Issue 32 April 2012
Worcestershire Record | 32 (April 2012) page: 17 | Worcestershire Biological Records Centre & Worcestershire Recorders
The new Dragonfly Atlas of England, Wales & Scotland
Mike Averill
This is the last year left to survey for the National Dragonfly Atlas and there is still time to visit those remote corners of Worcestershire. 2011 was the fourth of the five year project to remap the dragonflies in the UK mainland. In Worcestershire 86 recorders gathered 1312 new records which added to the previous three years to give 4588 in total (Table 1).
Table 1. Totals of records of dragonfly species in Worcestershire
| 2008 | 1107 |
| 2009 | 1172 |
| 2010 | 997 |
| 2011 | 1312 |
| Total | 4588 |
The main reason to re-survey is that the last major Atlas was published in 1990 and there have been considerable changes since then both in numbers of new species and their distribution. In that time Worcestershire has seen five new species including Yellow-winged Darter Sympetrum flaveolum; Red-veined Darter Sympetrum fonscolombii; Scarce Chaser Libellula fulva; Lesser Emperor Anax parthenope and Small Red-eyed damselfly Erythromma viridulum.
The results from the last four years in Worcestershire are summarised in the map (Fig. 1.) which shows how many species have been recorded in each 2×2 km square. The total is coded into nine groups as shown in the key. The map shows that many squares still only have two species or less recorded which hopefully can be rectified in 2012. There are a few sites coded to nine which means that there are between 17 and 21 species present as at the Grimley gravel pits and Upton Warren, but if possible recording in the blank areas is needed as the popular sites are well covered.
In 2011 new records helped to fill in some of the outlying areas around Tenbury Wells, but there are still large gaps in quite a few locations such as in the SW, SE and NE of the county.
Please send your records to mike.averill@blueyonder.co.uk and these should contain at least the basic information about the location of the dragonfly with grid reference, date, and species, but if you can give any estimate of numbers or breeding activity that would add a lot to the record. If you feel keen I can send a spreadsheet with a pick list of species already entered.
For details of the individual species records and where to record, you will be able to visit a new website dedicated to Worcestershire Dragonflies before the start of the new season.
If you are outside Worcestershire on holiday, don’t forget it is a national atlas. All records are welcome and I am happy to receive them all or you can enter them online via the British Dragonfly Society website: http://british-dragonflies.org.uk/content/submit-records.
Records from Herefordshire are also badly needed to bring the dataset up to date.
Image
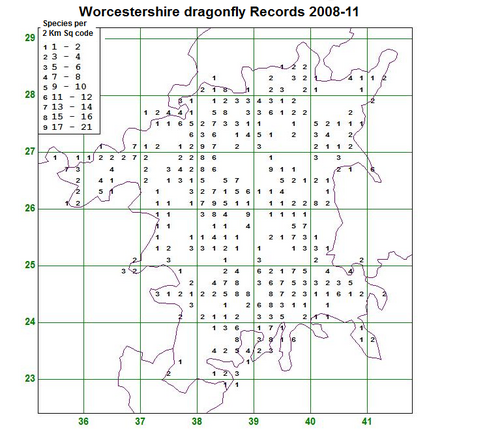
Fig. 1. Dragonflies recorded in Worcestershire 2008-11
Worcestershire Record | 32 (April 2012) page: 14 | Worcestershire Biological Records Centre & Worcestershire Recorders
Gonia picea (Robineau-Desvoidy, 1830) Tachinidae, Diptera and Melanimon tibialis (Fabricius, 1781) Tenebrioninae, Coleoptera, Recorded on the Devil’s Spittleful Nature Reserve 2012
John Bingham
The Devil’s Spittleful is undergoing much-needed restoration back to heathland and the work is producing many newly cut stumps (and many stumps ground down). On a visit on 22nd March 2012 a line of freshly cut birch stumps looked interesting as sap was oozing out and on several a bacterial/fungal flux had formed. Several insects were feeding on the stumps when I spotted and photographed an unusual large Tachnid fly: several were seen in this area. It was later identified as Gonia picea (Red Data Book Rare), a southern species with apparently few records for the Midlands and none for Worcestershire (NBN Gateway). Recent reports indicate that it has increased in numbers and range in 2012 due to the exceptional warm spring weather (as now in March 2012).
A distinctive fly with a unusual head, it is an early vernal species seen March to May after overwintering as an unenclosed adult. It is a parasitoid of Cerapteryx graminis (Antler Moth), Mythimna comma (Shoulder-striped Wainscot) (Noctuidae) and Polygonia c-album (Comma butterfly) (Nymphalidae). Its habitat is said to be long grass on chalk, meadows and wood margins. (Information taken from the Tachnid Recording Scheme Web pages). Perhaps we can now add acid grassland and scrub heathland to the list?
On the same day on a drier part of the adjacent the Rifle Range on bare sand near heather I collected a small beetle about 3-4mm long that looked slightly different from the norm. It was identified as Melanimon tibialis (tibiale) a Darkling Beetle. (Joy1932). This is a mainly coastal species with a few records inland and only one old and undated record from Worcestershire from Hartlebury Common. (NBN Gateway). Little information was found searching the web but it appears to burrow in soft sand and have a preference for sand dune locations in Britain.
References
Joy, N. H. 1932. A Practical Handbook of British Beetles. Vols I & II. Witherby, London.
Tachnid Recording Scheme web site http://tachinidae.org.uk/site/whatarethey.php
Images

Fig. 1. Gonia picea Tachinidae, Diptera, at Devil’s Spittleful. John Bingham.

Fig. 2. Melanimon tibialis Coleoptera, Tenebrioninae, Devil’s Spittleful. John Bingham.
Worcestershire Record | 32 (April 2012) page: 6 | Worcestershire Biological Records Centre & Worcestershire Recorders
Ichneumon sarcitorius Linnaeus 1758. Family Ichneumonidae. Found at Shavers End Quarry, Abberley, Worcestershire
John Bingham
On a visit to Shavers End quarry (Abberley Hills) on 19th March 2012 I was trying to photograph small solitary bees on a dry bank (unsuccessfully) when a large colourful Ichneumonid landed nearby. I grabbed a quick photograph before it flew off having no idea what species it might be.
Normally ichneumons are difficult to identify and beyond my skills and my reference works but this was quite large species (14mm) and very distinctive with the orange and black banding so I searched around for an identification on the web. A German site HymIS, Hymenoptera Information System, proved very useful and after some consideration I tentatively named it as Ichneumon sarcitorius.
I emailed the image and my tentative naming to Dr Gavin Broad of the Natural History Museum who kindly replied to confirm my identification. He added the following: ‘Ichneumon sarcitorius. This is a common and fortunately distinctive species. There is no other species that has that combination of red, black, red black then a white stripe towards the tip of the metasoma. The females overwinter as adults so can be out early in the year when it is a very polyphagous parasitoid of noctuid pupae’.
In Worcestershire it appears to be under recorded with three records, one from the Teme area near Rochford and two from near Pershore.
My thanks to Gavin Broad for his help.
Reference
MymIS web pages; http://www.hymis.de/fotos/pictures/list.php?tree=Ichneumonoidea.19&sub=yes&fam_level=1&tree_id=19&tree_status=plus&tree_seq=7&nav1=pictures
Image

Fig.1. Ichneumon sarcitorius at Shavers End. John Bingham
Worcestershire Record | 32 (April 2012) page: 22 | Worcestershire Biological Records Centre & Worcestershire Recorders
Isaria farinosa (Holmsk.) Fr.1832,. Trichomaceae. Found in Wyre Forest, Worcestershire
John Bingham
On 28th January 2012 I found a small white fungus growing in a clump of Polytrichum formosum moss in Hitterhill Wood, Wyre Forest, Worcestershire. The powdery spike was only 20mm long and grew out of the moss, so I carefully dug downwards to find the attachment. When extracted it was found to be attached to a small Lepidopteran larvae, possibly a micro moth of some type (Fig. 1.). Identification of this fungus would have been difficult but thankfully an illustration can be found in the Collins Photographic Guide Book on Fungi (Sterry & Hughes 2009) that clearly indicated it was Isaria (Paecilomyces) farinos.
Isaria farinosa is apparently quite a common fungus as there are some 565 records on the British Mycological Society database (BMS access online). However many records are multiple entries and a search for Worcestershire in the entries only yielded one record, although the ‘dot map’ indicates about six old 10km square records including the Wyre Forest SO77 10×10 km square. This last record I assume is attributable to Carlton Rea as he recorded Isaria farinosa in his fungus list for Wyre Forest (Rea 1923). The only other record I can find for the county was at Ockeridge Wood in 1912. Warwickshire is somewhat better recorded thanks to the 1980 fungus survey by Malcolm Clark et al (Clark 1980), but records were still limited and very scattered. For a supposedly common fungus this is certainly not the case for Worcestershire, although it must be very under recorded! Check your local moss for white powdery fungal spikes, my guess is you will find more.
There are several other common fungi that parasitise insects (Entomopathogenic fungi). Cordyceps militaris with the bright orange club shaped fruit-body being the most commonly recorded species, but Entomophthora muscae sl. (several species) is more frequent and obvious to find as it typically kills Muscid and Syrphid flies, leaving them dead perched on the top of grass stems with grey conidia spores bursting out of the abdomen.
References
Clark, M C. (ed). 1980 A fungus flora of Warkwickshire. British Mycological Society, London.
Sterry, P. & Hughes, B. 2009 Collins Complete Guide to British Mushrooms & Toadstools. Collins, London
Rea, C. 1923. Fungi of the Wyre Forest. Transactions of the Worcestershire Naturalists Club. 1924 pages 16-40.
