryophytes. Our broad aim is to assist everyone, especially those who are new to mosses and liverworts, to become more experienced and confident at identifying bryophytes. We have no formal membership or constitution and each person goes out at his/her own risk. Beginners are always very welcome, the only equipment needed is a hand lens (x10 or x20) and some paper packets for collecting specimens. In the field, the group will often work through the keys in the British Bryological Society’s Mosses and Liverworts of Britain and Ireland: a Field-guide (2010). Whilst we usually spend the morning concentrating on bryophyte identification, during the afternoon our attention tends to wander towards more general natural history, dependent on the interests of the individuals present on the day. Some people stay all day whilst some people leave at midday.
Please contact me for further details. ann@hillsecology.co.uk
Image
 Fig. 1. Worcestershire bryophyte group.
Fig. 1. Worcestershire bryophyte group.
Worcestershire Record | 33 (Nov 2012) page: 54 | Worcestershire Biological Records Centre & Worcestershire Recorders
Worcestershire Ancient Tree Record
Graham Hill
Records continue to be added to the database, which currently holds 3141 records of trees in Worcestershire (and a few just over the border). John Day kindly provided a long list of trees which he had recorded over several years; these are still being added to the database. Other records have been added as and when they arrive, and in a few cases where a record has been sent in of an existing record, the photographs have been very useful.
It is sad to see that the deadly Ash die-back fungus Chalara fraxinea has established itself in Britain, and according to The Guardian (6/11/2012) there are records in Worcestershire. The fungus is thought to have arrived from Denmark and attacks the Common Ash Fraxinus excelsior with devastating results. Three hundred and sixty four Ash trees are listed in the Worcestershire Ancient Tree Database scattered across the county (Fig. 1.). The largest Worcestershire Ash is on the boundary between Elmley Lovett and Hartlebury parishes – it has a girth of 6.8m and, according to the Forestry Commission Tree Age Calculator, could be over 500 years old. Of Worcestershire’s Ash trees, 27 have a girth of more than 5m, which suggest that many are more than 300 years old: it will be sad if we lose these trees from the landscape. It is sad, but there are echoes of Dutch Elm Disease here.
What can we do about our Ash trees? This is far from clear at the moment, but the Forestry Commission has issued advice (www.woodlandtrust.org.uk/en/about-us/faqs/your-woods/Documents/ash-fcalert.pdf). Perhaps the most important thing is BIOSECURITY and with a bit of effort we can slow the spread of Ash die-back.
It is unusual to find dated records of tree planting from several centuries ago. Brian Dawkins has spotted a date for Elms and Oaks planted along the western entrance drive to Madresfield Court. In her book ‘Madresfield – the real Brideshead’, Jane Mulvagh provides a date of 1646 for the planting of these trees, so those that remain are now 366 years old. Thank you, Brian, for that bit of information.
Do you ever wonder what use the Ancient Tree Record can be put to? Well a really good example emerged from Warwickshire recently (Falk 2012). You may be familiar with the proposed High Speed Railway (HS2) from London to Birmingham, it appears that the intended route is through Stoneleigh Park, Warwickshire, and will, potentially, affect a number of veteran trees there. Whilst I don’t want to be too controversial, but planting three trees for every veteran tree removed for the railway will take several centuries to be effective replacements!
The Worcestershire Ancient Tree Record was taken to the Tiddesley Wood Open Day again this year (2012), but this time we had the database running live onto a screen. This proved to be very popular and I had a lot of people stop by and talk about trees – generally local to them. We continue to collect records of Ancient Trees in Worcestershire, and put them onto the database eventually. If you know if any trees (or tree) that are old or notable please get in touch with us at Worcestershire Biological Records Centre records@wbrc.org.uk.
References
Falk, S. 2012. Tree-hunting in Warwickshire. British Wildlife, 24:21-30.
Mulvagh, J. 2008. Madresfield. The real Brideshead. Random House: London.
Image
 1. Distribution of Worestershire ash tree records, Worcestershire 18 Novemeber 2012.
1. Distribution of Worestershire ash tree records, Worcestershire 18 Novemeber 2012.
Worcestershire Record | 33 (Nov 2012) page: 66 | Worcestershire Biological Records Centre & Worcestershire Recorders
The Rediscovery of Frog Orchid (Coeloglossum viride (L.) Hartm.) in Worcestershire.
Roger Maskew
In early July this year while botanising on short, limestone grassland on the slopes of Broadway Park I came across seven scattered plants of Frog Orchid. This is one of the most important finds of recent years and is the first record of the species in the county for thirty-two years. Other orchid species present in the area were Early-purple Orchid Orchis mascula, Common Twayblade Neottia ovata, Common Spotted-orchid Dactylorhiza fuchsii, Bee Orchid Ophrys apifera and Pyramidal Orchid Anacamptis pyramidalis; other notable associates included Clustered Bellflower Campanula glomerata) Kidney Vetch Anthyllis vulneraria and Downy Oat-grass Avenula pubescens.
In southern Britain Frog Orchid is now mainly found from Sussex west to Wiltshire . However, apart from in the Peak District, it is a scarce plant in the Midlands. The most common habitat is well-drained calcareous grassland where it is usually seen as a small, greenish inconspicuous plant less than 15cm in height. It is therefore easily overlooked, although it can occur abundantly; for example I once found it difficult to avoid trampling numerous plants on the limestone at Durness NW Sutherland. On the other hand I have also found plants at least 25cm tall on wet rocky ledges at 900 metres on Ben More Assynt, Sutherland.
In Worcestershire, as elsewhere in much of England, Frog Orchid was also formerly frequent in unimproved pastures, but this habitat is now rare. It was first recorded in the county by Nash (1781) as abundant in meadows and pastures around Great Comberton and Pershore and the earliest specimen I have found was collected by William Mathews from Abberley in 1845. Lees (1867), in the first Flora for the county, lists it for odd sites in all four of his botanical districts and by the end of the 19th Century it had been found in at least twenty-five widely scattered sites with clusters of records from the Malvern and Wyre Forest districts. At the start of the 20th Century Amphlett & Rea (1909) stated “Downs, pastures, heaths. Local” and although there are few records from the 20th Century during the first two decades it was still to be found at Eastham in the Teme Valley and there are specimens in the herbarium of the Worcester Museum which were collected by Richard Towndrow from Little Malvern and Malvern Wells in 1910; it appears to have been last recorded in this district from West Malvern by Christine Dony in 1949. Humphreys (1917) mentions Woodgate near Bromsgrove and also that it was ”sparingly distributed” on the southern slopes of the Lickeys. Certainly during the first half of the 20th Century it was probably still more common and widespread than records suggest. Since then odd plants have been seen from time to time on the embankments of the earthworks at the summit of Bredon Hill. It was spotted here during a field meeting of the Worcestershire Naturalists’ in 1938 and again in 1979 and 1980. I have searched the exact spot several times in recent years without success. In 2002 I also unsuccessfully searched Eades Meadow with orchid expert Richard Bateman.
Suitable habitat still exists in the county and any damp unimproved grassland is worth searching, particularly those on base-rich soils, I have always thought the northern slopes of Bredon Hill look a good bet. Populations can fluctuate enormously from one year to the next, so any potential site is always worth visiting more than once.
References:
Amphlett, J. & Rea, C. 1909.The Botany of Worcestershire. Cornish Bros. Birmingham.
Humphreys, J. 1917. A Survey of the Flora of East Worcestershire. Proceedings of the Birmingham Natural History and Philosophical Society, Vol. XIV, pt. 2, pp. 61-86.
Lees, E. 1867. The Botany of Worcestershire. Worcestershire Naturalists Club. Worcester.
Nash, T.R. 1781. A catalogue of some rare plants. Collections for the History of Worcestershire. Vol. 1, pp. lxxxix-xc. John Nichols. London.
Image
 1. Frog Orchid Coeloglossum viride . Roger Maskew
1. Frog Orchid Coeloglossum viride . Roger Maskew
Worcestershire Record | 33 (Nov 2012) page: 30 | Worcestershire Biological Records Centre & Worcestershire Recorders
Brown Hairstreak caterpillar newly hatched
Paul Meers
I was lucky enough to be in the right place at the right time on Wednesday 2nd May 2012 at Feckenham Wylde Moor Reserve and checking our Brown Hairstreak eggs. Some were still intact but on one of the Blackthorn suckers two eggs had holes in them, one with this caterpillar right next to its egg. It must have only emerged just before I found it and so if I had been a little sooner I might have been able to see it half in half out which would have been a real bonus!
Images
 Fig. 1. Newly hatched brown hairstreak larva. Paul Meers.
Fig. 1. Newly hatched brown hairstreak larva. Paul Meers.
 Fig. 2. Newly hatched brown hairstreak larva. Paul Meers
Fig. 2. Newly hatched brown hairstreak larva. Paul Meers
Worcestershire Record | 33 (Nov 2012) page: 58-59 | Worcestershire Biological Records Centre & Worcestershire Recorders
Birds in Worcestershire – May to September 2012
Gavin Peplow
This summer has been virtually the wettest on record and certainly so in living memory. The few short brighter and warmer spells have been much appreciated, but in spite of (or on occasions perhaps due to) the weather, some interesting birds have been found.
Following April’s surprise influx, a further White Stork was seen in the Drakes Broughton area in late May, though this time a small ‘rubber’ ring was detected at the bottom of one of its legs, suggesting that it may have been of captive origin. Three Black-necked Grebes were at Bittell Reservoir for a day at the beginning of the month, with a further singleton there three weeks later. A drake Common Scoter also paused briefly at this site during the second week.
As is usual, May provided the bulk of the County’s wader highlights, albeit numbers are lower these days than a few years ago. Grimley attracted a wandering Avocet, parties of two and three Sanderling, a Little Stint and a Wood Sandpiper, with Lower Moor also visited by these last two species. A Bar-tailed Godwit lingered at Bittell for several days, with it or another, paying a short visit to Lower Park Farm to the north of Redditch. Late flooding at Longdon Marsh saw a peak of 16 Whimbrel drop in to feed, accompanied by a pair of Garganey. The star of the show though was a smart Red-necked Phalarope (Fig. 2.) which spent part of a day at Upton Warren before the regular forays of a local Peregrine encouraged it to move on – amazingly this is the fourth consecutive year that this species has visited the reserve!
A Sandwich Tern passed quickly through Bittell whilst two Black Terms were also seen there, with another reported over floodwater on the southern outskirts of Worcester.
A couple of Marsh Harriers drifted through Upton Warren, with the reserve also attracting a late Short-eared Owl at the start of the month, followed soon after by a passage Osprey.
A Nightingale sang from scrub near Diglis, Worcester for at least a couple of weeks and proved very popular and a couple of Quail calling late in the month at Woodrow and Honeybourne suggested that summer might finally be on its way!
Migration dwindled as June arrived, but a lingering Little Gull at Upton Warren was appreciated as was a Little Tern (by the few that saw it) that passed quickly through both there and Westwood. A party of 15 Crossbills on the Clent Hills were indicative of a small mid-year influx, whilst Red Kites continued to be seen frequently, with speculation that they should soon become a breeding species in the County (if this hasn’t in fact already happened!?)
The main event of June though, was an influx of Mediterranean Gulls to Upton Warren. Eleven were counted as being on site on one day at the end of the month with these birds lingering into July and at least 12 birds of varying ages involved in total. They were completely at home amongst the Black-headed Gull colony and one can only suspect that the display activity witnessed at this time is a pre-cursor to future breeding on the Reserve.
The persistent wet weather led to further flooding in the lower Severn and Avon valleys in early July and another four Mediterranean Gulls were seen amongst the large gathering of Black-heads at Bredon’s Hardwick. An adult Gannet (Fig. 3.) was found at Kemerton Lake at the start of the month, but sadly was sick and died overnight. Parties of three then two Common Scoter paused briefly at Upton Warren and two Sandwich Terns were detected calling as they flew over Kidderminster. The undoubted highlight of the whole summer however was an adult White-rumped Sandpiper that joined a Dunlin on receding floodwater at Grimley mid-month. Unfortunately for some it only stayed for a few hours one evening, but being only the second ever record for the County, it nevertheless attracted a lot of interest.
August began with a Great White Egret being located at Grimley (see www.carldayphotography.co.uk). (Fig. 1). It found the site much to its liking and stayed for a month, being joined by up to three Little Egrets at times. A Shag was also seen on one of the pits here briefly – a first record for the site. Several Black Terns passed through whilst the most interesting waders included individual Ruff at Longdon and Upton Warren, with a Spotted Redshank and a Knot also seen at this last site. A Wryneck was feeding on ants in a Stoulton garden towards the month end, whilst Marsh Harriers were seen over the two Nafford Reserves and at Upton Warren and an Osprey set up temporary residence at this last site into September (Fig. 4.).
The Great White Egret left Grimley in early September, but then presumably the same individual returned again 3½ weeks later. A Spotted Crake was found at Upton Warren Flashes one evening but then disappeared. A Sandwich Tern paused briefly at Grimley, as did a juvenile Marsh Harrier seen feeding on a dead Mute Swan there one evening. Up to four Garganey were equally elusive at this site, despite one bird lingering for ten days. Another Marsh Harrier, (perhaps the bird seen previously at Nafford) joined a spectacular gathering of at least 25 Buzzards and four Kestrels hunting rodents being disturbed during harvesting in a field near Bricklehampton in the second week.
The month concluded with three Common Scoter dropping in to Upper Bittell one afternoon and two Arctic Terns passing quickly through Bredon’s Hardwick.
Records compiled from reports received by Birdnews Midlands.
For all the latest information on birds currently within Worcestershire and the Midlands Region, call 09068-700247 (calls charged at 60p per minute).
Images
 Fig.1. Great White Egret at Grimley. Carl Day
Fig.1. Great White Egret at Grimley. Carl Day
 Fig.2. Red-necked Phalarope at Upton Warren.Peter Walkden
Fig.2. Red-necked Phalarope at Upton Warren.Peter Walkden
 Fig.3. Gannet at Kemerton Lake. Brian Stretch
Fig.3. Gannet at Kemerton Lake. Brian Stretch
 Fig.4. Osprey diving at Upton Warren. Alonza Garbett
Fig.4. Osprey diving at Upton Warren. Alonza Garbett
Worcestershire Record | 33 (Nov 2012) page: 60-62 | Worcestershire Biological Records Centre & Worcestershire Recorders
Birds in Worcestershire – November 2011 to April 2012
Gavin Peplow
With the exception of a cold snap in February, this (2011-2012) has been a much milder winter than the previous two. It’s also been very dry through to April when some wet and windy weather towards the end of the period providing some relief for farmers and gardeners alike, but proved somewhat disruptive to early breeding species.
A Black-necked Grebe that spent a few days at Westwood Lake at the beginning of November was a fairly typical record whilst a Red-throated Diver on the River Severn in Bewdley attracted a lot more attention, being the first record in the County for 14 years! Passage sea-duck included a Scaup at Pirton Pool and, more exceptionally, a sub-adult drake Eider along with a Red-breasted Merganser at Clifton Pit mid month.
The highlight of several interesting Geese records, were Beans (of the Tundra race) at Bittell Reservoir and then Bredon’s Hardwick at the end of the month. Elsewhere three White-fronted Geese appeared briefly at Lower Moor, with two again at Bittell and then a Brent Goose also paid a short visit to this last location.
Other highlights included a very mobile Great White Egret at Upton Warren, three Whooper Swans briefly at Clifton Pit, Hen Harriers passing over Chase End and through Westwood and at a peak of at least six Snow Buntings on the top of Worcestershire Beacon.
The Great White Egret again briefly visited Upton Warren at the beginning of December and the Tundra Bean Goose commuted between Ripple Pit and Bredon’s Hardwick into mid month. Further Red-breasted Mergansers were seen at Westwood during the run up to Christmas, with the second of these being an adult drake. A family party of four Bewick’s Swans took up residence at Pirton Pool and remained into the New Year. Elsewhere a Black Redstart was found on Worcestershire Beacon before all the Snow Buntings had finally moved on early in the month. Seven Waxwings were seen at Hewell Grange on a single date – an atypical record in a poor winter for the species. Gull watchers found a juvenile Glaucous at Throckmorton Tip in the third week and an Iceland Gull in the Westwood roost just after Christmas, whilst a Caspian Gull was also identified there and Little Gulls were seen at Grimley and again at Throckmorton Tip. A Brent Goose on the last day of 2011 was much welcomed by all the local Upton Warren birders, being the first record there for quite a few years.
The same Glaucous Gull returned to Throckmorton on New Years day and the Great White Egret again spent a very short time at Upton Warren during the second week. Mid-winter Little Egrets are still quite uncommon, so birds at Bittell and Upton Warren were good records. There were also unseasonal records of Knot at Upton Warren and Trimpley Reservoir, though neither bird lingered long. Up to 15 Crossbills were seen at Eymore Wood whilst Hawfinch numbers peaked at 20 at Chase End Hill during the month and a Black Redstart at Hartlebury Industrial Estate was a good find.
Unseasonal waders continued to be found into February during the cold spell, with another Knot, this time at Throckmorton Tip; three Sanderling also at this location and Grey Plovers at Bittell and then Grimley. Less unusual was a Ruff at this last location, whilst a couple of Avocets reappeared very early at Upton Warren from mid month.
There was a record influx of Iceland Gulls into northern Britain in December and January and by February a few of these trickled far enough south to be found in the County. Seven or eight birds were seen, with the highest concentration being three birds at Throckmorton Tip mid month. At the same time a Kumlein’s Gull was also found here – a still not fully classified form of Iceland Gull that normally shows some darker feathering in the wings.
March was very dry with some very sunny and warm weather during the second half of the month. The most exciting species was a Yellow-browed Warbler on the northern outskirts of Worcester. Having been heard calling early one morning by the finder, it remained faithful to hedgerows and gardens in the area well into April and was reported singing towards the end of its stay.
Early returning migrants were found widely across the County, with an Osprey spending time at both Upton Warren and Bittell attracting most attention.
Iceland Gulls continued to been seen regularly in the Throckmorton Tip area whilst a count of 76 Corn Buntings at Summerfield was the highest anywhere in the County for a good number of years.
April as always was a busy time with returning summer visitors and passage migrants making for interesting birding across the County. Most exceptionally was a party of nine White Storks at Abberley, photographed by the farmer on his mobile phone from a tractor as he was planting potatoes!! This is certainly an exceptional count for anywhere in the UK and a record for at least recent times, but sadly news filtered out well after these birds had moved on!
(Comment from Mike Morris who took the phone pictures (Figs. 1 to 9.) from a tractor cab. He said that the nine storks arrived together with some landing before the rest. The tractor drivers (nine of them!), who were planting a potato crop for Paines of Great Witley, did not know what the birds were at first, before working out that they were Storks. The birds arrived at around 3.30p.m. and stayed until after 7 p.m., feeding in the deep furrows, and getting really close to the tractors. They left together in the direction of Clows Top. (Editor’s insert. with thanks to Mike Southall for collecting this information.). The images can also be seen at www.temevalleywildlife.com
Also on the ‘large bird theme’, four Common Cranes circled over Ripple Pit one afternoon in the third week as they drifted east and typically were only seen by the lucky finders.
A male Hen Harrier was well photographed as it drifted over Upper Rochford at the beginning of the month, whilst Ospreys continued to be seen at Bittell and Upton Warren, with new arrivals over Drakes Broughton, Wassell Grove and Lower Moor.
A Hoopoe was typically elusive being only seen in flight over Coney Meadows near Droitwich mid month, whilst Ring Ouzel passage was strong with parties of up to 13 birds on Bredon Hill, seven on Walton Hill and lower numbers at several other sites.
Poor weather at the end of the month persuaded a few waders to drop into our wetland sites, with highlights being a Curlew Sandpiper at Grimley, Bar-tailed Godwit at Ripple and four Whimbrel at the Longdon Marsh Reserve. Small numbers of Arctic and Black Terns, along with several Little Gulls were found and a drake Garganey at Westwood was the first of the spring.
Records compiled from reports received by Birdline Midlands. For all the latest information on birds currently within Worcestershire and the Midlands Region, call 09068-700247 (calls charged at 60p per minute).
Images
 1. Storks at Abberley. Mike Morris
1. Storks at Abberley. Mike Morris
 2. Storks at Abberley. Mike Morris
2. Storks at Abberley. Mike Morris
 3. Storks at Abberley. Mike Morris
3. Storks at Abberley. Mike Morris
 4. Storks at Abberley. Mike Morris
4. Storks at Abberley. Mike Morris
 5. Storks at Abberley. Mike Morris
5. Storks at Abberley. Mike Morris
 6. Storks at Abberley. Mike Morris
6. Storks at Abberley. Mike Morris
 7. Storks at Abberley. Mike Morris
7. Storks at Abberley. Mike Morris
 8. Storks at Abberley. Mike Morris
8. Storks at Abberley. Mike Morris
 9. Storks at Abberley. Mike Morris
9. Storks at Abberley. Mike Morris
Worcestershire Record | 33 (Nov 2012) page: 69-73 | Worcestershire Biological Records Centre & Worcestershire Recorders
Plant Recording in SP13 in 2012
Bert Reid
Introduction
This year (2012) I decided to carry out a full survey of the vascular plants in the Worcestershire section of the 10×10 km square SP13. This area was primarily selected to update records for the Botanical Society for Britain and Ireland (BSBI) mapping project for the date class 2010 to 2019 in a part of the county where accurate boundaries are not easy to follow exactly on the ground.
The area surveyed is all within the Broadway parish, except for a very small area by the top of Fish Hill, where Worcestershire purchased part of Gloucestershire to create a parking and picnic site. This means that only parts of six tetrads are included, a little less than 10% of the hectad, although it feels like a lot more when climbing up and down Broadway Hill. Although it might be thought an insignificant part of Worcestershire, the Oolitic Limestone grasslands here are very important for locally scarce plants, and the area is directly connected to the main part of the Cotswold Hills to the south where many other rare and scarce plants are known, plants which could turn up here with luck.
I managed to find 542 species during the year. This compares with 604 species recorded between the start of the Worcestershire Flora Project in 1987 and the end of 2009. The 542 species are tabulated at the end of this report. The current records include 66 species not previously recorded in the area after 1986, suggesting that 128 post 1986 records have not been re-found. This seems to be a large number of losses, but many can be explained.
Lost Plants
The Worcestershire flora project ran from 1987 and never recorded more than 298 species in any year, that being 1992 when as well as the regular local recording by Terry Knight, a full flora outing was held there. It is almost impossible to fully cover the whole area at all seasons of a single year. In 2012 some plants must have simply been missed, especially plants only ever found once. Another reason for losses is changes to habitats. Fish Hill Quarry has changed significantly in recent years with the completion of quarrying, and plants like Deadly Nightshade, Blue Fleabane, Red Hemp-nettle, Corn Marigold and several garden escapes have disappeared. Arable fields have changed over the years, with increased use of fertilisers and herbicides. This almost certainly accounts for the loss of Venus’s Looking-glass and declines of a few other species. Another interesting loss is White Helleborine that was found in the Fish Hill picnic site between 1995 and 2006, although in the same site the Green-flowered Helleborine first noted here in 1992 is still present. Devil’s-bit Scabious was recorded in several grassy sites between 1979 and 1988, but has not been noted since, and changes to grassland management may be a factor.
The main groups of losses are clearly garden escapes and difficult or critical plants. About 45 species are obvious garden escapes or throw-outs. These plants are often casuals which change from year to year with horticultural fashions and with building policies. There have been significant changes in eastern part of Broadway, with many buildings renovated, and also a number of conversions in the Middle Hill House area. These changes have not only altered the species present but have also effected decisions about which plants are worth recording. The difficult and critical plants such as Hawkweeds, Eyebrights, Poplars, Willows, Roses, Brambles and Dandelions are outside the scope of most recorders, and even the most expert will only confidently deal with a few genera. This year I have done what I can with these, but there are still 25 taxa not found again.
Other losses are a miscellany of different species and reasons. Plants such as Silver Hair-grass, Pignut, Broom, Imperforate St.Johns-wort and Trailing St.Johns-wort are not usually found in calcareous areas but more often seen on more acid soils. Weather can be another reason. 2012 had a very dry spring and early summer but then became very wet. This followed a number of very dry summers, and may have accounted for failure to find Water-plantain, Marsh Marigold, Tufted Forget-me-not, Water Chickweed, Water-cress, Lesser Spearwort, Ragged Robin and Horned Pondweed. A number of lost plants had only been recorded at tetrad level before, since they were widespread in the county, making it difficult to follow up previous records.
Records before 1987 are much more difficult to assess. A further seven species were found in the period 1960-1986 but were not seen in 2012 – Downy Birch, Barberry, Wood Small-reed, Winter Aconite, Garden Peony, Adder’s-tongue and Pepper-saxifrage. These were all recorded by reliable botanists in well-defined areas. Earlier records are much more of a problem. Some are of very doubtful dates such as the Chalk Milkwort and other plants recorded by Hardaker from Broadway between 1902 and 1970. Others are not located sufficiently accurately to be sure they are in this hectad or even within the county. The 1898 record for Bird’s-nest Orchid was on a Worcestershire Naturalists Club field meeting from Buckland to Broadway, so could be in Gloucestershire. The final problem is doubts about identification. Cheddar Pink seems highly unlikely and Ribbed Melilot may have been confused with Tall Melilot. Despite the difficulties some of the historic records may re-appear, and I would not be surprised if plants like Chalk Milkwort, Fine-leaved Sandwort, Autumn Lady’s-tresses and Yellow Bird’s-nest crop up in the future. The next section on gains shows how plants with old records can be re-found with a bit of luck and effort.
Plant Gains
Eight of the gains have been recorded in the area prior to 1987. Five are of relatively recent times between 1979 and 1984. It is perhaps not surprising that Sainfoin, Norway Spruce, Grass Vetchling, Greater Butterfly-orchid and Green-winged Orchid have been found, as all of these are not rare in Worcestershire. My record of Yellow-wort from near Fish Hill Quarry had not been recorded since 1909 and was also not unexpected, but a small patch of Wood Vetch in Broadway Wood, last noted in 1908 was rather more of a surprise. Although this species is well-known further south in the Cotswolds and in parts of west Worcestershire, there have been no recent records in eastern Worcestershire. The oldest re-find for the hectad was Corn Buttercup, not seen since 1862 until I found a single plant in a bean field between Broadway Town and the A44. This was surprising for various reasons. Corn Buttercup is a scarce plant selected by the BSBI for checking in 2012, and I had followed up several old Worcestershire records without success. SP13 in Worcestershire has very little arable and this field was the only one with a crop suitable for the plant. Finally the plant has suffered such a great decline in recent years that it is now considered critically endangered nationally.
The other gains had never been recorded in the survey area. The most exciting plant of the year was Frog Orchid. I cannot claim this as my find, as it was spotted by Roger Maskew. He found it in steep calcareous pasture on Armley Bank, where he counted seven plants in two spots. He kindly told me of the find, so a week later I went to the same spots, where I found just four plants. A month later I found another two further down the slope. This was the first record in Worcestershire since a single plant was found in Bredon Hill fort in 1979, but this is in the East Gloucestershire vice-county and we need to go back to 1949 for the last record for our vice-county.
The other gains pale into relative insignificance. The A44 at Fish Hill provided quite a lot of new records. The salted edges of the main road produced Danish Scurveygrass, Buck’s-horn Plantain, Reflexed Saltmarsh-grass and Lesser Sea-spurrey, while the gravelly escape lane produced a surprise with a few plants of Sharp-leaved Fluellen. The salted road edge plants have increased for several years along Worcestershire’s main roads and it was good to record the extension of range in the far southeast, but the source of the Fluellen is not obvious. A single Round-leaved Fluellen was new in the same bean field as the Corn Buttercup, but despite considerable hunting, the Sharp-leaved could not be found there, but Black Mustard was added.
Common Centaury was found to be fairly widespread in road banks on the A44 in two monads, and I was surprised it had not been recorded before, but a small patch of Viper’s Bugloss by a footpath east of the underpass was more exceptional, as was a single plant of Hawkweed Oxtongue on the road bank. The Slender St.John’s-wort hidden away in the woods near Spring Hill House made up for the failure to find the Trailing St.John’s-wort, a plant of similar habitat requirements. Another surprising find was a few plants of Bristle Club-rush, growing with Toad Rush in a small damp flush area in rough grassland.
More detailed recording of difficult or critical plants accounts for the two subspecies of Tufted Hair-grass, the splitting of Glabrous and Common Whitlowgrass, Autumn Hawkweed, the hybrid between Hedge Bedstraw and Lady’s Bedstraw, and five Dandelion species. Other new plants include Chicory probably planted in improved grassland, a few Buckwheat plants in the bare edge of a flax field, and Eastern Rocket by a footway in Broadway Town. The other additions are nearly all garden plant escapes or alien trees and shrubs surviving in the wild. It is not appropriate to list these here, since a different set will probably turn up next year, but I was interested to see a single plant of Martagon Lily growing well away from any habitation half way up Coneygree Lane.