British Mycological Society. Checklist of the Fungi of the British Isles: http://www.fieldmycology.net/GBCHKLST/gbchklst.asp
Image

Fig. 1. Isaria farinosa growing from a lepidopteran larva. John Bingham
Worcestershire Record | 32 (April 2012) page: 13-14 | Worcestershire Biological Records Centre & Worcestershire Recorders
Snow Flea Boreus hyemalis (L., 1767 Mecoptera: Boreidae) in Wyre Forest
John Bingham
Many years ago during the late 1970’s or early 1980’s I recall whilst sitting on a moss covered bank in Wyre Forest partaking lunch spotting a small insect (5mm or so) crawling over a patch of thawing snow. At the time my interests in the smaller insects of the forest was limited but I did check the identity of this creature as it seemed rather unusual. Looking in the ‘Collins Insects book’ by Chinery the insect was described and a line drawing confirmed that it was a snow flea, Boreus hyemalis, (Fig. 1.).
I had forgotten about this old observation until recently when on 2ndDecember 2009 Jane Scott re-discovered the insect in Wyre in 2011. Following this discovery the Wyre Forest Study Group (WFSG) took up the hunt to locate more (Winnall 2009). My original site was at Longdon Orchard on the Shropshire side of the forest and Jane’s record was at Withybed Woods also Shropshire, so we had no record for Worcestershire from the Wyre Forest.
In Worcestershire Record No. 28. April 2010 Paul Whitehead reported his own observation of snow flea at Swinyard Hill (part of the Malvern Hills) in 1988. Paul suggests that a more apt common name for the insect is ‘Snow Scorpionfly’, which I agree is really a much a better name. Paul remarked that ‘B. hyemalis is a truly localised species in Worcestershire but one that is likely to occur more widely on the Malvern Hills’. I think the same is also true for the acid soils of the Wyre Forest landscape.
So inspired by these recent records the search was on. On a Wyre Forest Study Group meeting on 14th January 2012 we found more B. hyemalis (1 male and 1 female) at Jane’s Withybed Wood site. A few days later on 21stJanuary Denise Bingham found another single female at a new site, but still in Shropshire. On the 23rd January 2012 Denise and I visited Shelf Held Coppice on the Worcestershire side of the Forest, an area of mossy National Vegetation Classification W16 woodland that I felt sure would have B. hyemalis. My hunch paid off and we found several specimens within the mossy woodland floor (four females in total at two sites). The next foray into Wyre was on 28th January when Brett Westwood accompanied us. We looked in Hitterhill Wood and the forest near to Beaucastle, both areas in Worcestershire. Brett found two males at Hitterhill and Denise found one female at Beaucastle(Fig. 2.).
The typical habitat in Wyre Forest appears to be oak woodland, or open woodland glades with a sparse ground flora and areas of bare soil with scattered or extensive clumps of moss such as Polytrichum formosumand/or Dicranum scoparium. This acid woodland type occurs widely in Wyre Forest and also found at sites such as Chaddesley Woods or Hunthouse Wood (Fig. 3.).
Most books suggest both adults and larvae feed on moss but small soil invertebrates seem a more likely food for the adults. Finding B. hyemalis is really a matter of luck, they openly craw over the moss and have a ‘metallic sheen’ that can catch the sunlight making them easier to find. We found that if after about five minutes looking on a suitable clump of moss it reveals no insects them its best to move on to nearby clump. So as our esteemed editor said in Worcestershire Record No.28 ‘A challenge for next winter is to obtain (more) records in Worcestershire’.
References
Plant, C.W. 1994. Provisional atlas of the lacewings and allied insects (Neuroptera, Megaloptera, Raphidioptera and Mecoptera) of Britain and Ireland/ Edited for the Biological Records Centre by P.T. Harding, B.C. Eversham and H.R. Arnold. Huntingdon: Biological Records Centre.
Whitehead, P F. 2010. An overlooked Worcestershire insect, the Snow Flea Boreus hyemalis. Worcestershire Record 28:15.
Winnall, R. 2009. Snow Flea, Boreus hyemalis (L.,1767) (Mecoptera: Boreidae). Wyre Forest Study Group Review 2009. 10:42.
Buglife website. February 2012. http://www.buglife.org.uk/discoverbugs/bugofthemonth/snowflea.
Images

Fig. 1. Snow Flea Boreus hyemalis, Longdon, Wyre Forest. John Bingham.

Fig. 2. Snow Flea Boreus hyemalis at Beaucastle, Wyre Forest. John Bingham.

Fig. 3. Snow Flea Boreus hyemalis typical habitat in Wyre Forest John Bingham
Worcestershire Record | 32 (April 2012) page: 40-47 | Worcestershire Biological Records Centre & Worcestershire Recorders
Investigating the habitat parameters of the Noble Chafer Beetle Gnorimus nobilis in the Wyre Forest area
Anna Bunney
Dissertation Summary
This dissertation was completed as part of the Geography and Environmental Management course at the University of the West of England. This is an abstract from the whole dissertation. Please contact Anna Bunney if you wish to read the full version.
The aim of this research was to investigate the orchard habitat preferences of the Noble Chafer Gnorimus nobilis beetle focussing on a comparison of orchard habitats where G. nobilis is present and orchards where it is absent. This was to be achieved by comparing traditional orchard habitats and their surrounding habitats, and determining which of them do and which do not support G. nobilis. The location of this study is the Wyre Forest area (Grid Reference: SO749765), which spans the Worcestershire and Shropshire border. The field research aimed to establish if there was a significant relationship between the presence of G. nobilis and individual habitat parameters. This dissertation specifically focuses on identifying differences in the tree species, tree girth, orchard microclimate, surrounding orchard habitat, and orchard management in relation to the presence or absence of G. nobilis in traditional orchards in the Wyre Forest area. Identifying these habitat parameters could help to sustain populations of G. nobilis in the future.
The research question was; “Are there any significant differences in orchard habitat and surrounding orchard habitat where the Gnorimus nobilis beetle is found to be present and where it is found to be absent?”
G. nobilis is one of the rarest saproxylic invertebrate beetles in Britain, labelled as “Vulnerable” by the UK BAP (United Kingdom Biodiversity Action Plan) (Smith 2005), with its remaining number unknown (Eccleson 2007). Due to its rarity, a BAP was implemented in 1999 (UK BAP, 2010).
It is recognised that the most influential factor for G. nobilis presence is dead wood, as it is a saproxylic invertebrate (Alexander 2008; European Red List 2010). Saproxylic invertebrates are dependent on dead wood (Speight 1989). G. nobilis larvae feed on accumulations of wood mould in tree cavities (Smith, 2005). Fruit trees in traditional orchards are relatively short lived, producing decaying wood more quickly than most native hardwood trees, making them important refuges for saproxylic invertebrates (Worcestershire Biodiversity Partnership, 2008).
Furthermore, it is also recognised that G. nobilis preferentially inhabits traditional orchards in the Severn Basin area of Worcestershire, Herefordshire and Gloucestershire (PTES 2010). A traditional orchard is defined as having at least five full sized fruit trees, planted on permanent grasslands, which are widely spaced and allowed to reach a veteran state (Berkshire Biodiversity Action Plan, 2010). However, since the 1950s, traditional orchards have been removed for urbanisation, agriculture, and other changes in land use (National Trust, 2010). It is thought that 85% of Worcestershire’s traditional orchards have been lost in the last 100 years (Worcestershire Biodiversity Partnership, 2008). Therefore the species associated with these habitats are being lost, posing a great threat to saproxylic species, including G. nobilis. Therefore, conservation of these habitats is crucial, otherwise species like G. nobilis will become extinct (UK Biodiversity Action Plan (BAP), 2010).
Sixteen orchards were sampled in total. Once the owner’s permission was granted, site visits were undertaken to determine the suitability of the orchards for this study. This was based on Alexander’s (2002) visual grading scheme for assessment of traditional orchards for G. nobilis. Only orchards that had the grade of one or two were taken on to research in the final study, as they were composed of the required tree species in the right condition to contain G. nobilis.
On this initial site visit, it was noticed that there was a significant difference in orchard size between study sites. Some orchards contained less than ten trees; however, most orchards were much larger, with over fifty trees. Sampling all the trees in all orchards could have taken a considerable amount of time, and it would not have been representative to compare these much larger orchards with the smaller orchards. Therefore, it was decided that for orchards with less than ten trees, all trees would be sampled. In orchards with more than ten trees, a random selection of ten trees was taken (because small orchards mostly had ten or fewer trees in them). It was considered to be more important to study fewer trees in detail, looking into all rot holes, split bark and crevices, than a large number of trees, when there would not be enough time to study the tree in detail. An accurate result of G. nobilis positive and negative trees, and therefore positive or negative orchards, would be obtained.
However, in some of these orchards, saplings were apparent, which had been planted by the land owner. These saplings were too young to contain any dead wood, so would not be a suitable habitat for G. nobilis (PTES, 2010). These younger trees were eliminated from the study, so that a random sample of trees old enough to contain dead wood, could be taken. Trees which had rot holes that were not accessible either because they were too deep or too high up to sample, even by ladder, were also eliminated from this study, as they could not be studied in enough detail.
Individual trees were given numbers, which were marked on the map. Site maps were created so that when the sampling was carried out, a random selection of trees in the orchards could be chosen. Random samples were carried out using Microsoft Excel. The individual tree numbers were entered in a row on a spreadsheet, and each tree was assigned a random number by the Microsoft Excel random number tool. This row was then sorted from smallest random number to largest random number, and the first ten trees with the lowest assigned random number were the trees that were selected for sampling.