Plants found again
Nearly 480 plants are common to both the 1987-2009 and 2010-2012 periods. Here I will concentrate on the less widespread native or long established plants, especially those characteristic of the local habitats in SP13.
Limestone grassland is the most important habitat in SP13, and is characteristic of the higher parts of Broadway Hill. The richest areas are the SSSI grasslands by the Cotswold Way footpath and Armley Bank, both areas being unimproved sheep pasture. In Worcestershire, only Bredon Hill has similar geology. The unusual plants found here this year include Horseshoe Vetch, Squinancywort, Clustered Bellflower, Kidney Vetch, Carline Thistle, Autumn Gentian, Small Scabious, Common Rock-rose, Crested Hair-grass, Spiny Restharrow, and the more widespread Dwarf Thistle, Eyebright, Dropwort, Harebell, Spring Sedge, Meadow Oat-grass, Downy Oat-grass, Quaking Grass, Upright Brome and Tor-grass. On the lower slopes of the hill towards Broadway, the grassland is on landslip geology and has often been fertilised, giving a much less interesting flora. North of the A44, the grassland is again often improved, but here the locally uncommon Sheep’s Sorrel and Tormentil were found in small quantity.
The other major habitat in the survey area is woodland. Most of the woodland is old plantations, and there is little evidence of true ancient woodland. A typical local woodland is The Bank, stretching east and south above Middle Hill House. This has much Beech and Sycamore, with little shrub layer, and a restricted ground flora with Dog’s Mercury dominant. Yew, Woodruff, Sanicle and Wall Lettuce are locally frequent. Here and in similar local woods there are scattered Nettle-leaved Bellflower, Common Twayblade, Common Valerian and a good selection of ferns including Lady Fern and Hart’s-tongue. A special mention should be made of Coneygree Lane, which has a very different flora including Moschatel, Herb Paris, Wood Speedwell and Early-purple Orchid.
Aquatic and wetland plants are rare, with most interest around small flushes from springs and small streams trickling or rushing downhill. A flush in pasture above Broadway has a little Marsh Arrowgrass, Common Spike-rush and Small Sweet-grass in the abundant Rushes and Opposite-leaved Golden-saxifrage is found along the stream near Broadwaywood Cottages. The only other aquatic plant worth a mention is Great Horsetail, found in a number of spots where small streams reach the base of slopes.
Several locally rare plants were only found in one or two sites. Fern-grass turned up In Broadway town and on the A44; Small Toadflax on an arable edge and in the escape lane of the A44; Basil Thyme and Wild Mignonette both by the edge of Fish Hill Quarry; Dwarf Spurge in the bean field between Broadway and the A44; Round-leaved Crane’s-bill on the top of a stone wall beside the A44; a small patch of Common Gromwell on the woodland bank of the track from Middle Hill House by The Bank; and Rue-leaved Saxifrage on mossy roof or wall tops at Kites Nest and in Broadway town. Also in Broadway town were Common Stork’s-bill and Keel-fruited Cornsalad. The only site for Broad-leaved Helleborine was only just in the hectad on the track from Snowshill Road to Kite’s Nest, where 2 or 3 plants were on the edge of a fairly large colony mainly in SO93.
Just a few other plants are worthy of mention. The Ample-toothed Hawkweed has only been recorded in Worcestershire in SP13. It is a nationally rare introduction and was first confirmed locally in 2005 along the track from Middle Hill. This year I re-found the original plants and added three further spots nearby. Chinese Mugwort is an alien spreading from the London area and was first found in Worcestershire at the start of the track to Spring Hill House in 1990. It is still present in exactly the same spot, but the second county site had to wait until 2010 when I found it in two patches in Fladbury parish near Spring Hill Farm. Perhaps we need to hunt out more sites called Spring Hill! The final plant is the introduced True Service-tree by the verge at an edge of an orchard at the top of Broadway High Street. This was first formally recorded in 2011, although local people were aware of it much earlier.
Summary
This report shows some of the difficulties in recording and assessing the flora of a defined area. The past history of the site is often imperfectly known, even during the flora project period, and in earlier times the record is usually very poor. Even when well-localised records are available it is not always certain that losses are genuine. Plants are not always visible in a single year. The habitat may be temporarily unsuitable, with different crops in arable, grasslands grazed by the wrong amount or at the wrong time, small water features too dry or too wet, and woodlands cleared or replanted. Annual plants have good and bad seeding years and are well known to fluctuate in numbers dramatically. Other plants are known to rely on a seed bank or perennial underground system for many years. So not finding a plant is not necessarily mean a loss. Gains can be equally spurious. Just because nobody has recorded something before doesn’t mean it wasn’t there. Earlier botanist may have looked in the wrong place or wrong season, or may not have had the skills to recognise the plant. We all have blind spots with plant identification, and we all occasionally make silly, careless errors.
Whatever difficulties there are in assessing change, it is clear that the Broadway Hill area is still a very important part of Worcestershire’s flora. There are still a large number of rare or uncommon native plants where this area is the county stronghold. Most of the change recorded in 2012 gives a reasonably optimistic picture, with most losses being of casuals or non-native species.
Full list of 2012 species recorded
Acer campestre – Field Maple
Acer platanoides – Norway Maple
Acer pseudoplatanus – Sycamore
Achillea millefolium – Yarrow
Achillea ptarmica – Sneezewort
Adoxa moschatellina – Moschatel
Aegopodium podagraria – Ground-elder
Aesculus hippocastanum – Horse-chestnut
Aethusa cynapium ssp. cynapium – Fool’s Parsley
Agrimonia eupatoria – Agrimony
Agrostis capillaris – Common Bent
Agrostis gigantea – Black Bent
Agrostis stolonifera – Creeping Bent
Ajuga reptans – Bugle
Alcea rosea – Hollyhock
Alchemilla mollis – Soft Lady’s-mantle
Alliaria petiolata – Garlic Mustard
Allium ursinum – Ramsons
Allium vineale – Wild Onion
SandAlnus cordata – Italian Alder
Alnus glutinosa – Alder
Alnus incana – Grey Alder
Alopecurus myosuroides – Black-grass
Alopecurus pratensis – Meadow Foxtail
Anacamptis pyramidalis – Pyramidal Orchid
Anacanptis morio – Green-winged Orchid
Anagallis arvensis ssp. arvensis – Scarlet Pimpernel
Anemone nemorosa – Wood Anemone
Angelica sylvestris – Wild Angelica
Anisantha sterilis – Barren Brome
Anthemis punctata – Sicilian Chamomile
Anthoxanthum odoratum – Sweet Vernal Grass
Anthriscus sylvestris – Cow Parsley
Anthyllis vulneraria – Kidney Vetch
Antirrhinum majus – Snapdragon
Aphanes arvensis – Parsley-piert
Apium nodiflorum – Fool’s Water-cress
Aquilegia vulgaris – Columbine
Arabidopsis thaliana – Thale Cress
Arctium lappa – Greater Burdock
Arctium minus – Lesser Burdock
Arenaria leptoclados – Small Thyme-leaved Sandwort
Arenaria serpyllifolia – Thyme-leaved Sandwort
Armoracia rusticana – Horse-radish
Arrhenatherum elatius – False Oat-grass
Artemisia verlotiorum – Chinese Mugwort
Artemisia vulgaris – Mugwort
Arum maculatum – Lords-and-ladies
Asparagus officinalis – Garden Asparagus
Aspelenium scolopendrium – Hart’s-tongue
Asperula cynanchica – Squinancywort
Aster x salignus – Common Michaelmas-daisy
Athyrium filix-femina – Lady Fern
Atriplex patula – Common Orache
Atriplex prostrata – Spear-leaved Orache
Avena fatua – Wild-oat
Avenula pratense – Meadow Oat-grass
Avenula pubescens – Downy Oat-grass
Ballota nigra – Black Horehound
Bellis perennis – Daisy
Betula pendula – Silver Birch
Blackstonia perfoliata – Yellow-wort
Brachyglottis x jubar – Shrub Ragwort
Brachypodium pinnatum – Tor-grass
Brachypodium sylvaticum – False-brome
Brassica napus – Rape
Brassica nigra – Black Mustard
Briza media – Quaking-grass
Bromopsis erecta – Upright Brome
Bromopsis ramosa – Hairy Brome
Bromus hordeaceus – Soft-brome
Bryonia dioica – White Bryony
Buddleja davidii – Butterfly-bush
Buxus sempervirens – Box
Calendula officinalis – Pot Marigold
Calystegia sepium – Hedge Bindweed
Calystegia silvatica – Large Bindweed
Campanula glomerata – Clustered Bellflower
Campanula poscharskyana – Trailing Bellflower
Campanula rotundifolia – Harebell
Campanula trachelium – Nettle-leaved Bellflower
Capsella bursa-pastoris – Shepherd’s-purse
Cardamine flexuosa – Wavy Bitter-cress
Cardamine hirsuta – Hairy Bitter-cress
Cardamine pratensis – Cuckoo-flower
Carduus crispus – Welted Thistle
Carduus nutans – Musk Thistle
Carex caryophyllea – Spring Sedge
Carex flacca – Glaucous Sedge
Carex hirta – Hairy Sedge
Carex otrubae – False Fox-sedge
Carex pendula – Pendulus Sedge
Carex sylvatica – Wood-sedge
Carlina vulgaris – Carline Thistle
Carpinus betulus – Hornbeam
Castanea sativa – Sweet Chestnut
Catapodium rigidum – Fern-grass
Centaurea debeauxii – Chalk Knapweed
Centaurea montana – Perennial Cornflower
Centaurea scabiosa – Greater Knapweed
Centaurium erythraea – Common Centaury
Centranthus ruber – Red Valerian
Cerastium fontanum – Common Mouse-ear
Cerastium glomeratum – Sticky Mouse-ear
Cerastium tomentosum – Snow-in-summer
Chaenorhinum minus – Small Toadflax
Chaerophyllum temulum – Rough Chervil
Chamaecyparis lawsoniana – Lawson’s Cypress
Chamerion angustifolium – Rosebay Willowherb
Chelidonium majus – Greater Celandine
Chenopodium album – Fat-hen
Chenopodium polyspermum – Many-seeded Goosefoot
Chenopodium rubrum – Red Goosefoot
Chrysosplenium oppositifolium – Opposite-leaved Golden-saxifrage
Cichorium intybus – Chicory
Circaea lutetiana – Enchanter’s-nightshade
Cirsium acaule – Dwarf Thistle
Cirsium arvense – Creeping Thistle
Cirsium eriophorum – Woolly Thistle
Cirsium palustre – Marsh Thistle
Cirsium vulgare – Spear Thistle
Clematis vitalba – Traveller’s Joy
Clinopodium acinos – Basil Thyme
Clinopodium vulgare – Wild Basil
Cochlearia danica – Danish Scurvygrass
Coeloglossum viride – Frog Orchid
Conium maculatum – Hemlock
Convolvulus arvensis – Field Bindweed
Cornus sanguinea – Dogwood
Cornus sericea – Red-osier Dogwood
Corylus avellana – Hazel
Cotoneaster horizontalis – Wall Cotoneaster
Cotoneaster simonsii – Himalayan Cotoneaster
Crataegus laevigata – Midland Hawthorn
Crataegus laevigata x monogyna – Hybrid Hawthorn
Crataegus monogyna – Hawthorn
Crepis capillaris – Smooth Hawk’s-beard
Crepis vesicaria – Beaked Hawk’s-beard
Crocosmia x crocosmiiflora – Montbretia
Crocus tommasinianus – Early Crocus
Cyclamen hederifolium – Cyclamen
Cymbalaria muralis – Ivy-leaved Toadflax
Cynosurus cristatus – Crested Dog’s-tail
Dactylis glomerata – Cock’s-foot
Dactylorhiza fuchsii – Common Spotted-orchid
Daphne laureola – Spurge-laurel
Daucus carota ssp. carota – Wild Carrot
Deschampsia cespitosa ssp. cespitosa – Tufted Hair-grass
Deschampsia cespitosa ssp. parviflora – Tufted Hair-grass
Dianthus deltoides – Maiden Pink
Digitalis purpurea – Foxglove
Dipsacus fullonum – Wild Teasel
Dryopteris affinis – Golden-scaled Male-fern
Dryopteris borreri – Borrer’s Male-fern
Dryopteris dilatata – Broad Buckler-fern
Dryopteris filix-mas – Male-fern
Echium vulgare – Viper’s Bugloss
Eleocharis palustris ssp. vulgaris – Common Spike-rush
Elymus caninus – Bearded Couch
Elytrigia repens – Common Couch
Epilobium ciliatum – American Willowherb
Epilobium hirsutum – Great Willowherb
Epilobium montanum – Broad-leaved Willowherb
Epilobium obscurum – Short-fruited Willowherb
Epilobium parviflorum – Hoary Willowherb
Epilobium tetragonum – Square-stalked Willowherb
Epipactis helleborine – Broad-leaved Helleborine
Epipactis phyllanthes – Green-flowered Helleborine
Equisetum arvense – Field Horsetail
Equisetum telmateia – Great Horsetail
Erodium cicutarium – Common Stork’s-bill
Erophila glabrescens – Glabrous Whitlowgrass
Erophila verna – Common Whitlowgrass
Euonymus europaeus – Spindle
Eupatorium cannabinum – Hemp-agrimony
Euphorbia exigua – Dwarf Spurge
Euphorbia helioscopia – Sun Spurge
Euphorbia peplus – Petty Spurge
Euphrasia confusa x nemorosa – a hybrid Eyebright
Euphrasia nemorosa – Common Eyebright
Euphrasia sp. – Eyebright
Fagopyrum esculentum – Buckwheat
Fagus sylvatica – Beech
Fallopia baldschuanica – Russian Vine
Fallopia convolvulus – Black Bindweed
Fallopia japonica – Japanese Knotweed
Festuca ovina – Sheep’s-fescue
Festuca rubra – Red Fescue
Ficaria verna – Lesser Celandine
Ficaria verna ssp. fertilis – a Lesser Celandine
Filipendula ulmaria – Meadowsweet
Filipendula vulgaris – Dropwort
Fragaria vesca – Wild Strawberry
Fraxinus excelsior – Ash
Fumaria officinalis – Common Fumitory
Fumaria officinalis ssp. wirtgenii – a Common Fumitory
Galanthus nivalis – Snowdrop
Galega officinalis – Goat’s-rue
Galeopsis tetrahit agg. – Common Hemp-nettle [agg.]
Galeopsis tetrahit sens.str. – Common Hemp-nettle
Galium album – Hedge Bedstraw
Galium x pomeranicum – Hybrid Yellow Bedstraw
Galium aparine – Cleavers
Galium odoratum – Woodruff
Galium verum – Lady’s Bedstraw
Gentianella amarella – Autumn Gentian
Geranium dissectum – Cut-leaved Crane’s-bill
Geranium x oxonianum – Druce’s Crane’s-bill
Geranium molle – Dove’s-foot Crane’s-bill
Geranium pratense – Meadow Crane’s-bill
Geranium pusillum – Small-flowered Crane’s-bill
Geranium robertianum – Herb-robert
Geranium rotundifolium – Round-leaved Crane’s-bill
Geum urbanum – Herb Bennet
Glechoma hederacea – Ground-ivy
Glyceria declinata – Small Sweet-grass
Glyceria fluitans – Floating Sweet-grass
Glyceria notata – Plicate Sweet-grass
Hedera helix ssp. helix – Common Ivy
Helianthemum nummularium – Common Rock-rose
Helleborus foetidus – Stinking Hellebore
Helminthotheca echiodes – Bristly Oxtongue
Heracleum sphondylium – Hogweed
Hieracium argillaceum – Southern Hawkweed
Hieracium consociatum – Sociable Hawkweed
Hieracium sabaudum – Autumn Hawkweed
Hieracium sect. Sabauda – a Hawkweed
Hieracium sp. – a Hawkweed
Hieracium sylvularum – Ample-toothed Hawkweed
Hippocrepis comosa – Horseshoe Vetch
Holcus lanatus – Yorkshire-fog
Holcus mollis – Creeping Soft-grass
Hordeum murinum – Wall Barley
Hordeum secalinum – Meadow Barley
Hyacinthoides non-scripta – Bluebell
Hyacinthoides x massartiana – Hybrid Bluebell
Hypericum androsaemum – Tutsan
Hypericum calycinum – Rose-of-Sharon
Hypericum hirsutum – Hairy St. John’s-wort
Hypericum perforatum – Perforate St. John’s-wort
Hypericum pulchrum – Slender St. John’s-wort
Hypericum tetrapterum – Square-stalked St. John’s-wort
Hypochaeris radicata – Cat’s-ear
Ilex aquifolium – Holly
Ilex x altaclerensis – Highclere Holly
Inula conyzae – Ploughman’s-spikenard
Iris foetidissima – Stinking Iris
Isolepis setacea – Bristle Club-rush
Juglans regia – Walnut
Juncus articulatus – Jointed Rush
Juncus bufonius – Toad Rush
Juncus conglomeratus – Compact Rush
Juncus effusus – Soft Rush
Juncus inflexus – Hard Rush
Kickxia elatine – Sharp-leaved Fluellen
Kickxia spuria – Round-leaved Fluellen
Knautia arvensis – Field Scabious
Koeleria macrantha – Crested Hair-grass
Lactuca serriola – Prickly Lettuce
Lamiastrum galeobdolon ssp. argentatum – a Yellow Archangel
Lamium album – White Dead-nettle
Lamium amplexicaule – Hen-bit Dead-nettle
Lamium maculatum – Spotted Dead-nettle
Lamium purpureum – Red Dead-nettle
Lapsana communis – Nipplewort
Larix decidua – Larch
Larix x marschlinsii – Hybrid Larch
Lathyrus grandiflorus – Two-flowered Everlasting-pea
Lathyrus nissolia – Grass Vetchling
Lathyrus pratensis – Meadow Vetchling
Leontodon hispidus – Rough Hawkbit
Leontodon saxatilis – Lesser Hawkbit
Lepidium coronopus – Swine-cress
Lepidium didymum – Lesser Swine-cress
Leucanthemum vulgare – Oxeye Daisy
Leucanthemum x superbum – Shasta Daisy
Ligustrum ovalifolium – Garden Privet
Ligustrum vulgare – Wild Privet
Lilium martagon – Martagon Lily
Linaria purpurea – Purple Toadflax
Linum catharticum – Fairy Flax
Linum usitatissimum – Flax
Lithospermum officinale – Common Gromwell
Lobelia erinus – Garden Lobelia
Lolium multiflorum – Italian Rye-grass
Lolium perenne – Perennial Rye-grass
Lonicera nitida – Wilson’s Honeysuckle
Lonicera periclymenum – Honeysuckle
Lotus corniculatus – Common Bird’s-foot-trefoil
Lotus pedunculatus – Large Bird’s-foot-trefoil
Lunaria annua – Honesty
Luzula campestris – Field Wood-rush
Lycium barbarum – Duke of Argyll’s Teaplant
Lysimachia nummularia – Creeping-Jenny
Lysimachia punctata – Dotted Loosestrife
Mahonia aquifolium – Oregon Grape
Malus pumila – Apple
Malus sylvestris – Crab Apple
Malva moschata – Musk-mallow
Malva neglecta – Dwarf Mallow
Malva sylvestris – Common Mallow
Malva x clementii – Clements Tree-mallow
Matricaria chamomilla – Scented Mayweed
Matricaria discoidea – Pineapple Weed
Meconopsis cambrica – Welsh Poppy
Medicago arabica – Spotted Medick
Medicago lupulina – Black Medick
Medicago sativa ssp. sativa – Lucerne
Melica uniflora – Wood Melick
Melilotus altissimus – Tall Melilot
Melissa officinalis – Balm
Mentha aquatica – Water Mint
Mentha arvensis – Corn Mint
Mentha x villosa – Apple-mint
Mercurialis perennis – Dog’s Mercury
Milium effusum – Wood Millet
Moehringia trinervia – Three-nerved Sandwort
Mycelis muralis – Wall Lettuce
Myosotis arvensis – Field Forget-me-not
Myosotis sylvatica – Wood Forget-me-not
Neottia ovata – Common Twayblade
Nigella damascena – Love-in-a-mist
Odontites vernus – Red Bartsia
Onobrychis viciifolia – Sainfoin
Ononis repens – Common Restharrow
Ononis spinosa – Spiny Restharrow
Ophrys apifera – Bee Orchid
Orchis mascula – Early-purple Orchid
Origanum vulgare – Wild Marjoram
Oxalis acetosella – Wood-sorrel
Oxalis corniculata – Procumbent Yellow-sorrel
Oxalis exilis – Least Yellow-sorrel
Papaver dubium – Long-headed Poppy
Papaver lecoqii – Yellow-juiced Poppy
Papaver rhoeas – Common Poppy
Papaver somniferum – Opium Poppy
Parietaria judaica – Pellitory-of-the-Wall
Paris quadrifolia – Herb Paris
Pastinaca sativa – Wild Parsnip
Pentaglottis sempervirens – Green Alkanet
Persicaria capitata – Pink-headed Persicaria
Persicaria lapathifolia – Pale Persicaria
Persicaria maculosa – Redshank
Phleum bertolonii – Smaller Cat’s-tail
Phleum pratense sens.str. – Timothy
Phragmites australis – Common Reed
Picea abies – Norway Spruce
Picea sitchensis – Sitka Spruce
Picris hieracioides – Hawkweed Oxtongue
Pilosella aurantiaca – Fox-and-cubs
Pilosella officinarum – Mouse-ear-hawkweed
Pimpinella saxifraga – Burnet-saxifrage
Pinus sylvestris – Scots Pine
Plantago coronopus – Buck’s-horn Plantain
Plantago lanceolata – Ribwort Plantain
Plantago major – Greater Plantain
Plantago media – Hoary Plantain
Platanthera chlorantha – Greater Butterfly-orchid
Poa angustifolia – Narrow-leaved Meadow-grass
Poa annua – Annual Meadow-grass
Poa compressa – Flattened Meadow-grass
Poa humilis – Spreading Meadow-grass
Poa nemoralis – Wood Meadow-grass
Poa pratensis – Smooth Meadow-grass
Poa trivialis – Rough Meadow-grass
Polygala vulgaris – Common Milkwort
Polygonum arenastrum – Equal-leaved Knotgrass
Polygonum aviculare – Knotgrass
Polystichum setiferum – Soft Shield-fern
Populus alba – White Poplar
Populus x canescens – Grey Poplar
Populus nigra fastigiate cultivars – a Black-poplar
Populus x canadensis – Hybrid Black Poplar
Potentilla anserina – Silverweed
Potentilla erecta – Tormentil
Potentilla reptans – Creeping Cinquefoil
Potentilla sterilis – Barren Strawberry
Poterium sanguisorba ssp. sanguisorba – Salad Burnet
Primula veris – Cowslip
Primula vulgaris – Primrose
Prunella vulgaris – Selfheal
Prunus avium – Wild Cherry
Prunus domestica – Wild Plum
Prunus laurocerasus – Cherry Laurel
Prunus spinosa – Blackthorn
Pseudofumaria lutea – Yellow Corydalis
Pteridium aquilinum – Bracken
Puccinellia distans – Reflexed Saltmarsh-grass
Pulmonaria officinalis – Lungwort
Quercus x rosacea – Hybrid Oak
Quercus robur – Pedunculate Oak
Ranunculus acris – Meadow Buttercup
Ranunculus arvensis – Corn Buttercup
Ranunculus auricomus – Goldilocks Buttercup
Ranunculus bulbosus – Bulbous Buttercup
Ranunculus repens – Creeping Buttercup
Reseda lutea – Wild Mignonette
Reseda luteola – Weld
Rhinanthus minor – Yellow-rattle
Ribes rubrum – Red Currant
Ribes uva-crispa – Gooseberry
Robinia pseudoacacia – False Acacia
Rosa arvensis – Field Rose
Rosa x dumalis – a hybrid Rose
Rosa canina Group Transitoriae – Dog Rose
Rosa multiflora – Many-flowered Rose
Rosa rubiginosa – Sweet Briar
Rubus armeniacus – a bramble
Rubus caesius – Dewberry
Rubus cockburnianus – White-stemmed Bramble
Rubus fruticosus agg. – Bramble
Rubus idaeus – Raspberry
Rubus ulmifolius – a bramble
Rumex acetosa – Common Sorrel
Rumex acetosella – Sheep’s Sorrel
Rumex conglomeratus – Clustered Dock
Rumex crispus – Curled Dock
Rumex x pratensis – a dock
Rumex obtusifolius – Broad-leaved Dock
Rumex sanguineus – Wood Dock
Sagina filicaulis – Slender Pearlwort
Sagina procumbens – Procumbent Pearlwort
Salix alba – White Willow
Salix caprea – Goat Willow
Salix cinerea – Grey Willow
Salix fragilis – Crack Willow
Salix viminalis – Osier
Sambucus nigra – Elder
Sanicula europaea – Sanicle
Saxifraga tridactylites – Rue-leaved Saxifrage
Scabiosa columbaria – Small Scabious
Schedonorus arundinaceus – Tall Fescue
Schedonorus giganteus – Giant Fescue
Scorzoneroides autumnalis – Autumnal Hawkbit
Scrophularia auriculata – Water Figwort
Scrophularia nodosa – Common Figwort
Sedum acre – Biting Stonecrop
Sedum album – White Stonecrop
Sedum spurium – Caucasian-stonecrop
Sempervivum tectorum – House-leek
Senecio erucifolius – Hoary Ragwort
Senecio jacobaea – Common Ragwort
Senecio vulgaris – Groundsel
Sherardia arvensis – Field Madder
Silene dioica – Red Campion
Silene x hampeana – Hybrid Campion
Silene latifolia – White Campion
Silene vulgaris – Bladder Campion
Sinapis arvensis – Charlock
Sison amomum – Stone Parsley
Sisymbrium officinale – Hedge Mustard
Sisymbrium orientale – Eastern Rocket
Solanum dulcamara – Bittersweet
Solanum nigrum – Black Nightshade
Soleirolia soleirolii – Mind-your-own-business
Solidago canadensis – Canadian Goldenrod
Sonchus arvensis – Perennial Sow-thistle
Sonchus asper – Prickly Sow-thistle
Sonchus oleraceus – Smooth Sow-thistle
Sorbus aria – Whitebeam
Sorbus aucuparia – Rowan
Sorbus domestica – Service-tree
Spergularia marina – Lesser Sea-spurrey
Stachys sylvatica – Hedge Woundwort
Stellaria graminea – Lesser Stitchwort
Stellaria holostea – Greater Stitchwort
Stellaria media – Common Chickweed
Symphoricarpos albus – Snowberry
Symphytum x uplandicum – Russian Comfrey
Symphytum officinale – Common Comfrey
Syringa vulgaris – Lilac
Tamus communis – Black Bryony
Tanacetum parthenium – Feverfew
Tanacetum vulgare – Tansy
Taraxacum boekmanii – Bokman’s Dandelion
Taraxacum densilobum – Close-lobed Dandelion
Taraxacum hamatum – Hook-lobed Dandelion
Taraxacum lacistophyllum – Cut-leaved Dandelion
Taraxacum laticordatum – Decumbent Dandelion
Taraxacum oblongatum – Oblong-leaved Dandelion
Taraxacum oxoniense – Oxford Dandelion
Taraxacum pannucium – Green-stalked Dandelion
Taraxacum pseudohamatum – False Hook-lobed Dandelion
Taraxacum rhamphodes – Robust Dandelion
Taraxacum sp. – Dandelion agg.