As the PTES (2010) have identified, the best way of determining the presence of G. nobilis is by looking for its frass. This is an easy way of assessing presence, and causes no damage to the host tree, or any species inhabiting the tree. The pellets provide good evidence that the larvae have been living in the tree, and can be substantiated by seeing the larvae or parts of the dead beetle (PTES 2010). Larvae live deep within the tree hollows and are not always detectable, even when present. Searching for them could damage or destroy the host tree (Alexander, 2005), whereas frass can be studied without damaging the host tree (Smith 2005).
In this study, sampling was carried out by carefully scooping into any rot holes, crevasses, or behind any bark in the sample tree with an extended spoon to excavate a small amount of wood mould. In some cases, wood mould was easily accessible and G. nobilis frass was identifiable visually. However in most cases, the extended teaspoon was used to scoop into rot holes and crevasses in the tree to obtain a wood mould sample because the frass can be found quite deep within the tree. The wood mould sample was then spread out on a sheet of white paper to examine for frass. Frass is approximately 3mm long, and somewhat resembles mouse droppings (PTES, 2010). This process was repeated in every part of the sample tree where wood mould could have potentially been found.
When sampling for frass in the trees, it was easy to determine when there was a definite positive result if it resembled the frass seen in G. nobilispositive trees at Tiddesley Orchard (SO927462), Pershore, a G. nobilispositive orchard owned by the Worcestershire Wildlife Trust. The G. nobilisfrass stood out well as it was much larger and the colour was either much darker or lighter (depending on age) than the wood mould or any other invertebrate frass, such as woodlouse frass. It was also easy to define a G. nobilis negative tree, as only very fine wood mould was found. However, in some cases, it was hard to be certain. When an unsure sample was taken from the tree, it was put in a sampling bag and sent to G. nobilis expert Harry Green for checking.
A second, but far less reliable way to determine G. nobilis presence was to look for adult beetles. However, as they are only in flight for four to six weeks in early summer, it is a rare occurrence to see them (Smith 2003). This research was undertaken in November so it was not a suitable or reliable method to assess G. nobilis presence.
All trees selected for sampling in the orchards were looked at thoroughly, in all hollows, rot holes and crevasses, to search for frass. Once all sample trees were examined, the orchards were classified as an orchard containing, or not containing G. nobilis. For example if one tree in an orchard of thirty trees contained G. nobilis frass, it would be classified as a G. nobilis positive orchard.
To investigate the habitat preferences of G. nobilis the following set of data was collected in all orchards:
Species of tree and tree girth at every randomly selected orchard tree.
Immediate adjacent habitat – agriculture (pesticides being applied or not), housing, orchards, woodland, industry, roads.
Management techniques applied within the orchard.
Orchard microclimate – Orchard aspect, tree density, orchard size.
Raw data for this report was collected in the field over a period of five days from the 1st November to the 5th November 2010. In total, 158 trees were sampled across sixteen orchard sites.
28 trees were observed as G. nobilis positive.
12 orchards were recorded as G. nobilis positive.
4 orchards were recorded as G. nobilis negative.
The highest number of G. nobilis positive trees was found at Cherry Orchard, Bowcastle Farm.
Previously unrecorded G. nobilis sites were found at Hole Farm, The Cottage, Dreamskerry, Far Forest School, Hawkbatch Farm, The Latchetts and Starrs Hill.
One orchard, Tarn, was recorded in previous Worcestershire Biological Records Centre surveys as G. nobilis positive, however in this study there was no evidence of G. nobilis in this orchard.
The results of this study are summarised in table 1.
| Habitat Parameter | Relationship with G. nobilisPresence? | Evidence |
| Tree Characteristics: | ||
| Tree Species – Tree Level | P. domestica preferred by G. nobilis | 33.33% of Prunus domestica trees contained G. nobilis. |
| Tree Species – Orchard Level | P. avium preferred by G. nobilis | 83.87% of Prunus avium trees were in G. nobilis positive orchards. |
| Statistical Analysis of Tree Species Preference | None | H0 was accepted – G. nobilis has no preference for particular orchard tree species. |
| Orchard Tree Species Composition | G. nobilis appear to prefer more diverse orchards | G. nobilis positive orchards contained a higher number of different tree species than in G. nobilis negative orchards. |
| Tree Girth – Tree Level | G. nobilis was found in trees with a larger mean tree girth, and a smaller tree girth range than G. nobilis negative trees. | Mean tree girth of G. nobilis positive trees was 138.29cm, whereas G. nobilis negative trees had a mean tree girth of 127.02cm. The range of tree girth was 155cm in G. nobilispositive trees, and 230cm in G. nobilis negative trees (see Discussion). |
| Tree Girth – Orchard Level | Not a considerable difference in mean tree girth between G. nobilispositive orchards and G. nobilis negative orchards. Tree girth range was lower in G. nobilis positive orchards. | Mean tree girth of G. nobilis positive orchards was 131.54cm, whereas in G. nobilis negative orchards mean tree girth was 129.03cm. A difference of only 2.51cm. Tree girth range was 30cm larger in G. nobilis negative orchards than G. nobilis positive orchards. |
| Tree Girth – Statistical Analysis – Tree Level | None | H0 was accepted for all tree species – There was no difference in median girth between specific fruit trees with G. nobilis present and specific fruit trees with G. nobilis absent. |
| Tree Girth – Statistical Analysis – Orchard Level | Significant difference (at the 95% confidence level) in median girth of P. aviumtrees between orchards which had G. nobilispresent, and those where it was absent. No difference between girth of G. nobilis positive and negative orchards with any other tree species. | P-value for P. avium was 0.028, therefore the H1 hypothesis was accepted, that there was a difference in median girth of P. avium trees between orchards with G. nobilispresent and those where it was absent. |
| Adjacent Orchard Surrounding Habitat | G. nobilis seemed to prefer a higher diversity of surrounding habitat, with slightly higher proportion of orchards. | Surrounding habitat of G. nobilispositive orchards was composed of 18% orchards, in G. nobilis negative orchards, this was 12%. There were 6 different categories of surrounding habitat in G. nobilis positive orchards, compared with 4 categories in G. nobilis negative orchards. |
| Orchard Management | Less intense management and therefore a higher amount of dead wood could be preferred by G. nobilis | Less intense management techniques were more prominent in G. nobilispositive orchards. In orchards where dead wood was left and no management combined had the highest percentage of 38% in G. nobilis positive orchards and 19% in G. nobilis negative orchards. In G. nobilis negative orchards, there were no orchards where dead wood was intentionally left in situ. |
| Orchard Microclimate: | ||
| Tree Density | None | Statistical analysis showed that there was no difference in orchard tree density and G. nobilis presence (H0accepted). However, 75% of G. nobilisnegative orchards did have tree densities of 94 trees per hectare and above. |
| Orchard Size | None | H0 was accepted, there was no difference in orchard size between G. nobilis positive orchards and G. nobilis negative orchards |
| Orchard Aspect | None | 41.6% of G. nobilis positive orchards were situated on a South facing slope. 75% of G. nobilis negative orchards were on non South facing slopes. |
Table 1. Summary of the results of the study.
Discussion
On the tree level, P. domestica trees had the highest percentage of G. nobilis presence (33.33%), with P. avium second (22.58%). However, on the orchard level, P. domestica was the least preferred (33.33%) and P. avium was most favoured (83.87%). This could suggest that G. nobilispreferably use individual trees of this species to inhabit rather than just orchards with this tree in. However, P. domestica had to be eliminated from statistical tests as there was a low abundance of the species in this study. The results in this report suggest that P. avium trees are also preferred over other species (except P. domestica) for colonisation, as G. nobilis are commonly found in purely P. avium orchards; in orchards where P. aviumand other species are present; and G. nobilis colonised a high number of P. avium trees (21 Trees, 22.58% of all G. nobilis positive trees).
Heartwood decay fungi such as Sulphur Polypore Laetiporus sulphurous are a common feature of old traditional orchards (Natural England, 2010). These fungi do not harm the host tree, but attack the cellulose of the tree cells resulting in red rot and white rot, creating cavities in the tree, which G. nobilis and other wildlife can exploit (Natural England, 2010). The fungi soften the wood making it accessible for saproxylic invertebrates to feed on (Woodland Trust, 2011). G. nobilis preferentially eat away at the wood mould inside the tree column, which has been attacked by L. sulphurous (Woodland Trust, 2011). L. sulphurous is a particularly common on P. domestica and P. avium trees (Smith, 1988). This could suggest why P. domestica and P. avium trees were favoured by G. nobilis in this report. Because of these fungi, trees have softer wood mould contained within them, which is easier for G. nobilis to digest.
A higher diversity of tree species in each individual orchard was possibly preferred by G. nobilis, which has not been shown in previous studies.
In G. nobilis positive orchards and trees the tree girth of all species, accounted together, had a larger girth. In G. nobilis negative orchards and trees the range of tree girths was smaller. This suggests there was a small range of large tree girths where the dead wood was suitable for G. nobilis. PTES (2010) have stated that G. nobilis inhabits orchard fruit trees with a girth of between 62cm and 116cm, a range of 54cm. However, in this study, G. nobilis was found in trees between 55cm and 210cm; a much larger range of 155cm. This could be due to different tree species producing wood mould at different ages (Dubois et al. 2009) who stated that saproxylic beetles preferred or avoided species according to the amount of wood mould found in the tree, this being due to the characteristics of the tree itself.