Taraxacum subbracteatum – Dark-bracted Dandelion
Taraxacum subhamatum – Large Hook-lobed Dandelion
Taxus baccata – Yew
Thuja plicata – Western Red-cedar
Thymus polytrichus – Wild Thyme
Tilia cordata – Small-leaved Lime
Tilia cordata x platyphyllos (T. x vulgaris) – Lime
Tilia platyphyllos – Large-leaved Lime
Torilis japonica – Upright Hedge-parsley
Tragopogon pratensis – Goat’s-beard
Tragopogon pratensis ssp. minor – Goat’s-beard
Trifolium campestre – Hop Trefoil
Trifolium dubium – Lesser Trefoil
Trifolium hybridum – Alsike Clover
Trifolium medium – Zigzag Clover
Trifolium pratense – Red Clover
Trifolium repens – White Clover
Triglochin palustris – Marsh Arrowgrass
Tripleurospermum inodorum – Scentless Mayweed
Trisetum flavescens – Yellow Oat-grass
Triticum aestivum – Bread Wheat
Tussilago farfara – Colt’s-foot
Typha latifolia – Bulrush
Ulex europaeus – Gorse
Ulmus glabra – Wych Elm
Ulmus procera – English Elm
Urtica dioica – Common Nettle
Valeriana officinalis – Common Valerian
Valeriana officinalis ssp. sambucifolia – Common Valerian
Valerianella carinata – Keeled-fruited Cornsalad
Verbascum nigrum – Dark Mullein
Verbascum thapsus – Great Mullein
Veronica agrestis – Green Field-speedwell
Veronica arvensis – Wall Speedwell
Veronica beccabunga – Brooklime
Veronica chamaedrys – Germander Speedwell
Veronica filiformis – Slender Speedwell
Veronica hederifolia ssp. hederifolia – an Ivy-leaved Speedwell
Veronica hederifolia ssp. lucorum – and Ivy-leaved Speedwell
Veronica montana – Wood Speedwell
Veronica persica – Common Field-speedwell
Veronica serpyllifolia – Thyme-leaved Speedwell
Viburnum lantana – Wayfaring-tree
Viburnum opulus – Guelder-rose
Vicia cracca – Tufted Vetch
Vicia hirsuta – Hairy Tare
Vicia sativa ssp. segetalis – Common Vetch
Vicia sepium – Bush Vetch
Vicia sylvatica – Wood Vetch
Vicia tetrasperma – Smooth Tare
Vinca major – Greater Periwinkle
Viola arvensis – Field Pansy
Viola hirta – Hairy Violet
Viola odorata – Sweet Violet
Viola reichenbachiana – Early Dog-violet
Viola riviniana – Common Dog-violet
Viola x wittrockiana – Garden Pansy
Worcestershire Record | 33 (Nov 2012) page: 69-73 | Worcestershire Biological Records Centre & Worcestershire Recorders
Worcestershire Record | 33 (Nov 2012) page: 63-66 | Worcestershire Biological Records Centre & Worcestershire Recorders
Moonwort Botrychium lunaria near Lodge Hill Farm, Wyre Forest
John Robinson
Lodge Hill Farm
Moonwort Botrychium lunaria had been recorded previously from Worcestershire (Bredon Hill, Bewdley, Habberley valley, Hartlebury Common, Astley, Shelsley, Lickey monument) although somewhat erratically in the past. The record by Jordon in 1864 referring to “somewhere near Bewdley” may have been our site.
It had been regularly recorded from the North meadow at Lodge Hill Farm, but not every year, during my time as warden of the Wyre Forest National Nature Reserve (NNR) from 1976 and also during my subsequent 13 years of retirement. There are reports of 11 plants being found in 1992, and two in 1999 on the same site.
Moonwort was mentioned in the original Wyre Forest NNR Notification, along with Adder’s-tongue Fern, in relation to the meadows at Lodge Hill. To my knowledge it was never found at any other sites within the Wyre Forest, but may have been present. It always appeared in a relatively small area of the Lodge Hill meadow. Although not recorded every year it is very easily missed due to varying characteristics of grass growth in each season. It is also worth noting that it was not searched for in every year. At first emergence it is small, insignificant, and very, very easily missed.
Originally, before the declaration of the nature reserve, the meadow was managed in a quite a “haphazard” way. It was grazed by the odd milk cow and calf, perhaps a few sheep and a single horse which the then owner used with his cart as transport to market and for other farm work. Some other areas away from that were cut for hay – mostly by hand.. There were anthills scattered about the grassland.
Because it was difficult to replicate this type of ad hoc grazing etc the Nature Conservancy Council (NCC) prepared a management plan and the section where the moonwort occurred was included in the “cut for hay” section. A little later the area in the NE corner was left alone for ease of getting the hay baler round the cramped corner. Later we did manage to get a grazing regime into operation using sheep at first and then cattle later and the hay making in the North section of the North meadow ceased completely in approximately the mid 1990’s. The grazing was kept fairly light, as to avoid too much anthill poaching and to replicate as near as possible the historical light sporadic animal based management. In recent years the meadows have been grazed from about July, according to the season, and grazing moved around to various other meadows in the reserve and to those connected with the Grow with Wyre scheme.
In those early days the rabbit population was high and also the Fallow Deer herd was substantial, and this kept the sward ideally short after the larger animals were taken off at the end of their grazing stint.
During my warden days whenever the Moonwort it was found I would place an upturned hanging basket above the plant to prevent unwelcome grazing by whatever.
The way I found Moonwort in 2012 was quite amusing. Fran Flanagan, the Natural England Scientific Officer, visited the reserve and asked if I had seen any Moonwort this year. I said I hadn’t looked and he asked if I would show him the site roughly where it was usually found. We walked to the general area and I pointed to the ground and I said – “It’s usually around here somewhere. Oh – there it is, look!!!”
I am sure he thought I had found it previously but I hadn’t – it was pure luck. I am still not sure he believed me!
The date was May 2nd 2012and I covered it with the basket. I then photographed it every few days until its final demise on about July 18th. It was therefore visible for about 12 weeks from emergence. It has to be found in the early Spring when the grass is low but the danger is that over-searching at that time can easily lead to its trampling as at that stage it is only 4-5 cms in height. I am sure that well meaning searches in the past have lead to trampling and so inaccurate “absent” results may have been submitted. In the final stages the plant was approximately 11-12 cms high and if the grass had not been kept down for photography it would be almost impossible to see.
Calendar
May 2nd found and protected. 4-5 cms.
June 6th leaf lobe fully open.
June 28th spore shoot fully unfurled.
July 18th spore shoot limp and starting to shrivel.
Illustrated by Fig. 01. to Fig. 09. Fig. 10 shows spores blowing in the wind.
Images
 Fig. 01. Moonwort 02 May2012 Lodgehill. John Robinson
Fig. 01. Moonwort 02 May2012 Lodgehill. John Robinson
 Fig. 02. Moonwort 06 May2012 Lodgehill. John Robinson
Fig. 02. Moonwort 06 May2012 Lodgehill. John Robinson
 Fig. 03. Moonwort 01 June2012 Lodgehill. John Robinson
Fig. 03. Moonwort 01 June2012 Lodgehill. John Robinson
 Fig. 04. Moonwort 04 June2012 1 Lodgehill. John Robinson
Fig. 04. Moonwort 04 June2012 1 Lodgehill. John Robinson
 Fig. 05. Moonwort 10 June 2012 Lodgehill. John Robinson
Fig. 05. Moonwort 10 June 2012 Lodgehill. John Robinson
 Fig. 06. Moonwort 18 June2012 Lodgehill. John Robinson.
Fig. 06. Moonwort 18 June2012 Lodgehill. John Robinson.
 Fig. 07. Moonwort 25 June2012 Lodgehill. John Robinson
Fig. 07. Moonwort 25 June2012 Lodgehill. John Robinson
 Fig. 08. Moonwort 8 July2012 Lodgehill. John Robinson
Fig. 08. Moonwort 8 July2012 Lodgehill. John Robinson
 Fig. 09. Moonwort 16 July2012 Lodgehill. John Robinson
Fig. 09. Moonwort 16 July2012 Lodgehill. John Robinson
 Fig. 10. Moonwort producing spores 19 June 2012. John Robinson
Fig. 10. Moonwort producing spores 19 June 2012. John Robinson
Worcestershire Record | 33 (Nov 2012) page: 25 | Worcestershire Biological Records Centre & Worcestershire Recorders
Interesting Behaviour of Solitary Wasp, Cerceris arenaria
Jane Scott
For several years a colony of the attractive solitary wasp Cerceris arenariahas occupied nests between the old paving slabs at the back of my house in Astley Burf. Like all species of solitary wasp they provision their burrow with a particular type of prey and in the case of C. arvensis this is always weevils. The paralysed prey is carried in flight below the body of the wasp and is never released prior to being dragged down into the nest burrow. If the wasp has to relinquish hold on the prey even for a very short time it is discarded, thus avoiding the possibility that it has, in the meantime, been parasitized.
On 21st July 2012 I noticed one of the wasps arrive at its nest carrying a large Vine Weevil, which it tried for some time to pull into the nest hole but this was far too small to accommodate such a large prey. After a struggle it gave up the attempt and ejected the hapless, paralysed creature which just lay inert near the nest entrance. The wasp then emerged and immediately began furiously to dig out the burrow, presumably to accept larger prey. In so doing, it ejected a further three or four smaller weevils with which it had previously provisioned its nest. All of the prey was just abandoned around the entrance and no attempt was made to carry them back into the enlarged nest. The burrow continued to be used, presumably by the same individual (Figs. 1-4.)..
This action seemed to be an exaggerated response by this particular wasp as on previous observations prey that is too large would be discarded but no further steps taken to enlarge the burrow. Some ‘tidying up’ does occur from time to time following disturbance of the soil cone around the nest, but this is usually restricted to a small amount of clearance of the entrance to the burrow and the re-establishment of the neat cone around the top.
Images
 Fig. 1. Cerceris arenaria attempting to pull Vine Weevil into nest. Jane Scott
Fig. 1. Cerceris arenaria attempting to pull Vine Weevil into nest. Jane Scott
 Fig. 2. Cerceris arenaria and discarded weevils. Jane Scott
Fig. 2. Cerceris arenaria and discarded weevils. Jane Scott
 Fig. 3. Cerceris arenaria digging and discarded weevils. Jane Scott
Fig. 3. Cerceris arenaria digging and discarded weevils. Jane Scott
 Fig. 4. Cerceris arenaria digging and discarded weevils. Jane Scott
Fig. 4. Cerceris arenaria digging and discarded weevils. Jane Scott
Worcestershire Record | 33 (Nov 2012) page: 12 | Worcestershire Biological Records Centre & Worcestershire Recorders
Corizus hyocyami in a Malvern Garden
Frank Screen
I was pleased to see the photograph of Corizus hyocyami in the April edition of Worcestershire Record (32) as this confirmed my identification of this bug. On 25th May I found this bright red bug with black markings in a wild patch in my garden and immediately thought of Pyrrhcoris apterus. However my books did not confirm this. I photographed the bug again on 31st May, this time on an unopened dandelion seed head with its rostrum pushed into the seed head. On 1st June on the same dandelion was a mating pair (Fig. 1.). They were being molested by a Sloe Shieldbug Dolycoris baccarum who considered the spot to be its own and even tried to dislodge them by pushing its head under them.
Image

Fig. 1. Corizus hyocyami mating pair in dandelion seed head with Slow Bugs Dolycoris baccarum nearby. Frank Screen
Worcestershire Record | 33 (Nov 2012) page: 4 | Worcestershire Biological Records Centre & Worcestershire Recorders
Ash feeding moths
Tony Simpson
There has been speculation about the possible effects of Ash “Die Back” on our insect populations and I thought some information on local moths associated with this tree would be of interest.
Ash has few associated moths probably because it is a chemically well protected species and the larvae feeding on it need to be highly specialised to cope with this. Some Ash feeders also feed on related Privet species which are also in the Oleaceae family, as are Forsythia and Lilac.
Amongst the larger moths the common Centre-barred Sallow Atethmia centrago overwinters as an egg and feeds, mainly on the flowers, in the spring. Similarly the larva of the Dusky Thorn Ennomos fuscantaria feeds exclusively on this tree, as does the more uncommon Tawny Pinion Lithophane semibrunnea.
Amongst the micromoths the uncommon Zelleria hepariella feeds in a web on the young leaves in June; the common Prays fraxinella mines leaves and shoots causing drooping of young leaves in the spring; the recently separated Prays ruficeps also feeds in a web on leaves as well as mining shoots; the common Pyralid, Euzophera pinguis, feeds under the bark of mature trees, as does the rare Tortrix, Pammene suspectana.
All these species are therefore likely to be seriously affected.
The recently spreading Coronet Craniophora ligustri seems mainly to feed on Ash but is said to also feed on Privet. Ash Pug Eupithecia fraxinata is nowadays usually considered to be a form of Angle-barred Pug Eupithecia innotata which has coastal populations on Sea Buckthorn and Tamarisk in the U.K, but inland it is a rare Ash feeding resident with only three confirmed modern Worcestershire records. The nationally rare Barred Tooth-striped Trichopteryx polycommata feeds on Ash as well as more usually on Privet, but our last local record was in 1979. I have seen Privet Hawk Moth Sphinx ligustri larvae on Ash in West Wales but Privet is the usual food plant and we have only two modern records of this species.
The common micromoths Caloptilia syringella (also on Privet and Lilac) and Pseudargyrotoza conwagana (also in berries of wild Privet) are Ash feeders, as is Caloptilia cuculipennella (also on Privet) a recent arrival in the county.
It is possible loss of Ash will significantly affect these species but as they do apparently have alternative food plants they may not be so seriously affected. A lot will depend on how quickly and completely Ash is lost from the environment.
Worcestershire Record | 33 (Nov 2012) page: 4 | Worcestershire Biological Records Centre & Worcestershire Recorders
Worcestershire Record | 33 (Nov 2012) page: [[null]] | Worcestershire Biological Records Centre & Worcestershire Recorders
Buzzards, Hares and Crows
Mike Southall
On the 2nd of June 2012 I was discing a field near Elmley Lovett prior to planting. The field had some weedy areas, which had been sprayed off. I disturbed a young hare which ran away a short distance. A dark-plumaged Buzzard began hovering five metres over the hares location. The Buzzard swooped, but the hare managed to escape. A second bird, the female, with a white crescent on her chest, joined in the attack and after several tumbles the hare seemed doomed.
At this point I decided to even the odds, and set off diagonally across the field at full speed. I managed to get between the desperate hare and the birds close behind. As I dismounted the tractor, the hare continued to run and was attacked by a Carrion Crow which had joined in the melee. The young hare dived into thick cover under a large pylon in the centre of the field.
One of the Buzzards flew back to the pylon about ten minutes later. It perched low down and was obviously looking for the hare. It failed to spot it, and was eventually mobbed and driven off by a Carrion Crow.
The Buzzards nested nearby and had not been able to spot the hare until it was disturbed. A little later, I disturbed an adult hare in another field, around 300 metres away, which was possibly the mother.
Worcestershire Record | 33 (Nov 2012) page: [[null]] | Worcestershire Biological Records Centre & Worcestershire Recorders
Worcestershire Record | 33 (Nov 2012) page: 30-31 | Worcestershire Biological Records Centre & Worcestershire Recorders
Mining bees and their cleptoparasites
Geoff Trevis.
Whilst idly mulling over the factors affecting the distribution and status of mining bees I wondered if the cleptoparasites found on a given site would be good predictors of the species of mining bee that should be present. Clearly the cleptoparasites depend on the host bees but the converse is not true – the mining bees will indicate which cleptoparasites could be there but not which should be there. As a model I decided to use Nomada bees which mainly parasitise the nests of Andrena mining bees and occasionally Melitta species. Nomada are particularly useful because many are black and yellow and thus easily seen and collected whilst many Andrena are small or brown and camouflaged or both and hence are not so readily seen.
The initial port of call was to look up the host Andrena(s) for each Nomadaspecies. This provided the first hitch. The information for many species is difficult to find, even on the internet, and when data are available there seems to be considerable uncertainty. In many cases the nomadas are noted as parasitizing a couple or so species definitely and a few others probably or possibly. There is a good area of research here for anybody with the time and patience!
To narrow down the predictions of which species should be present a number of other factors had to be considered. Of these, the most important and obvious was the habitat and the presence of flowers on which the mining bees are known to forage. Andrena lapponica, for example, is known to forage on bilberry hence it will not be found on sites without this plant. At the moment its only known Worcestershire location is the Wyre Forest though I have hopes of finding it elsewhere e.g. on the Malvern Hills where there are extensive patches of bilberry. However, other factors to be considered are climate and the geographical distribution of the species in question. The bilberry on the Malverns is exposed to the elements at altitude and this might not suit A. lapponica – certainly I have yet found it despite some searches. Habitat too is important as many andrenas are fairly restricted in their habitat choice. For instance, the common A. chrysosceles is found in woodland clearings and along rides whilst A. fuscipes is an ericaceous heath specialist.
It would be tedious to list the results for all 47 sites for which I analysed the data but the results for a few illustrate the predictive value of cleptoparasites and the difficulty in accumulating data. Some sites have received a lot of attention and are well recorded but many others have received scant attention despite their potential for hymenoptera (and other species!). Again it would be tedious to list all the sites but it is worth noting those for which fairly comprehensive lists are available. These are Beckford Gravel Pit, Rosemary Winnall’s land at Bliss Gate, Caddicroft Farm near Drakes Broughton, meadows at Drakes Broughton, Hartlebury Common, Kemerton Lake reserve, Kempsey Common, Rough Hill Orchard near Defford, Tiddesley Wood near Pershore, Windmill Hill near the Littletons, and Wyre Forest. The sharp-eyed might notice the absence of Devil’s Spittleful but I will come back to that later. I will not list the other sites but I have selected Burlish Top and Grafton Wood to illustrate those with intermediate levels of recording and Burley Dene Meadows and the Knapp and Paper Mill Reserve (surprisingly) as ones with poor levels.
Nine species of Nomada bees and twenty four species of Andrena have been recorded at Hartlebury Common. However, four of the Andrena have not been recorded there since the mid-nineteen nineties. Comparison of the lists for the two groups shows that potentially a further seven species of Andrena should be there, these being Andrena carantonica, A. thoracica, A. clarkella, A. apicata, A. minutula, A. semilaevis and A. subopaca. A. carantonica is accorded universal status; A thoracica is a heathland specialist; A. clarkella and A. apicata (Na) are early species mainly associated with sallow; and A. minutula, A. semilaevis and A. subopaca are very small species and are the hosts for Nomada flavoguttata. Despite the lack of records for the last three species N. flavoguttata is present.
Tiddesley Wood has records of eight species of nomada and fifteen species of Andrena. Nonetheless a further six Andrena species should be found and two more, A. apicata (Nb) and A. varians (Nb) could also be present. The six relatively common species indicated by the Nomada cleptoparasites are A. nigroaenea, A. angustior, A. flavipes, A. semilaevis, A. clarkella and A. helvola. A. nigroaenea, A. angustior and A. varians and all are widely distributed and polylectic. As noted above, A. clarkella and A. apicata are early species on sallow whilst A. semilaevis is common and particularly associated with hogweed. The last of the expected species, A. helvola, is the only woodland specialist.
Burlish top, an intermediately recorded site, has three species of Nomadaand four species of Andrena. One Nomada, N. flavopicta, parasitises Melittaspecies rather than Andrena and a suitable host, Melitta leporina, has been recorded. Undoubtedly more Nomada remain to found but even with those already noted another four Andrena could be present. These are A. bimaculata, A. tibialis, A. fuscipes and A. denticulata. A. bimaculata and A. tibialis are uncommon (Nb and Na respectively) but A. bimaculata is relatively common and seems to favour sandy soils of the type found on Burlish Top. A. fuscipes is a fairly common heather heath specialist and A. denticulata is a widespread but local species foraging on yellow asteraceae.
There are records of four Nomada species at Grafton Wood but only two records of Andrena. Potentially A. carantonica, A. nigroaenea, A. minutula, A. semilaevis, A. subopaca, A. bicolour and A. angustior could be present and undoubtedly some of these are, to support the Nomada population.
The poorly recorded sites have few records either of Nomada or Andrenaspecies. Burley Dene Meadows has received one recording day visit and the only cleptoparasite noted was Nomada flava. The hosts for this species are A. carantonica and A. nigroaenea. A. carantonica was found but A. nigroaenea might also be there. The variety of habitats at Knapp and Papermill should have a wide range of mining bees and their parasites. However, only three species of Nomada, N. flava, N. favoguttata and N. marshamella have been recorded accompanied by four Andrena species, A. helvola, A. flavipes, A. fulva and A. haemorrhoa. Based only on the limited range of cleptoparasites found the Andrena mining bee list could be extended to include A. carantonica, A. nigroaenea, A. minutula, A. semilaevis, A. subopaca and A. trimmerana. These and most of the other under recorded sites would amply repay further surveys.
Devil’s Spittleful is an interesting case. As one of our premier heathland sites, and hence also one of our best sites for hymenoptera, it should be in the well recorded category with a long list of species. Indeed at first glance this would appear to be the case. However, a look at the dates of the records indicates a different picture. Several rare species of Andrena have been found there but for most of these the latest record is in the mid 1990’s when surveys were being carried out by Dr. Michael Archer and Colin Plant. Since then little work has been done, which is unfortunate as the opportunity to assess the impact of recent management work has been lost – we have no baseline.
Overall, this brief review of the Andrena mining bees and their Nomadacleptoparasites shows that using the Nomada species to identify missing Andrena is of only limited value, even on well recorded sites. This is because some Nomada bees may have several hosts and because even with sustained recording Nomada species are missed. However, it is worthwhile having some indications of species that may be present and are worth looking for.
Another question was raised by looking carefully at the Andrena/Nomadapairs. Why is one of our rarest nomad bees, N. signata (RDB2) associated with one of our commonest mining bees, A. fulva?
Worcestershire Record | 33 (Nov 2012) page: 30-31 | Worcestershire Biological Records Centre & Worcestershire Recorders
Worcestershire Record | 33 (Nov 2012) page: 62 | Worcestershire Biological Records Centre & Worcestershire Recorders
A flock of Hadada Ibises (Bostrychia hagedash (Latham, 1790)) (Ciconiiformes: Threskiornithidae) in the Bredon Hill area of Worcestershire during 2005
P.F. Whitehead
Paul F. Whitehead
Moor Leys, Little Comberton, Pershore, Worcestershire WR10 3EH email: paul@thewhiteheads.eu
Preamble
This account stems from an observation that took place over seven years ago and deals with a Worcestershire sighting of a group of Hadada Ibises Bostrychia hagedash (Latham, 1790), an African member of the stork family for which there are apparently no published British records of wild birds. As any scientist will confirm, there is an onus of responsibility to publish data and these might, at some later date, be placed in the context of changing population dynamics. There is some evidence to suggest that Hadada Ibises are presently expansive and in parts of South Africa they are domesticated scavengers. In Africa the species has a huge sub-Saharan range extending in the north to Ethiopia.
Discussion
At 21.25 hrs near to dusk on 6 June 2005 an extraordinary cacophony of sound was heard approaching Little Comberton settlement from the south (SO 94 30 m O.D.). As the sound drew nearer people stopped working on their allotments and gathered to await sight of what might be producing it. Eventually six birds were observed, gaining height from the south and overflying the edge of the village following the low contours of Bredon Hill; the syncopated cacophony can only be described as reminiscent of football rattles. The exact flight-call sound [not the frequently-used raucous single crow-like call] can be heard at 24 seconds into the video http://www.youtube.com/watch?v=ErhMjSevu14&feature=related and at 30 seconds into the video http://www.youtube.com/watch?v=u7mUevTfJnE&feature=player_detailpage.
Fortunately one of the birds peeled off to within 300 m of me when it was possible to see from its extended neck and down-curved linear beak that it was a rather chunky-looking ibis; the wings however seemed to be somewhat rounded and the rather languid flapping flight showed a very slight rolling motion without any extended glides. Due to the poor light I can only describe the birds as being ‘dark’ and appearing somewhat larger than a crow. Having little idea of which ibis they might be I contacted Harry Green since one of them passed over his home. I was later informed by Brett Westwood that they could well be African Hadada Ibises which are named onomatopoeically. This was confirmed when I subsequently heard sound recordings.
All the birds were evidently in good condition and showed marked social cohesion. They seemed to have arisen from a point along the wooded foot-slopes of Bredon Hill nearer to Elmley Castle, to have over-flown Little Comberton and then continued over Great Comberton, which might have taken them to the floodplains of the River Avon.
Hadada Ibises are occasionally kept in captivity in European collections. The related African Sacred Ibis Threskiornis aethiopicus (Latham, 1790) is naturalised in continental Europe after having escaped from collections there and is now showing up more frequently in Britain
Worcestershire Record | 33 (Nov 2012) page: 62 | Worcestershire Biological Records Centre & Worcestershire Recorders
Worcestershire Record | 33 (Nov 2012) page: 57 | Worcestershire Biological Records Centre & Worcestershire Recorders
A Glossy Ibis Plegadis falcinellus (L., 1766) (Ciconiiformes: Threskiornithidae) in flight over Winchcombe, Gloucestershire
P.F. Whitehead
Paul F. Whitehead, Moor Leys, Little Comberton, Pershore, Worcestershire WR10 3EH. email: paul@thewhiteheads.eu
At 1209 hrs on 4 October 2012 a Glossy Ibis (Plegadis falcinellus) passed over Winchcombe town, Gloucestershire (SP 02). Flying at a height of only some 70 m it was possible to discern the uniformly vinous plumage against a clear blue sky and the extended neck, long beak and trailing legs, a unique and unmistakeable combination.
The Glossy Ibis flies in a distinctive fashion. The wings appear to be rigid, flexing at the axilla rather than the carpus, with a slight flicking action. Migrating birds, of which the largest flock I have seen numbered 117, have a peculiar and distinctive flight action with sequential down-glides, creating a strange effect of rise and fall in a flock. The Winchcombe bird did not glide at all.
The appearance of this bird, which in all probability passed into Worcestershire, immediately preceded a significant influx of Glossy Ibises into southern England which then penetrated northwards.
Worcestershire Record | 33 (Nov 2012) page: 57 | Worcestershire Biological Records Centre & Worcestershire Recorders
Worcestershire Record | 33 (Nov 2012) page: 33-34 | Worcestershire Biological Records Centre & Worcestershire Recorders
A review of Callicerus sp. (Col., Staphylinidae) in the English midland region
P.F. Whitehead
Observing the athetine rove beetle Callicerus rigidicornis (Erichson, 1839) in flight on the Birlingham floodplain during April 2012 reminded me how scarce and poorly understood this genus of beetles is in Britain. Most British overviews of Coleoptera confirm the scarcity of Callicerus spp. which occur mostly to the south of the Pennines. It seems opportune to review what is known of Callicerus in the midland area and make some assessment of its conservation status.
Two species of Callicerus occur in Britain and these are by far the most widespread of the seven European species. The smaller Callicerus obscurusGravenhorst, 1802 attains some 3.0 mm in length whilst large female C. rigidicornis may occasionally exceed 5.0 mm in length. Males of C. rigidicornis may be distinguished from females by the short keel on their first abdominal tergite, a feature lacking in male C. obscurus. The males of C. obscurus can be distinguished by their conspicuously elongate apical and penultimate antennomeres, much more so than those of the females.
Callicerus obscurus Gravenhorst, 1802
Callicerus obscurus is known to me from eight Worcestershire and two Warwickshire specimens. These include female spring dispersants from February to April (the one in flight at Broadway on the rather early date of 23 February 1990 coincided with one of the warmest February days on record there when the temperature reached 19oC). Only one male has been observed, in flight at Little Comberton on 30 April 2010, indicative of male spring dispersal.
Ecology. In Worcestershire, female C. obscurus have been observed wintering in litter and moss in ancient escarpment woodland blocks on the south side of Bredon Hill where at Westmancote three examples were found by woodland springs during March 1992. During 1992 and 1994 examples were found amongst winter wheat on arable land at ADAS Drayton, Warwickshire (Corbett & Whitehead, unpublished; cited by Lane, Wright & Forsythe, 2002) where Jurassic clays inhibit water transmission. This range of habitats is mirrored exactly by Duff (1993) citing seven records from Somerset spanning a period of 107 years. Atty (1983) could cite only two Gloucestershire records including a 1914 example also in a well-incised wooded Cotswold Hill valley. A relatively clear picture can therefore be reconstructed of a localised, possibly under-recorded insect with a preference for somewhat humid closed-canopy woodland shrouding water bodies or artesian drainage.
Regional records. Single specimens except where stated.
Warwickshire (Corbett & Whitehead, unpublished; cited by Lane, 2002).
Drayton ADAS, SP15, 40 m O.D., in winter wheat on arable farmland, 9 March 1992 and 9 June 1994 (crop in ear).
Worcestershire
Broadway, SP03, 91 m O.D., wooded valley, in flight, 23 February 1990.
Westmancote, SO93, 130 m O.D., woodland floor litter, 9 March 1992.
Westmancote, 145 m O.D., near or at springs in ancient woodland, 13 March 1992, 16 March 1992, 18 March 1992 (2).
Westmancote, 145 m O.D., scrub woodland, at weeping cut of freshly ‘ringed’ ash tree, 10 May 1994.
Little Comberton, SO93, 30 m O.D., in flight near hill stream, 30 April 2010.
Callicerus rigidicornis (Erichson, 1839)
All three Worcestershire specimens of C. rigidicornis known to me are females encountered during their spring dispersal; the records range from the Cotswold Hills to the Malvern Hills implying that the species is localised but widespread. It should not be presumed that this dispersal is long-distance or anything other than local.