Tree girth of all tree species were different in G. nobilis positive trees compared to G. nobilis negative trees: mean 138.29cm in G. nobilis positive trees and 127.02cm in G. nobilis negative trees. This could equate to about ten years age difference, which could affect how much dead wood is in the tree (PTES, 2010). The larger trees could therefore be older, and produce more wood mould (Ranius, 2002) which G. nobilis larvae could utilise as a food source.
P. avium trees had a significant difference in tree girth between the orchards that did and did not contain G. nobilis (p-value 0.028 at 95% confidence level). G. nobilis positive orchards contained P. avium trees 25.61cm larger on average than in G. nobilis negative orchards. This could be due to the age of the P. avium trees; older trees could contain more suitable dead wood conditions for G. nobilis (Dubois et al., 2009). In Ranius’ (2002) study, several saproxylic species occurred with higher frequencies in trees with a larger girth, this being due to larger amounts of wood mould found.
These results could, however, simply suggest colonisation at different times. The older trees could have sustained G. nobilis populations for a much longer time than populations in the younger trees (Ranius, 2002). The larger the tree girth, which signifies older trees, the longer species can persist because of the larger amounts of wood mould present (Ranius, 2002).
The small girths at The Cherries Orchard consisted of four different species of tree; P. domestica (55cm GBH), Malus domestica (80cm GBH), P. communis (65cm GBH) and P. avium (95cm GBH). Therefore this suggests that it is age and not tree species that G. nobilis prefers (Alexander, 2002). However, the P. communis tree with a GBH of 65cm had a rot hole, created by a woodpecker. (GBH = Girth at Breast Height). This rot hole, where woodpeckers have acted as allogenic engineers, may not have been caused by the natural decay process of a tree, but it could also contain G. nobilis(Jones et al., 1994). The softest part of a tree is usually chosen by woodpeckers, though the outer layer must be strong enough to support the excavated cavity (Jones et al. 1994) and in this case the softest part of the tree is the rotten core. This could suggest that the harder outside of the tree had not started decaying, and therefore the decay inside the tree was not exposed for G. nobilis colonisation but as the woodpecker pecked through the harder tree exterior, the wood mould inside became accessible for G. nobilis to inhabit (PTES, 2010). The M. domestica (GBH 80cm) and the P. avium (GBH 95cm) at The Cherries were both dead. Therefore, dead wood was available as the tree was being broken down by organisms in the natural nutrient recycling process of decomposition, so producing soft dead wood that G. nobilis can digest (Whitehead 2003; Blakesley and Buckley 2010). These trees could have died at a young age, hence their relatively small girths, but as they have died, they can provide the dead wood that G. nobilis favours (PTES, 2010).
It is suggested that small clusters of G. nobilis positive orchards interact at some levels. This was shown in the results from the surrounding orchard habitat. In G. nobilis positive orchards, 41% of the surrounding habitat was fields and 18% was orchards. The high proportion of fields could have been old orchards which have been cleared for agriculture. The proportion of orchard surrounding habitat around the G. nobilis positive orchards in this report could suggest that G. nobilis interact with these surrounding orchards on the metapopulation scale for food and mates. These metapopulations could be areas that G. nobilis has been forced to occupy, since their first choice of habitat locations have been altered (Keble 2001). The fields present unsuitable habitat for G. nobilis, whilst the orchards are the suitable habitat within which they interact. 18% of the orchard surrounding habitat is woodland, which could mean that G. nobilis interacts with the ancient woodland of the Wyre Forest. There are two suggestions as to why this might be. Franc et al. (2007) suggested that saproxylic beetles interact with woodland as it provides a corridor of dead wood for colonisation. A higher number of different surrounding habitats occur in G. nobilis positive orchards. As Økland et al. (1996) observed a richer mosaic of different habitats increased saproxylic species presence, due to the different habitats provided, creating the specific niches for saproxylic species.
In G. nobilis negative orchards there was a higher proportion of woodland surrounding habitat (25%) compared to G. nobilis positive orchards (18%). Denser woodland may not facilitate the movement of adults (Kindlmann, 2005) and a large canopy provides a habitat which is too cold and damp for G. nobilis (Alexander, 2008). For example at Lodge Hill Farm a G. nobilisnegative orchard is surrounded by woodland on three sides. It could be a G. nobilis negative orchard because colonisation of this orchard could be difficult, as G. nobilis may not be able to penetrate through the dense woodland to find this orchard site (Franc et al. 2007). Lodge Hill Farm was not in close enough proximity (within 700m) to any other G. nobilis positive orchards for possible colonisation.
As Whitehead (2003) identified, the largest distance that G. nobilis has been recorded to fly is 700m. Fig. 1. shows G. nobilis positive sites, and a 700m buffer zone around each one. This shows that there could possibly be two metapopulations in the area, one around The Cottage and Dreamskerry (2 and 4), and another around Bowcastle Farm (7, 8 and 9), Hole Farm (10) and Uncllys (7). These two areas had G. nobilis populations within 700m of each other, therefore they could potentially interact at some level, for food sources, trees to colonise and mates (Levins, 1969).
However, other G. nobilis positive orchards such as Far Forest School (11), The Latchetts (16), and Hawkbatch Farm (3) are not in close proximity to any other G. nobilis positive orchards. It could be that these orchards were once connected by other orchards, but have suffered from habitat fragmentation. As Whitehead (2006) identified, these isolated G. nobilispositive orchards could prove that beetles and larvae can survive in one tree for several generations. These G. nobilis populations could have inhabited the trees in these orchards utilising the wood mould resource for many years (Whitehead, 2007). As the larvae develop, their mandibles become larger and stronger, enabling the larvae to eat their way through the tree trunk where the wood may be a lot harder and where there is less water and oxygen (Whitehead, 2006).
It can be seen that three out of four G. nobilis negative orchards are not within a 700m flight distance of any G. nobilis positive orchards. This would suggest that it is hard for G. nobilis to colonise these negative orchards as they are not in close proximity to any G. nobilis populations. However, Brooklands was in very close proximity to a G. nobilis positive site: The Cherries. Brooklands was managed as a garden, so there was no dead wood present, suggesting why G. nobilis had not been found inhabiting this orchard (PTES, 2010). In all G. nobilis negative orchards, dead wood had been removed by management practices such as pruning. In G. nobilispositive orchards, 27% of orchards had the dead wood left intentionally, and 11% of G. nobilis positive orchards had no management, which both meant that a considerable amount of dead wood was present. This would suggest that the higher the proportion of dead wood, the greater the chances are of species like G. nobilis inhabiting these orchards, as a dead wood resource is crucial to their life cycle (Speight 1989; Alexander 2008; PTES 2010; Whitehead 2003). In 1% of G. nobilis positive orchards, orchard trees had been pollarded, and in 11% of G. nobilis positive orchards, pruning was apparent. Pollarding and pruning can create crevices and rot holes as the cuts created by a saw mimic natural tears providing colonisation sites for saproxylic species (Fay and Berker 1997; Forestry Commission 2002). Therefore, 50% of G. nobilis positive orchards in the Wyre Forest area involved management practices that encouraged dead wood to form on living trees, or kept dead wood in situ in the orchards, without clearance.
The UK BAP (2010) stated that one of the threats to G. nobilis is the loss of nectar and pollen sources through inappropriate management of orchard grassland, such as intense grazing. In G. nobilis negative orchards, the largest proportion of management was grazing (39%), which could be why G. nobilis are not found in these orchards as a food source is not available. The grazing animals could have eaten the plants such as Hogweed Heracleum sphondylium and Yarrow Achillea millefolium before they come into flower so the nectar that G. nobilis feed on was not available.
Even though it did not show up on statistical tests (p-value 0.5852 at 95% confidence level), a majority of G. nobilis positive orchards had the lowest tree densities. This is probably due to the warm microclimate that a low tree density creates. Each individual tree is more sun exposed as there is less shading (Ranius, 2002). Beetles in tree hollows prefer a stable warm temperature and lack of moisture (Kelner-Pillault 1974), which is what a south facing slope provides. It could also mean that the umbel flowers which G. nobilis commonly feeds on have enough light to grow on the orchard floor (Database of Insects and their Food Plants, 2010), as they are species that need plentiful amounts of light to grow (Heywood, 1971).
Orchard size did not affect whether or not G. nobilis was present (p-value 0.2029 at 95% confidence level). This suggests that it is the characteristics of the habitat inside the orchard, rather than the size of the orchard, which could attract or deter G. nobilis.
In relation to orchard aspect, there was not a strong relationship between G. nobilis presence and South facing slopes. 75% of G. nobilis negative orchards were on not on south facing slopes, which is as expected due to the colder microclimate. Out of all G. nobilis positive sites, 41.6% were on south facing slopes, a far lower percentage than predicted. Ranius and Jansson (2000) found that saproxylic beetles preferred sun exposed trees, but the relationship was not found in Ranius’ (2002) survey. Sun exposure may have changed in the study sites during the last few years, as a majority of the trees now had shading due to dense surrounding vegetation (Ranuis, 2002). However, the saproxylic invertebrates still inhabited the trees because of favoured conditions in the past. The only G. nobilis positive orchard in this survey with dense surrounding vegetation was The Cottage but this was sun exposed on a South facing slope.
On closer inspection, looking at microclimate as a whole, there does seem to be a relationship. Apart from Far Forest School, all non South facing slopes had the lowest tree densities. The Cherries had the highest tree density, but as this was on a South facing slope. Tree density did not seem to deter G. nobilis. G. nobilis positive orchards either need to be south facing or have a low tree density, because G. nobilis being cold blooded, need heat (Ball 1985).