Ecology. In France C. rigidicornis is most usually associated with leaf litter in deciduous woodland (M. Tronquet, in litt., 24 April 2012) and there seems little doubt that this species is somewhat hygrophilous like its congener and close relatives in the genus Aloconota; the affinity with Aloconota is evident from the structure of the female genitalia. In Worcestershire records range from 13 m altitude at Birlingham to 300 m altitude in the Malvern Hill’s Happy Valley. This last, nectaring on flowers of gorse Ulex europaeus L., and the one from Broadway were both in wooded valleys. The Birlingham specimen may be associated with plantations of hybrid poplar, which are now mostly felled and although not colline it is located in a valley bottom. An association with sheltered woodlands is confirmed by Atty (1983) who recorded a Gloucestershire specimen at Toadsmoor near Middle Lypiatt during 1914. Given what is now known, the single Warwickshire specimen found by Professor F. W. Shotton at Shuttington during May 1966 is likely to have dispersed from humid woodland by the pools at nearby Alvecote. Callicerus rigidicornis is not cited by Duff (1993) and neither species is cited by Tomlin (1949) or Hallett (1954) for Herefordshire or by Darby (2009) for Wiltshire. According to Sharp (1908) C. rigidicornis was noted at sea-level on the estuarine banks of the River Mersey. This may reflect a greater tolerance of exposure in the west of the country, a well-known phenomenon in a number of more usually woodland beetles but in the midlands neither C. rigidicornis nor C. obscurus can be regarded as primary floodplain species (Lott, 2009; Whitehead, 1992). In the midlands C. rigidicornis is likely to favour damp or somewhat closed-canopy broadleaved woodland and is up-to-now unknown on arable farmland.
Regional records. All single specimens.
Worcestershire
Broadway, SP03, 91 m O.D., partially wooded valley, in flight, 15 May 1985.
Malvern Hills, Happy Valley, SO74, 300 m O.D., wooded valley, nectaring at flower of gorse Ulex euoropaeus L., 28 April 2007.
Birlingham floodplain, SO94, 13 m O.D., in flight, 22 April 2012 (specimen 5.2 mm in length).
Conservation status of Callicerus spp. The inclusion of beetles in U.K. Nature Conservation 12:2 is warranted on the criteria of their frequency and distribution (Hyman, 1994); Callicerus is excluded from that text. Widespread localised species may nonetheless diagnose sites and situations which reflect clearly on special or ancient landscape features and it is my view that Callicerus, like various other athetine rove beetles, belongs in this category.
Acknowledgements
Some of these findings arose during research projects for ADAS Drayton, Warwickshire and the Kemerton Conservation Trust, Worcestershire. Marc Tronquet kindly summarised knowledge of C. rigidicornis in France.
References
Atty, D.B. 1983. Coleoptera of Gloucestershire. Cheltenham, published privately.
Darby, M. 2009. Wiltshire beetles; history, status, distribution and use in site assessment. Malthouse Books, Salisbury.
Duff, A.G. 1993. Beetles of Somerset. Taunton: Somerset Archaeological and Natural History Society.
Hallett, H.M. 1954. The Coleoptera of Herefordshire: first supplement, pp. 279-282 in: Transactions of Woolhope Naturalist’s Field Club, Hereford.
Hyman, P.S. (revised Parsons, M.S.) 1994. A review of the scarce and threatened Coleoptera of Great Britain 2. UK Nature Conservation 12. Peterborough: Joint Nature Conservation Committee.
Lane, S.A., Wright, R.J. & Forsythe, T.G. 2002. An atlas of Warwickshire beetles. Warwickshire Biological Records Centre.
Lott, D.A. 2009. Rare beetles from the lower Soar valley in Leicestershire and Nottinghamshire. British Journal of Entomology and Natural History22:217-233.
Sharp, W.E. 1908. The Coleoptera of Lancashire & Cheshire, pp. 1-75. Lancashire & Cheshire Entomological Society.
Tomlin, J.R. le B. 1949. Herefordshire Coleoptera 1. Woolhope Naturalist’s Field Club, Hereford.
Whitehead, P.F. 1992. The floodplain Coleoptera of the River Avon, Worcestershire, England, with provisional diagnoses of ancient assemblages. Elytron 6:15-33.
Worcestershire Record | 33 (Nov 2012) page: 33-34 | Worcestershire Biological Records Centre & Worcestershire Recorders
Worcestershire Record | 33 (Nov 2012) page: 32-33 | Worcestershire Biological Records Centre & Worcestershire Recorders
Acicula fusca (Montagu, 1803) (Mollusca: Gastropoda, Architaenioglossa, Aciculidae) extant at Cleeve Hill, Gloucestershire VC33 SO92, with observations on its ecology and palaeogeography
P.F. Whitehead
Introduction
Although the distribution map of Acicula fusca (Montagu, 1803) shown by Kerney (1999) now requires some updating, there remains a clear picture of a declining species over large parts of central and eastern England. In the oceanic west and south-west the populations are rather more stable and examples may be found in relatively open sites such as sea-cliffs. In Worcestershire, recent discoveries or confirmations of A. fusca come from Death’s Dingle (SO66, 21st June 2003) and Seckley Ravine (SO77, 27th June 2004) (the records were published anonymously in Worcestershire Record 15 and 17 respectively) and to the south in Watsonian Worcestershire on the wooded north Cotswold escarpment near Snowshill (Knight, 2007).
Scrutiny of the distribution of A. fusca in Gloucestershire (http://data.nbn.org.uk; Kerney, 1999) provides a catalogue of ancient species-rich Cotswold woodlands such as Buckholt, Cranham, Siccaridge, Frith and Workman’s to which the Worcestershire woodland sites are complementary in quality. A combination of scree, subangular rock clasts, artesian drainage and sometimes tufa is one which A. fusca is known to favour.
Acicula fusca on Cleeve Hill
Cleeve Hill is delineated on its westernmost edge by limestone crags and thermoclastic scree and is noteworthy for its biota including invertebrates. The only previous record of A. fusca on Cleeve Hill that I have traced was of one found by Peter Tattersfield on 26th April 1986 (SO92) and away from woodland the pulmonate A. fusca becomes much scarcer in the midlands where it is sensitive to anthropogenic and climatic impacts and to desiccation.
The live specimen of A. fusca cited here was observed on 12th April 2012 at a depth of 20 cms in thermoclastic scree at 290 m O.D. This is a habitat that requires to be investigated with care and I regarded further searches as counterproductive on the basis that the hold of A. fusca there would be tenuous at best. The nearest woodland to the site is a mature dominantly high-canopy beech wood 100 m to the south; this woodland is also recognised for its invertebrate interest but up to now has apparently not yielded A. fusca.
The habitat of Acicula fusca
Acicula fusca is regarded as an oceanic climate woodland snail (Kerney, 1977a; 1977b; Limondin-Louzouet & Preece, 2004) sometimes of wet spots (Janus, 1965). However, A. fusca has several Palaearctic congeners which characterise scree in the Alps and other mountain ranges (Kerney & Cameron, 1979). It may well be, at least in the midlands, that woodland preference of A. fusca is determined partly by the rarity of crags with actively developing scree such as exist on Cleeve Hill. Further north along the north Cotswold escarpment where scree is well-developed, it is more rarely active and is quite often fixed by vegetation and soils which render it unsuitable for the pulmonate A. fusca. This process of scree-fixing renders it suitable for a superficially similar but unrelated subterranean mollusc Cecilioides acicula (Muller, 1774) which occurs in such habitat on Cleeve Hill within 20 m of A. fusca and down slope of it. Cecilioides acicula is not anthropophobic and is widespread in Worcestershire even occurring in stable undisturbed loamy sediments in old urban gardens. With its requirement for airspaces, the open mantle cavity lung of A. fusca is able to function optimally in interstices which may also partly explain its occurrence on the lightly vegetated rock floors of Cotswold beechwoods.
The range of Acicula fusca
Fig.1. shows the west European range of Acicula fusca (after Boeters, Gittenberger & Subai 1989). Note that this is an indication of distribution not of frequency and that whilst appearing widespread in Britain, some of the map dots may represent one or a few specimens.
The range of A. fusca is determined by climate. It avoids climatic continentality (Pfleger & Chatfield, 1983) and like its Cleeve Hill associates Pomatias elegans (Müller, 1774) and Abida secale (Draparnaud, 1801) has a Palaearctic range (Fig. 1) largely governed by winter temperature (Boeters, Gittenberger & Subai, 1989; Limondin-Louzouet & Preece, 2004). Evans (1975) points out that A. secale is essentially west European but is able to withstand temperatures as low as -10oC in the Alps. It should be recalled however that A. secale, which is also unknown in Scandinavia, is able to utilise scree interstices to avoid marked fluctuations of climate in the same way as A. fusca. Further confirmation of the relict status of A. fusca comes from its fossil record which extends back into the Miocene of central Europe.
The modern geographical range of A. fusca (Fig. 1) is worthy of scrutiny from another standpoint. The populations focus in Britain and northern Spain and one is entitled to ask, if they are disparate, whether each is indicative of a past refugium. Were that to be so, where in north-west Europe did A. fusca survive the last ice age? South of the Euro-Britannic land bridge, in south west Ireland, in both or in neither?
References
Boeters, H.D., Gittenberger, E., & Subai, P. 1989. Die Aciculidae (Mollusca: Gastropoda Prosobranchia). Zoologische Verhandelingen 252. Leiden.
Evans, J. G. 1975. The environment of early man in the British Isles. Unwin Brothers, Surrey.
Janus, H. 1965. The young specialist looks at land and freshwater molluscs. Burke, London.
Kerney, M.P. 1977a. British Quaternary non-marine Mollusca: a brief review. pp. 31-42 In: Shotton, F.W. (ed.) British Quaternary studies: recent advances. Clarendon Press, Oxford.
Kerney, M.P. 1977b. A proposed zonation scheme for Late-glacial and Postglacial deposits using land Mollusca. Journal of Archaeological Science4:387-390.
Kerney, M.P. 1999. Atlas of the land and freshwater molluscs of Britain and Ireland. Harley Books.
Knight, T. 2007. Provisional atlas of land snails in south-east Worcestershire. Worcestershire Record 23:54-71.
Kerney, M.P. & Cameron, R.A.D. 1979. A field guide to the land snails of Britain and north-west Europe. Collins, London.
Limondin-Louzouet, N. & Preece, R.C. 2004 Molluscan successions from the Holocene tufa at Germain-le-Vasson, Normandy (France) and their biogeographical significance. Journal of Quaternary Science 19:49-54.
Pfleger, V. & Chatfield, J. 1983. A guide to the snails of Britain and Europe. Hamlyn.
Image

(C)PFWHITEHEAD
Fig_1 The west European range of Acicula fusca
Worcestershire Record | 33 (Nov 2012) page: 34-35 | Worcestershire Biological Records Centre & Worcestershire Recorders
Coproporus immigrans Schülke, 2006 (Col., Staphylinidae) new to the English Midlands
P.F. Whitehead
Paul F. Whitehead
Moor Leys, Little Comberton, Pershore, Worcestershire WR10 3EH email: paul@thewhiteheads.eu
Introduction
The Tachyporinae is a well-known sub-family of rove beetles often comprising shiny brightly-coloured beetles in shades of yellow orange and black and between one and 11 mm in length. Many are moisture-loving and frequently have a characteristic plan-form narrowing behind from a rounded pronotum and small head.
On 26 June 2012 at the firewood processing site known as Tiddesley Wood Barn (SO94 46 m O.D.) I had the opportunity to examine a pile of wet wood chippings, a habitat well-known for its ability to attract a variety of synanthropic and warm-stenothermic invertebrates (e.g. Whitehead, 1994). It soon became clear that this pile of wood chippings supported a population of a small uniformly brown tachyporine with significantly broad foreparts attenuated behind. A further feature of this beetle was its distinctive highly mobile abdomen which was continually elevated and rotated unlike other tachyporines seen by the writer (Fig. 1.).
Fig. 1. Coproporus sp. ♂, on the basis of its plan-form probably C. immigrans Schülke, 2006. (Image courtesy of Professor Lech Borowiec).
Discussion
The site is located amongst open pastureland and old orchards close to the edge of Tiddesley Wood near Pershore, a well-known Worcestershire Wildlife Trust woodland actively managed for its conservation interest. Tiddesley Wood Barn receives cordwood and trunkwood from many of Worcestershire Wildlife Trust’s woodlands. It therefore acts as a biological assembly-point for invertebrates from VC37 some of which eventually die out; at the same time it provides instant habitat for colonists from nearby. In essence the Coleoptera fauna bears some similarity to that of a timber yards. In this case, the on-site conversion of felled trees to firewood, often by hand, creates a mosaic of wood-based habitats some of which have no analogues in nature. Having gleaned a considerable amount of biological data from this site I eventually hope to say more about it, suffice to say that the ‘deadwood’ invertebrate fauna of the site is quite different in composition to that of the nearby regional wet woodlands of the ancient Forest of Horewell.
A literature search implied that the tachyporines belonged to the genus Coproporus which is most usually associated with moribund trees; it may also be found associated with ants of the genus Camponotus and synanthropically amongst soft fallen fruit. Using a high-powered microscope I determined voucher specimens as Coproporus immigrans Schülke, 2006. Schülke (2006) cited no British records of C. immigrans which came too late for inclusion in his review.
Coproporus species are essentially thermophilous in warm-temperate regions. During the late 1990s observers in a number of north-west European countries, including Norway, became aware of Coproporus as a novelty. It was first recorded in Britain during 2004 (Denton, 2005) where the species is now thought to be widely established in the Home Counties (Sage, 2012) and elsewhere so that its occurrence in Worcestershire was only a matter of time. C. immigrans can be separated from the central and south European Coproporus colchicus Kraatz, 1858 by its more vaulted body and by fine details of the last abdominal tergite which, unless one is very familiar with the genus, require a microscope to resolve. Where I have found C. colchicus in southern Europe the association in that case has been with coniferous trees.
The epithet ‘immigrans’ is quite often employed in the binomials of adventive or tramp species which have invaded from elsewhere. On the basis of its occurrence in south-eastern Australia Schülke regards C. immigrans as of potentially Australian origin, although by the speed of its spread in north-western Europe further work may be required to confirm its range and origin. Coproporus is especially speciose in Asia (Smetana, 2004). C. immigrans is likely to turn up at other sites in Worcestershire, but unlike many tramp species has not yet been found in compost.
Acknowledgements
I thank Worcestershire Wildlife Trust and Harry Green M.B.E. for permission to visit the site. I am grateful to Dr Michael Schülke for keeping me up-to-date with his output and to Professor Lech Borowiec for the use of one of his exceptionally fine images.
References
Denton, J., 2005. The beetles of Surrey – a checklist. Surrey Wildlife Trust.
Sage, B., 2012. The first records of Coproporus immigrans Schülke and Pseudomedon obscurellus (Erichson) (Staphylinidae) for Norfolk. The Coleopterist 21:79.
Schülke, M., 2006. Drei neue Adventivarten der europäischen Staphyliniden-Fauna, mit Bemerkungen zu Coproporus colchicus Kraatz (Coleoptera, Staphylinidae, Tachyporinae). Entomologische Blätter102:173–201.
Smetana, A, 2004. Tachyporinae, pp. 330-352 In Lobl, I. & Smetana, A. (eds), Catalogue of Palaearctic Coleoptera 2. Stenstrup: Apollo Books, 942 pp.
Whitehead, P.F., 1994. Rural breeding populations of Hypoponera punctatissima (Roger) (Hym., Formicidae) in Worcestershire. Entomologist’s Monthly Magazine 130:194.
Image

Fig. 1 Coproporus sp. male, on the basis of its plan-form probably C. immigrans Schülke, 2006. (Image courtesty of Professor Lech Borowiec).
Worcestershire Record | 33 (Nov 2012) page: 35 | Worcestershire Biological Records Centre & Worcestershire Recorders
Lipoptena cervi (L., 1758) (Diptera, Hippoboscidae) in a Worcestershire gymnasium
P.F. Whitehead
Paul F. Whitehead, Moor Leys, Little Comberton, Pershore, Worcestershire WR10 3EH email: paul@thewhiteheads.eu
Introduction
The Hippoboscidae is a family of highly distinctive flies which are blood-sucking ectoparasites of mammals and birds. The genera Crataerina and Ornithomya are well-known on House Martins and passerine birds respectively and occur widely in this area. Many species are flightless and may cause consternation if they appear on people, when, by virtue of their exceptionally well-developed tarsal claws, they demonstrate an ability to move in any direction with equal facility. They are very tenacious to the host animal hence the occurrence of Hippobosca in tins of corned beef.
Discussion
On 30 June 2012 I observed an adult hippoboscid Lipoptena cervi (L.) on the glass door of a gymnasium on the first floor of, and well inside, a health complex in south Worcestershire (SP04). This species is capable of flight although according to Hutson (1984) this is mostly between August and December. Lipoptena cervi is usually associated with deer. No deer occur in the nearby area and none have been confirmed in the gymnasium; equally it is inconceivable that this fly could navigate through some 100 metres of a closed building to gain access to an area which has no opening windows.
I would contend that this fly was introduced to the gymnasium by a human on which according to Hutson (op. cit.) there are ‘stray’ records. Hopefully a precedent for breeding on them has not been set.
Reference
Hutson, A.M., 1984. Keds, flat-flies and bat-flies. Handbooks for the identification of British insects 10(7). Royal Entomological Society of London.
Worcestershire Record | 33 (Nov 2012) page: 35 | Worcestershire Biological Records Centre & Worcestershire Recorders
Worcestershire Record | 33 (Nov 2012) page: 35 | Worcestershire Biological Records Centre & Worcestershire Recorders
Speckled Bush-cricket Leptophyes punctatissima (Bosc, 1792) (Orth., Tettigoniidae) eating the fruit valves of Hoary Stock in Worcestershire
P.F. Whitehead
Paul F. Whitehead, Moor Leys, Little Comberton, Pershore, Worcestershire WR10 3EH email: paul@thewhiteheads.eu
On 6 October 2012 an enterprising female Speckled Bush-cricket Leptophyes punctatissima (Bosc) was observed consuming, with some alacrity, the surface tissues of a fruit-valve of a Hoary Stock (Matthiola incana (L.) W. T. Aiton) growing in a sunny spot in my garden in Little Comberton (SO94) (Fig. 1). As this scarce plant has only been grown here for two years it must indicate the wide-ranging tastes of this bush-cricket.
Image

Fig. 1 Female Speckled Bush-cricket masticating and consuming the fruit-valve of a Hoary Stock, Little Comberton, 6 October 2012
Worcestershire Record | 33 (Nov 2012) page: 36-38 | Worcestershire Biological Records Centre & Worcestershire Recorders
Greenhouse safari! Invertebrates visiting greenhouses
Rosemary Winnall
We amateur entomologists are well used to sidling up to the windowsills in the houses of friends and acquaintances to take a furtive look for stranded insects. We often make wistful glances up to those covered light fitments through which small dark shadows speak of riches beyond our reach.
This summer, however, I have taken a particular interest in greenhouses and take delight in visiting the conservatories and greenhouses of friends and relations in order to get a sneaky look at the residents or visiting fauna! Indeed, our own greenhouse has provided a wealth of interest this summer by both day and by night. Perhaps creatures were coming in to shelter from the rain!
Residents included a large toad (Fig. 1.) that spent summer days beneath the flowerpots, beautifully marked Yellow Slugs Limacus flavus (Fig. 2.) and a large Leopard Slug Limax maximus (Fig. 3.) that used to venture out to feed on algae at night. I went down at 10.00pm one night and found a line of Lasius niger ants moving their pupae from under a slab in the garden to a new nest inside the greenhouse! Perhaps they were moving into warmer quarters for the winter because they couldn’t stand their waterlogged nest any longer! As I watched, a large snail Cornu aspersum slowly made its way across the line of ants. I am not sure what the ants did, but the snail soon started to produce a lot of protective frothy bubbles (Fig. 4.) as it made its way into the greenhouse to feast on our plants!
There were all kinds of small creatures up on the glass windows on sunny days. Andrena fulva (Fig. 5.)was a conspicuous visitor early in the year, Odynerus spinipes in June, and the smaller Campanula bee Chelostoma campanularum (Fig. 6.) in late summer. I had good views of some of the smaller flies against the glass, many of which are easily overlooked amongst foliage in the garden. I spotted the small hoverfly Neoascia podagrica, soldier flies Beris morrisii (Fig. 7.) with its distinctive spined scutellum, and the splendidly coloured female Sargus bipunctatus. Picture-winged Flies were easily spotted with their distinctive wing patterning: Palloptera muliebris (Fig. 8.), Urophora jaceana (Fig. 9.) Tephritis formosa amongst others.
Moth flies (Fig. 10.) were of particular interest but only the males can be identified, and most of the ones I spotted were females! There were tiny Chalcid wasps and the occasional Ruby-tailed Wasp, (Fig. 11.) and I could get close up views of the horse fly Haematopota pluvialis (Fig. 12.) to see the beautiful colours its eyes.
These flying insects attracted predators and there were several different spiders in residence and the distinctive harvestman Dicranopalpus ramosuson the wall. When flying ants ventured in many casualties were found in spiders’ webs and this enabled me to check out the winged queens and males of different ant species, for example Formica fusca (Fig. 13.).
Our greenhouse provided a warm dry environment for many creatures this year, and it was a great place to entomologise out of the rain! I can recommend it!
Images
 Fig. 01. Common Toad. Rosemary Winnall
Fig. 01. Common Toad. Rosemary Winnall
 Fig. 02. Yellow Slug (Limacus flavus). Rosemary Winnall
Fig. 02. Yellow Slug (Limacus flavus). Rosemary Winnall
 Fig. 03. Leopard Slug (Limax maximus). Rosemary Winnall
Fig. 03. Leopard Slug (Limax maximus). Rosemary Winnall
 Fig. 04. Garden Snail (Cornu aspersum) with Lasius niger ants carrying pupae. Rosemary Winnall
Fig. 04. Garden Snail (Cornu aspersum) with Lasius niger ants carrying pupae. Rosemary Winnall
 Fig. 05. Female Andrena fulva. Rosemary Winnall
Fig. 05. Female Andrena fulva. Rosemary Winnall
 Fig, 06. Campanula Bee Chelostoma campanularum. Rosemary Winnall
Fig, 06. Campanula Bee Chelostoma campanularum. Rosemary Winnall
 Fig. 07. Scutellum of soldier fly Beris morrisii.Rosemary Winnall
Fig. 07. Scutellum of soldier fly Beris morrisii.Rosemary Winnall
 Fig. 08 Female Palloptera muliebris. Rosemary Winnall
Fig. 08 Female Palloptera muliebris. Rosemary Winnall
 Fig. 09. Male Urophora jaceana. Rosemary Winnall
Fig. 09. Male Urophora jaceana. Rosemary Winnall
 Fig. 10. Female Moth Fly. Rosemary Winnall
Fig. 10. Female Moth Fly. Rosemary Winnall
 Fig. 11. Ruby-tailed Wasp Trichrysis cyanea. Rosemary Winnall
Fig. 11. Ruby-tailed Wasp Trichrysis cyanea. Rosemary Winnall
 Fig. 12. Horsefly Haematopota pluvialis. Rosemary Winnall
Fig. 12. Horsefly Haematopota pluvialis. Rosemary Winnall
 Fig.13. Queen ant Formica fusca. Rosemary Winnall
Fig.13. Queen ant Formica fusca. Rosemary Winnall
Worcestershire Record | 33 (Nov 2012) page: 48-50 | Worcestershire Biological Records Centre & Worcestershire Recorders
Water Shrew bones in an Owl Box
Rosemary Winnall
It has long been accepted that the identification of bones from owl pellets is a good way of obtaining records of small mammals. This is something I have been doing for some time and I am always on the look out for local roosting and breeding holes used by Tawny Owls or Barn Owls, as these can provide a regular source of pellets.
Barn Owls are known to have roosted and bred regularly in artificial nest boxes at Haye Farm near Bewdley (SO778741) for about ten years from 1998, although during the last four years they have only been seen intermittently. During the spring of 2012 the farmer, Stuart Norgrov,e cleaned out one of his eight owl boxes and asked me if I wanted the contents for checking. I was enthusiastic, but when they arrived in two large fertiliser bags via Brenda Rea, I realised that I had grossly underestimated the size of the task, and there was an enormous amount of material to sort! The owl box from which this came (Owl Box 2) is a large black drum high in a veteran hedgerow oak tree about 400m from the farm buildings (see Figs. 1 and 2). I had already received a box containing the debris from one of the other owl boxes – a smaller wooden triangular owl box that had been up inside one of the barns (Owl Box 1).
We could not, of course, presume that all the material came from Barn Owl pellets. We knew that Kestrels, Tawny Owls and Little Owls had all held territories in the vicinity of the farm, and these, as well as other creatures, might have used the box at some time. But nevertheless, any small mammal records that we could obtain would make valuable records for this locality.
Firstly I recruited the help of Mick Blythe and his battery-powered pooter. (We didn’t like the thought of sucking up creatures from such material with standard pooters as you can imagine!). We opened the bags and collected anything that moved – moths, flies and beetles went into the tubes and were saved for later identification. Caterpillars were extracted and all lepidoptera given to Tony Simpson. Beetles went to John Meiklejohn and Mick took the flies for identification.
Then we donned gloves and sorted through the material bit by bit, taking out skulls and lower jaws. These were then soaked overnight in hot soapy water with disinfectant, after which identification could begin. First I sorted the skulls and lower jaws into those of voles, mice, shrews and others, and it was a long business with 836 skulls examined and many more lower jaws checked. Then I went back and identified the species in each group, and it was then that I got a surprise (see table below for the results)!
Whilst checking the shrew bones, I soon realised that I had the lower jaws and skulls of some Water Shrews (see photos 3, 4, and 5)! I had not found these in owl pellets before! Dormice yes, but not Water Shrews! This is an excellent record of a small mammal that is so rarely seen and recorded in Worcestershire. The difficulty is that we don’t know how recently the Water Shrews were caught – some time within the last 14 years! Are they still present and if so, where do they live? (See Figs. 3, 4, & 5.)
The Mammal Society conducted a Water Shrew Survey during 2004/2005 and I was interested to find out how they had gained evidence that this species was present. Shrews need to consume very regular amounts of food and soon die if they are left in traps for too long. Indeed, without a license to trap shrews Longworth traps could not be used. The Mammal Society gained most information by setting up feeding tunnels, collecting the droppings, and checking their contents. This seemed the way forward.
So I have made some feeding tubes by cutting 4cms. wide white tubing into 20cms. lengths and covering one end of each with gauze held with a rubber band. I have received permission to place these tubes, baited with dried mealworms, around the watercourses on the farm to see if I can obtain any evidence that these endearing little mammals are present. It might not work, but it is worth a try. Watch this space!
Many thanks are due to Stuart Norgrove for his enthusiasm for the wildlife around his farm, and for his interest in this project.
Species |
Owl Box 1 skulls |
Box 1% of species |
Owl Box 2 skulls |
Box 2% of species |
Total skulls |
Total % |
| Field Vole | 172 | 70% | 406 | 68.7% | 578 | 69% |
| Common Shrew | 31 | 12.6% | 87 | 14.7% | 118 | 14% |
| Wood/Yellow-necked Mouse | 28 | 11.4% | 43 | 7.3% | 71 | 8.5% |
| Bank Vole | 8 | 3.2% | 25 | 4.2% | 33 | 3.9% |
| Pygmy Shrew | 3 | 1.2% | 22 | 3.7% | 25 | 2.9% |
| Water Shrew | 2 | 0.8% | 4 | 0.7% | 6 | 0.7% |
| Rat | 1 | 0.4% | 4 | 0.7% | 5 | 0.6% |
| Bird | 2 | 0.8% | 1 | 0.17% | 3 | 0.35% |
| TOTAL | 245 | 591 | 836 |
Table of results from identifying skulls in material from two owl boxes from Haye Farm near Bewdley.
Images
 Fig. 1. Hayes Farm owl box 2
Fig. 1. Hayes Farm owl box 2
 Fig. 2. View down to Haye Farm from Owl Box 2 tree
Fig. 2. View down to Haye Farm from Owl Box 2 tree
 Fig. 3. Close up of Water Shrew lower jaw to show lack of cusps on front tooth
Fig. 3. Close up of Water Shrew lower jaw to show lack of cusps on front tooth
 Fig. 4. Difference in skull size left Water Shrew, middle Common Shrew, right Pygmy Shrew
Fig. 4. Difference in skull size left Water Shrew, middle Common Shrew, right Pygmy Shrew
 Fig. 5. Lower jaw bones top Pygmy Shrew, middle Common Shrew, bottom Water Shrew
Fig. 5. Lower jaw bones top Pygmy Shrew, middle Common Shrew, bottom Water Shrew