At this stage in the research, there were only some habitat parameter patterns that emerged. The current literature suggests patterns of habitat parameters can be explained. There are possible anomalies and limitations to this report, such as Tarn, which pose further questions for their findings. At the beginning of this study sixteen orchards were identified: six orchards with recorded G. nobilis populations and ten without. When undertaking this research new populations were recorded in a higher number of orchards than previously expected. This will contribute to the Worcestershire Biological Records Centre’s records of the species in this area. However, a previously recorded G. nobilis positive orchard came out as G. nobilisnegative in this study: Tarn. This is most likely to be due to the random methods of sampling in this report. This highlights the need for more research into G. nobilis in the Wyre Forest area.
However, it needs to be noted that although habitat parameter relationships have emerged with relation to G. nobilis presence, and there is literature to support these explanations, no real conclusions can be made, due to a data set with only four G. nobilis negative orchards. It is hoped that future research can use the data in this report as a baseline for further studies on G. nobilis in the Wyre Forest area.
As this study relied on the presence or absence of G. nobilis frass which may persist in the wood mould over several years, the occupancy patterns observed may not reflect recent habitat changes, but may be to a result from the situation in the past (Ranius 2002).
References
Alexander, K. 2004. The Noble Chafer Gnorimus nobilis in Gloucestershire- a Report on the 2003 Survey, Unpublished Report for the People’s Trust for Endangered Species.
Alexander, K. 2005. The Noble Chafer Gnorimus nobilis in Gloucestershire- a Report on the 2004 Survey, Unpublished Report for the People’s Trust for Endangered Species.
Alexander, K. 2008. The special importance of traditional orchards for Invertebrate conservation, with a case study of the BAP species the Noble Chafer Gnorimus nobilis, in Rotherham, I. 2008. Orchards and Groves: Their History, Ecology, Culture and Archaeology, Landscape and Ecology, 7.
Alexander, K. 2010. Saproxylic Beetles, In: Newton, A. 2010. Biodiversity in the New Forest, Information Press, Oxford.
Allen, A. 1960. The history and present day status of Gnorimus variabilis L. (Col., Scarabaeidae) in Britain, Entomologist’s Record and Journal of Variation, 72: 129-132.
Baille, J., Hilton-Taylor, C., and Stuart, S. 2004. 2004 IUCN Red List of Threatened Species: A Global Assessment. International Union for Conservation of Nature, Cambridge, UK.
Ball, G.1985. Taxonomy, Phylogeny and Zoogeography of Beetles and Ants, Dr W. Junk Publishers, Dordrecht
Berkshire Biodiversity Action Plan. 2010) Traditional Orchards, [online] Available at: http://www.berksbap.org/traditional-orchards-1 [Accessed 20.08.2010]
Blakesley, D., and Buckley, G. 2010. Managing Your Woodland for Wildlife, Pisces Publications, Newbury
Boatman, N., Crocker, D., Hart, A., Roelofs, W., Smith, R., Holland, J., Lutman, P., Brown, V., and Ramsay, A. 2004. Development of a scheme for the assessment of risks to wider biodiversity arising from the use of pesticides, Final report to DEFRA
Bouget, C., Brustel, H., and Zagatti, P. 2008. The French information system on saproxylic beetle ecology (FRISBEE): An ecological and taxonomical database to help with the assessment of forest conservation status, Revue d’ Ecologie (la Terre et la Vie) 10:33-36.
Buse, J., Schröder, B., and Assmann, T. 2007. Modelling habitat and spatial distribution of an endangered longhorn beetle- a case study for saproxylic insect conservation, Biological Conservation, 137: 372-381.
Callaway, R., Reinhart, K., Moore, G., Moore D., and Pennings, S. 2002. Epiphyte host preferences and host traits: mechanisms for species-specific interactions, Population Ecology, 132(2):221–230.
Database of Insects and their Food Plants, (2010), Coleoptera >> Scarabaeidae >> Gnorimus nobilis (L.) [online], Available at: http://www.brc.ac.uk/DBIF/invertebratesresults.aspx?insectid=4100, [Accessed 16.10.2010].
Davies, K., Margules, C., and Lawrence, J. 2000. Which traits of species predict population declines in experimental forest fragments?, Ecology, 81: 1450–1461.
Dubois, G., Vignon, V., Delettre, Y., Rantier, Y., Vernon, P., and Burel, F. 2009. Factors affecting the occurrence of the endangered saproxylic beetle Osmoderma eremite (Scopoli, 1763) (Coleoptera: Cetoniidae) in an agricultural landscape, Landscape Planning 91:152-159.
Eccleson, P. 2007. Noble Chafer found in Kent [online], Available at: http://www.telegraph.co.uk/earth/earthnews/3317524/Noble-chafer-beetle-found-in-Kent.html [Accessed 23.09.2010].
Ehnström, B. 2001. Leaving dead wood for insects in boreal forests – suggestions for the future, Scandinavian Journal of Forest Research 3: 91–98.
European Red List. 2010. An Introduction to Saproxylic Beetles [online], Available at: http://ec.europa.eu/environment/nature/conservation/species/redlist/beetles/introduction.htm, [Accessed 08.01.2011].
Fay, N., & Berker, N. 1997 Veteran Trees Initiative: Specialist Survey Method, English Nature.
Ferris-Kaan, R., Londsdale, D., and Winter, T. 1993. The Conservation Management of Dead Wood in Forests, In: Research Information Note 241, Forestry Authority, Holt Alice.
Forestry Commission. 2010. Wyre Forest Orchard Restoration Underway [online], Available at: http://www.forestry.gov.uk/newsrele.nsf/WebPressReleases/E2CCBE6B9123CE108025754B0053A3AC [Accessed 1.10.2010].
Forestry Commission. 2002. Life in the Dead Wood, A Guide to managing deadwood in Forestry Commission Forests, Forest Enterprise, Bristol.
Franc, N., Götmark, F., Økland, B., Nordén, H., and Paltto, H. 2007. Factors and Scales Potentially Important for Saproxylic Beetles in Temperate Mixed Oak Forest, Biological Conservation, 136:86-98
Fry, R., and Lonsdale, D., (1991), Habitat Conservation for Insects – A Neglected Green Issue, The Amateur Entomologists’ Society, London.
Google Maps, (2011), Google Maps [online], Available at: http://maps.google.co.uk/ [Accessed 05.01.2011].
Green, H. 2004. The Noble Chafer Gnorimus nobilis: records needed, Worcestershire Record, 16:10-11 [online], http://www.wbrc.org.uk/WorcRecd/Issue%2016/noble_chafer.htm [Accessed 10.08.2010]
Green, P., and Peterken, G. 1997. Variation in the Amount of Dead Wood in the Woodlands of the Lower Wye Valley, UK in Relation to the Intensity of Management, Forestry Ecology and Management, 98:229–238.
Groombridge, B. 1992 Global biodiversity : Status of the Earth’s Living Resources – World Conservation Monitoring Centre, Chapman & Hall, London.
Grove, S. 2002. Saproxylic Insect Ecology and the Sustainable Management of Forests, Annual Review of Ecology, Evolution and Systematics, 33:1–23.
Hampshire Biodiversity Partnership. 2001. Noble Chafer, Gnorimus nobilis: Species Action Plan [online], Available at: http://www.hampshirebiodiversity.org.uk/pdf/PublishedPlans/NobleChaferVerIISAP.pdf, [Accessed 10.10.10].
Harding, P., and Rose F. 1986. Pasture-woodlands in Lowland Britain; A Review of Their Importance for Wildlife Conservation, Institute of Terrestrial Ecology, Huntingdon, UK.
Heywood, V. 1971. The Biology and Chemistry of the Umbelliferae: Linnean Society of London Journal of Botany, Academic Press, New York.
Hickin, N. 1978 The Natural History of an English Forest; The Wild Life of Wyre, Westmid Supplies, Shrewsbury.
Holland, P., and Steyn, D. 1975. Vegetation responses to latitudinal variations in slope angle and aspect, Journal of Biogeography, 2:179-183.
Hulme, M., Turnpenny, J., and Jenkins, G. 2002. Climate Change Scenarios for the United Kingdom: the UKCIP02 Briefing Report, Tyndall Centre for Climate Change Research, Norwich, UK.
IUCN. 2010. The IUCN Red List of Threatened Species [online], Available at: http://www.iucnredlist.org/ [Accessed 10.08.2010].
Jones, C., Lawton, J., and Shachak, M. 1994. Organisms as ecosystem engineers, Okios, 69:373-386.
Jonsson, B., and Kruys, N. 2001. Ecology of Woody Debris in Boreal Forests, Ecological Bulletins, 49:1–283.
Keble, D. 2001. Wild Things; Noble Chafer Beetle, [online] Available at: http://www.telegraph.co.uk/gardening/3292356/Wild-things-noble-chafer-beetle.html [Accessed 25.08.2010].
Keech, D. 2000. Common Ground Book of Orchards: Community, Conservation and Culture, Common Ground, London.
Kelner-Pillault, S. 1974. Ecological Study of the Stand Entomology Terraux Hollow Tree (Chestnuts and Willows), Bulletin d’ Ecologie, 5:123–156.
Key, R. 1992. What are Saproxylic Invertebrates? In: Kirby, K., & Drake C. 1992). Dead Wood Matters: The Ecology and Conservation of Saproxylic Invertebrates in Britain. 1992, English Nature, English Nature Science: 7.
Key, R. 2011. Noble Chafer (Gnorimus nobilis) [online], Available at: http://www.arkive.org/noble-chafer/gnorimus-nobilis/image-A6264.html#text=All [Accessed 24.01.2011].
Kindlmann, P., Aviron, S., and Burel, F. 2005 When is landscape matrix important for determining animal fluxes between resource patches? Ecological Complex, 2:150–158
Kirby, K. 1992. Accumulation of dead wood: a missing ingredient in coppicing?, in G P Buckley [editor], Ecology and Management of Coppice Woodlands, Chapman and Hall, London.
Kouki, J., Löfman, S., Martikainen, P., Rouvinen, S., and Uotila, A. 2001. Forest fragmentation in Fennoscandia: linking habitat requirements of wood-associated threatened species to landscape and habitat changes, Scandinavian Journal of Forest Research, 3:27–37.
Krejčík, S. 2008. Gnorimus nobilis Linnaeus [online], Available at: http://www.eol.org/pages/141887, [Accessed 01.12.2010].
Levins, R. 1969. Some demographic and genetic consequences of environmental heterogeneity for biological control, Bulletin of the Entomological Society of America, 15:237-240.
Martikainen, P., Siitonen, J., Kaila, L., Punttila, P., and Rauh, J. 1999. Bark beetles (Coleoptera, Scolytidae) and associated beetle species in mature managed and old-growth boreal forests in southern Finland, Forest Ecology and Management, 116:233–245.
Marrotte, E. 2003. Why Fruit Trees Fail to Bear [online], Available at: http://www.uri.edu/ce/factsheets/sheets/fruittreesfail.html [Accessed 21.03.2011].
National Trust. 2010. Apples and Orchards, [online] Available at: http://www.nationaltrust.org.uk/main/w-chl/w-countryside_environment/w-nature/w-nature-orchard-restoration/w-nature-orchards-history.htm [Accessed 10.08.2010].
Natural England. 2010. Traditional Orchards: Orchards and Wildlife [online], Available at: http://naturalengland.etraderstores.com/NaturalEnglandShop/TIN012 [Accessed 23.03.2011].
Økland, B., Bakke, A., Hagvar, S., and Kvamme, T. 1996. What factors influence the diversity of saproxylic beetles? A multi-scaled study from a spruce forest in southern Norway, Biodiversity and Conservation, 5:75–100.
People’s Trust for Endangered Species 2010. The Noble Chafer beetle [online] Available at: http://www.ptes.org/?page=170 [Accessed 08.08.2010].
Prime, C. and Deacock, R. 1970. Trees and shrubs and their identification in summer or winter, Heffer, Cambridge
Rackham, O. 2001. Trees and Woodland in the British Landscape: The Complete History of Britain’s Trees, Woods and Hedgerows, Phoenix Press, London.
Ranuis, T. 2002. Influence of stand size and quality of tree hollows on saproxylic beetles in Sweden, Biological Conservation, 103 (1):85-91.
Ranius, T., and Jansson, N. 2000. The influence of forest re-growth, original canopy cover and tree size on saproxylic beetles associated with old oaks, Biological Conservation, 95:85–94.
Ranius, T., and Jansson, N. 2002. A comparison of three methods to survey saproxylic beetles in hollow oaks, Biodiversity and Conservation, 11:1759-1771.
Ranney, J., Bruner, M., and Levenson, J. 1981. The Importance of Edge in the Structure and Dynamics of Forest Islands, In Burgess, R. & Sharpe, D. Forest island dynamics in man-dominated landscapes, Springer, New York.
Read, H. 2000. Veteran Trees: A Guide to Good Management, Natural England.
Rosenberg, B. 1983. Microclimate: The Biological Environment, John Wiley and Sons, London.
Saint-Germain, M., Drapeau, P., and Buddle, C. 2007. Host-use patterns of saproxylic phloeophagous and xylophagous coleoptera adults and larvae along the decay gradient in standing dead Black Spruce and Aspen, Ecography, 30:737-748.
Saunders, D., Hobbs, R., and Margules, C. 1991. Biological Consequences of Ecosystem Fragmentation: A Review, Conservation Biology, 5:18–32.
Similä, M., Kouki, J., and Martikainen, P. 2003. Saproxylic beetles in managed and semi-natural Scots Pine forests: quality of dead wood matters, Forest Ecology and Management, 174:365–381.
Smart, M., and Winnall, R. 2006. The Biodiversity of Tree Traditional Orchards within the Wyre Forest SSSI in Worcestershire: A Survey by the Wyre Forest Study Group, English Nature Research Report 707: 1-89.
Smith, I. 1988. European Handbook of Plant Diseases, Blackwell Publishing, Oxford.
Smith, M.(2004. Just Leave the Dead to Rot, The Guardian newspaper, March 25th 2004, London.
Smith, M. 2005. Matt Smith’s Report 2005; Worcestershire Noble Chafer Survey, Unpublished Report to People’s Trust for Endangered Species
Smith, M. 2003 Saproxylic beetles in Britain, an overview of the status and distribution of four biodiversity action plan species, In Bowen, C., Proceedings of the second pan-European conference on Saproxylic Beetles, PTES, London.
Smith, R., and Smith, T. 2002. Elements of Ecology, 5th Edition, Benjamin Cummings Publishing, New York.
Speight, M. 1989. Saproxylic Invertebrates and their Conservation, Council of Europe, Strasbourg.
UK Biodiversity Action Plan. 2010. Species Action Plan; The Noble Chafer Gnorimus nobilis [online] Available at: http://www.ukbap.org.uk/UKPlans.aspx?ID=326#1 [Accessed 12.10.2010].
UK Biodiversity Steering Group. 1999. Action Plans: Volume IV-Invertebrates, English Nature, Peterborough, England.
Väisänen, R., Biström, O., Heliövaara, K. 1993. Sub-cortical Coleoptera in dead pines and spruces: is primeval species composition maintained in managed forests? Biodiversity and Conservation, 2:95–113.
Vernon, P., and Vannier, G. 2001. Freezing susceptibility and freezing tolerance in Palaearctic Cetoniidae (Coleoptera), Canadian Journal of Zoology, 79:67–74.
Wermelinger, B., Flückiger, P., Obrist, M., and Duelli, P. 2007. Horizontal and vertical distribution of saproxylic beetles (Col., Buprestidae, Cerambycide, Scolytine) across sections of forest edges, Journal of Applied Entomology, 131(2):104-114.
Wheater, C., and Cook, P. 2003. Using Statistics to Understand the Environment, Routledge, London.
Whitehead, P. 1990. Further Observations on Gnorimus nobilis (L.) (Col., Scarabaeidae) in Worcestershire. Entomologist’s Monthly Magazine, 126:110.
Whitehead, P. 1990a. Rare Beetles from an apple tree in Worcestershire, Entomologist’s Monthly Magazine, 126:236.
Whitehead, P. 1997. Invertebrates of pome and stone fruit orchards in the English Wyre Forest, July 1997, with particular regard to Gnorimus nobilis(L., 1758) (Col., Scarabaeidae) pp. 1-8, Unpublished Report for English Nature.
Whitehead, P. 1999. The 1999 Worcestershire Survey for Gnorimus nobilis(L., 1758) (Col., Scarabaeidae), Unpublished Report for the People’s Trust for Endangered Species.
Whitehead, P. 2000. The 2000 Gloucestershire survey for Gnorimus nobilis(L., 1758) (Col., Scarabaeidae), Unpublished Report for the People’s Trust for Endangered Species.
Whitehead, P. 2000a. The 2000 Worcestershire Survey for Gnorimus nobilis(L., 1758) (Col., Scarabaeidae), Unpublished Report for the People’s Trust for Endangered Species.
Whitehead, P. 2001. The 2001 Gloucestershire Survey for Aleurosticus nobilis (L., 1758) (Col., Scarabaeidae), Unpublished Report for the People’s Trust for Endangered Species.
Whitehead, P. 2001a. The 2001 Worcestershire Survey for Aleurosticus nobilis (L., 1758) (Col., Scarabaeidae), Unpublished Report for the People’s Trust for Endangered Species.
Whitehead, P. 2001b. Evidence of the Noble Chafer Aleurosticus nobilis (L., 1758) (Col., Scarabaeidae) in Warwickshire during 2001, Unpublished Report for the People’s Trust for Endangered Species.
Whitehead, P. 2003. The Noble Chafer Aleurostictus nobilis (L., 1758) (Col., Scarabaeidae) in Britain, Proceedings of the Second pan-European conference on Saproxylic Beetles; People’s Trust for Endangered Species, London.
Whitehead, P. 2006. Larvae of Noble Chafer Aleurostictus nobilis (L.) (Col., Scarabaeidae) developing terrestrially in Gloucestershire, Entomologist’s Monthly Magazine, 142:228.
Whitehead, P. 2007. Noble Chafer Aleurostictus nobilis (L., 1758) (Col., Scarabaeidae) Breeding externally on a pear tree, Entomologist’s Monthly Magazine, 143:206.
Woodland Trust. 2011. Ancient Trees and Wildlife [online], Available at: http://frontpage.woodland-trust.org.uk/ancient-tree-forum/information/ecology/natural_info.htm#Fungi and trees [Accessed 25.03.2011].
Worcestershire Biodiversity Partnership. 2008. Traditional Orchards Habitat Action Plan, H2 Traditional Orchard BAP, Unpublished Report.
WWF. 2004. Deadwood- Living forests, WWF Report October 2004.
Image
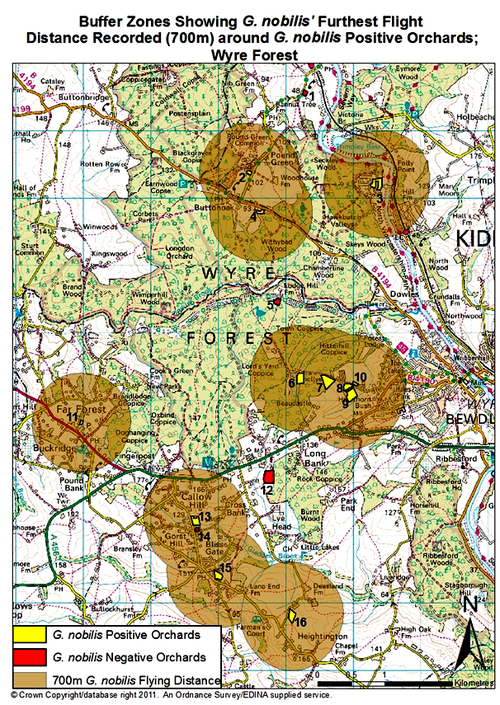
Fig. 1. Map of the Gnorimus nobilis positive and negative sites in this study. The buffer zones represent 700m around G. nobilis positive orchards; the largest recorded distance of G. nobilis flight (Whitehead 2003).
Worcestershire Record | 32 (April 2012) page: 25-32 | Worcestershire Biological Records Centre & Worcestershire Recorders
Notes on plant bugs (Heteroptera) in Worcestershire
| Harry Green | Introduction |
| Brett Westwood | Fallen’s Leatherbug Arenocris falleni in Worcester |
| Brett Westwood | Masked Hunter on the hearth Reduvius personatus |
| Jane Scott | Interesting Bugs at Woodbank, Astley Burf 2011 |
| Jane Scott | Forget-me-not Bug – Sehirus luctuosus |
| John Bingham | Trapezonotus (Trapezonotus) arenarius (L, 1758). (Lygaeidae) (Heteroptera) |
| John Bingham | Dicranocephalus medius (Mulsant & Rey, 1870) (Hemiptera) Status: Nb |
| John Bingham | Short notes on some Hemiptera found near Kidderminster |
| Harry Green | Corizus hyoscyami, a striking red and black bug |
| Harry Green | Brassica Bug Eurydema oleracea at Upper Blackstone Farm, Bewdley |
| various | Picture gallery |
Introduction
Harry Green
Since John Partridge’s (2004) review of the records of bugs (Hemiptera: Heteroptera) for Worcestershire followed by further encouragement in 2006 more people have taken an interest in the group aided by the excellent British Bugs website (http://www.britishbugs.org.uk ) and several books, notably Evans & Edmondson (2005), Hawkins (2003) and the re-issue of Southwood & Leston’s (1959) classic in 2005 as a paperback far less costly than the scarce original. John also contributed two further reports in 2007 (Pied Shieldbug and a rare bug found in a tussock) and John Meiklejohn (2011) wrote of red and black bugs. There is now also a considerable exchange of pictures taken by digital cameras via email amongst local naturalists which is helping many people to identify bugs and to generate more records.
The classification of True Bugs is complex but clearly set out by Barnard (2011). There is a picture of Pentatoma rufipes, a shieldbug, on the cover.
The range and abundance of several species in the county appear to be changing. John Partridge (2006) wrote of Corizus hyoscyami ‘The bright colouring of this bug should ensure we get more records’. As he predicted this species has become commoner and now often attracts the attention of natural history photographers. On the other hand The Bishops Mitre Aelia acuminate, which became commoner a few years ago, appears to have declined. The Western Conifer Seed Bug Leptoglossus occidentalis, an introduced species from North America, arrived in Worcestershire in 2011 (Holder 2011). The Box Bug Gonocerus acuteangulatus (Curran 2011) has been found in Edgbaston, Birmingham, and can be expected to arrive in Worcestershire before long. The Cow-wheat Bug Adomerus(Sehirus) biguttatus has been rediscovered at Monkwood and from this stimulus was searched for and found in Wyre Forest (Green 2011).
The following compilation (in no particular order) of notes from several active bug-watchers highlights some recent discoveries. There are also pictures of commoner species. Hopefully more people will now be encouraged to study and record bugs. They are an interesting and sometimes colourful group.
This compilation does not attempt cover all the bugs recorded in Worcestershire.
Fallen’s Leatherbug Arenocris falleni in Worcester
Brett Westwood
On August 10th 2011 a posse of naturalists visited Cherry Orchard on the east bank of the Severn south of Diglis in Worcester. The site is managed mainly by Worcester City Council and has a range of habitats including scrub, grassland and riverbank, but the main focus of the day was the area of river sand dredged from the Severn several years ago and dumped to create a substantial ‘dune’. While the conchologists sifted it for the remains of aquatic molluscs, some of us searched the bare sand for invertebrates. It was good to see the sand-loving, ginger-socked solitary bee Dasypoda hirtipes and the red-and black bug, Corizus hyoscyami consolidating their positions in Worcestershire, but my curiosity was drawn to some mottled greyish-brown squash-bugs hiding underneath the mats of fruiting Stork’s-bill Erodium cicutarium on the bare sand. They were beautifully camouflaged against the sand, but I had never seen anything like them before. On consulting Evans and Edmondson (2005) it was clear from photographs and description that these were Fallen’s Leatherbugs Arenocoris falleni which can be distinguished from similar species by the V-shaped cluster of spines on the pronotum. There are also no spines near the apex of the rear femur and the foodplant, Stork’s-bill, is an important clue to identification. Fallen’s Leatherbug is supposed to be coastal in distribution from Norfolk around the English and Welsh south coasts, but records inland are scarce and this appears to be the first record for Worcestershire VC37. How the bugs found this remote site we may never know, but this bug is worth searching for in other sandy places where Stork’s-bill grows.
Masked Hunter on the hearth Reduvius personatusBrett Westwood
Brett Westwood
On the evening of August 16th 2011 I was watching television in the living room of my house in Stourbridge, when from the corner of my eye I noticed a dark shape moving in a sinister fashion across the stone fireplace. Instead of the expected Tegenaria spider, this turned out to be a flattish primeval-looking creature completely covered in dust and sand grains. The camouflage was so complete that it was obviously no accident. A vague memory stirred of bug nymphs which do this, and after a short search, I was able to confirm the beast as Reduvius personatus, the Masked Hunter. This is a large predatory reduviid bug which generally lives in human habitation and feeds on silverfish, flies, harvestmen and, alarmingly, on bedbugs, though there is no evidence of the last in the house. Adults are black and can fly well, hence their other name of Flybugs, but the nymphs are wingless and cover themselves with granules of sand, mortar and dust, applying a new coating after each moult. They live for several months, stalking their prey under this camouflage. In the USA studies have revealed that they can over-winter twice in this nymphal state. The adults and nymphs can pierce human skin with their mouthparts.
It seems that though this bug was once relatively frequent in dwelling houses they are now scarce and Worcestershire Biological Records Centre (WBRC) has only eleven records, with only one other in the north of the county at Chaddesley Corbett. Jane Scott (pers.comm.) also confirms that she has recorded the adults at Astley Burf near Stourport-on-Severn, so it may simply be overlooked or perhaps under-recorded.
Interesting Bugs at Woodbank, Astley Burf 2011
Jane Scott
2011 proved to be a very interesting year, with two new species recorded and several records for a third species, which I had only seen very occasionally in the last few years.
Ceraleptus lividus – first individual was seen on 19th April having presumably overwintered as an adult, with two further individuals seen much later in the year, on 5th September and 6th October respectively. This species seems to be found in warm, dry situations, where the nymph feeds on various leguminous plants, many of which we have in the wilder parts of the garden.
Stictopleurus punctatonervosus – first one found on catmint on 17thOctober. This one had a damaged wing case whereas the second one, found two days later on lavender, was in good condition. A third was seen on 9thNovember, also on lavender. Care with identification is needed as this species can easily be confused with a closely related species Stictopleurus abutilon. It is suggested that both these species have a preference for plants in the Compositae family.
There are good images of all these species on British Bugs website, which was of particular help in distinguishing between the two Stictopleurusspecies. Stictopleurus abutilon is also illustrated by Kevin McGee(2007, 2009) http://www.wbrc.org.uk/WORCRECD/Issue/2027/uncommon_insects_mill_meadow.htmand http://www.wbrc.org.uk/WORCRECD/Issue/2023/records_of_note_2007.htm
Thanks go to Rosemary Winnall who spent time getting some excellent images of my specimens and to John Meiklejohn for his confirmation of my identification.
The third bug which made its presence felt in the garden was Corizus hyoscyami, a black and red bug that has expanded its range in the past few years. I recorded this several times in the garden between 13th July and 28th August, with three individuals seen at the same time on 22nd July. They seemed particularly attracted to lavender, but it may be that they are just easier to spot thereon, although I did also find one on the flowers of Rudbeckia.
We (Dave & Jane Scott) also recorded Corizus hyoscyami species from the following sites:
Longdon Orchard in Wyre Forest – Found under reptile refugia on 20th April. This specimen was very pale and lacked the bright red colouration, presumably as it had overwintered.
Deasland Farm, Nr Bewdley – One found on the flowerhead of ragwort on 3rd August.
Monkwood Reserve near Grimley – One seen on Bramble along main ride on 25th October
No doubt the warm, dry, late summer and autumn proved very beneficial for these insects, so it will be very exciting to see whether any of them make further appearances in 2012.
Forget-me-not Bug – Sehirus luctuosus
Jane Scott
For a number of years now I’ve been regularly seeing this small shiny black shieldbug in my garden in Astley Burf and had rather assumed this to be common in Worcestershire in suitable habitat. After chatting to fellow recorders, it became obvious that very few of them had ever seen this bug, a fact confirmed by the few records received at Worcestershire Biological Records Centre since the year 2000. There have been only fourteen records, seven of which were provided by Kevin McGee (2007) and with one exception, his were from the area around Drakes Broughton where he lived.
In my experience, the adult is most easily seen from the beginning of April until the end of July, with nymphs seen from mid June. They use warm dry situations with a plentiful supply of their foodplant, which not surprisingly is Forget-me-not species. I almost always see them on the ground below the Forget-me-nots Myosotis sp., either amongst the leaf debris or on bare soil. Rarely do I find them on the plants themselves. When disturbed they move very fast and will quickly disappear under the leaf litter or below the surface of the soil. Most are in the ‘gardened’ area but I also find them occasionally in the wilder, south facing area behind the house, which is a mixture of woodland and acid grassland.
I have singularly failed to get a good shot of the adult, but luckily I was able to get one or two pictures of a nymph found wandering about on a path in the garden. I’m hoping that 2012 will be the year that I finally manage to get a reasonable photograph of an adult.
I hope this short article will encourage others to have a good look around any Forget-me-nots in their own garden, or if they haven’t got this plant, to maybe consider introducing it by collecting some seed and scattering it in a suitable place in their plot. As long as you leave some to seed at the end of the season, they will self perpetuate freely.
Strangely, I have never found the bug in the wild, but have to admit to never having seriously searched for it. I hope to remedy this omission in the coming 2012 season.
Trapezonotus (Trapezonotus) arenarius (L, 1758). (Lygaeidae) (Heteroptera)
John Bingham
This ground bug is very similar to others in the general group but can be differentiated by the thin marginal strip to the pronotum with the mid and hind tibiae usually black. British Bugs website is helpful in sorting out the various species. (Bantock & Botting). So far no other Trapezontus species has been found in Worcestershire (as yet!). Southwood & Leston (1959) states they overwinter in adult stage and mate May and June after which they die with a new generation appearing as adult by August. Their habitat is varied but usually has dry soils such as heaths, light sandy soils and dry woodlands.
There are three records on NBN Gateway for Worcestershire:
Recorders day Grafton Wood WBRC recording day 11th July 1998.
One collected from sandy ground at Cherry Orchard, Worcester on 3rd August 2005 by K McGee, confirmed by P.F. Whitehead.
Recorders Day, Lodge Farm, Wyre Forest SO6365 on 10th June 2006, K McGee.
Also found in 2011 on the Recorders Day at Wilden Marsh on 6th August 2011 on sandy grassland SO8273. This last record is where I photographed it on acid grassland typical of those found around Kidderminster in an area of short turf with bare ground heavily grazed by rabbits.
It is not a particularly rare species and is widely distributed over England but there are only very scattered records. It is nearly absent from the south coast. It may be much commoner in Worcestershire than records suggest on any light, dry soil.
Dicranocephalus medius (Mulsant & Rey, 1870) (Hemiptera) Status: Nb
John Bingham
Somewhat surprisingly this is a very scarce species in the Midlands although the main food plant, Wood Spurge, is common. There are only two local records on NBN Gateway:
Wyre Forest.
Haugh Wood, Herefordshire.
It is on the County list for Worcestershire (Partridge 2004).
This species feeds on wood spurge Euphorbia amygdaloides, generally in open sheltered situations within woodlands. It can be found on the plant but it is well hidden. Observed mating on Wood Spurge in Wyre Forest on 26th May 2007. More often it occurs near to the food plant on the ground, typically amongst fairly open vegetation or on bare ground such as recently coppiced plots or areas of conifer clear-fell where there is a flush of wood spurge plants. Most record dates are in late May or early June in the Wyre Forest. Records from Surrey were made on 17th June (Hawkins 2003). Adults overwinter and then mate in May, the new generation appearing from August onwards (Bantock & Botting http://www.britishbugs.org.uk).
Despite the abundance of wood spurge in many Worcestershire woods there appear to be no records of Dicranocephalus medius except from Wyre Forest. It may need large areas of ancient semi-natural woodland to survive and presumably a good supply of wood spurge. As yet I personally have yet to see it on the Worcestershire side of the forest although it must be present? Despite the abundance of the food plant in Wyre, especially in recent years with ongoing coppice and conifer clearance, I have only managed to record it from three locations. It is elusive to say the least!
It should benefit from ride management to maintain open sunny conditions typical for many butterflies, or a good structured wood edge habitat or open glade situation.
Short notes on some Hemiptera found near Kidderminster
John Bingham
Eurydema oleracea found in a cleared conifer plot with regenerating birch woodland on hedge garlic [garlic mustard, Jack-by-the-hedge] Alliaria petiolata, Wyre Forest at Bell Coppice, Shropshire. This was the first record for the county 22nd May 2007. I’ve not seen this on the Worcestershire side of Wyre.
Zicrona caerulea found along a ride at Birchen Park just north of Wyre Forest, only scattered records for Shropshire, found 22nd March 2011.
Dicyphus epilobi found in garden at Kidderminster on Christmas day 2011, a very late date so presumably overwinters.
Nabis ferus found in my Kidderminster garden on low herbage on 11thSeptember 2011.
Rhabdomiris striatellus said to be associated with oak so would expect this to be common in Wyre Forest – found on 17th June 2011 along the disused railway line Worcestershire part of the Forest.
Rhyparochromus pini a southern species associated with heather found on 4th May 2011 at Longdon Wood, Shropshire, on a forest ride and on bare ground created by heathland restoration at Pound Green Common , Worcestershire on 10th September 2011.
Aelia acuminata turned up for the first time in my garden at Kidderminster on 10th September 2011.
Eurygaster testudinaria found in herb rich vegetation near Dowles Brook, Wimperhill Wood, Shropshire. The first time I have recorded this species in Wyre Forest, 10th July 2011.
Aneurus laevis often found under the bark of fruit or hawthorn trees, but any tree might do, scattered over Wyre Forest, 30th September 2010.
Corizus hyoscyami, a striking red and black bug
Harry Green
Corizus hyoscyami does not appear in the list of county records given by John Partridge (2004). Three records are mentioned by him in 2006 but the total had only reached five when John Meiklejohn (2011) wrote about red and black bugs in Worcestershire. His summary was of records received by 2010; additional records for 2010 arrived at the WBRC in 2011. However, 2011 itself was a bumper year for the species in Worcestershire with records from all over the county. Most bug-watchers found them and further records came in via the Worcestershire Wildlife Trust’s Facebook or Blog pages. The bugs were often seen on flowers, especially several species of thistles, and attracted the attention of photographers.
It is worth noting that British Bugs website reports another bug of somewhat similar appearance: Arocatus longiceps (Family: Lygaeidae). To quote the site: ‘This red and black bug is distinctive, although be sure to rule out the rhopalid Corizus hyoscyami. A recent immigrant to the UK, first noted in 2007 and by the end of this year extremely abundant on plane trees in parts of London. This species has recently expanded its range across Europe from the eastern Mediterranean. Initially thought to be Arocatus roeselii, the bug was found in profusion in the grounds of the Natural History Museum, prompting widespread news coverage. It feeds on the seeds of Plane, producing several generations before overwintering communally under bark. Flies readily in warm conditions, when the orange abdomen is conspicuous. Adult found all year, Length 6-7 mm’. This is perhaps one to arrive in Worcestershire in the future.
Corizus hyoscyami is distinctly hairy (well-shown in Pete Smith’s picture) which distinguishes it from similar-looking species.
Brassica Bug Eurydema oleracea at Upper Blackstone Farm, Bewdley
Harry Green
On 6th July 2011 I swept one Eurydema oleracea from mixed ‘arable weed’ vegetation within an arable field at Upper Blackstone Farm owned by Worcestershire Wildlife Trust. This is a scarce species of southern England reaching as far north at Gloucestershire (Evans & Edmondson 2005) although the more up-to-date British Bugs web site states ‘Well distributed in southern and central England’. As mentioned by John Meiklejohn (2011) there are two earlier Worcestershire records made by Kevin McGee (2007) and John Partridge but apparently none since in Worcestershire. John Bingham mentions elsewhere in these notes ‘found in a cleared conifer plot with regenerating birch woodland on hedge garlic Alliaria petiolata, Wyre Forest at Bell Coppice, Shropshire, 22nd May 2007. Not seen in the Worcestershire side of Wyre Forest’.
The species is found on a wide range Brassicaceae including Garlic Mustard Alliaria petiolata and Horse-radish Armoracia rusticana. Kevin McGee’s 2007 records were of bugs found on oilseed rape flowers.
These records suggest that the species may be extending its range northwards so it is worth searching for and obtaining records to check progress, if any.
The species can be as pest of brassica crops in Russia and possibly elsewhere according to http://www.agroatlas.ru/en/content/pests/Eurydema_oleracea/ attacking your plants.
The Brassica bug occurs in a variety of colour forms
Picture gallery
The following pictures show a range of bugs found in Worcestershire. Many are common, a few less so. Some species have a variety of colour forms as illustrated by the Tortoise Shieldbug Eurygaster testudinaria.
The Sloe Bug Dolycoris baccarum and Green Shieldbug Palomena prasinaare widespread and common. The larvae of Gorse Bug Piezodorus lituratusfeed on the seed pods of gorse and related Genisteae. Has been found on laburnum. The only record from my garden in Little Comberton was of one on a defunct nest of the wasp Dolichovespula media in a laburnum tree! (Green – personal observation)
Green shield bug Palomena prasina is common. The overwintering adults are much darker in colour.
Tortoise Shieldbug Eurygaster testudinaria is widespread in damp grasslands, often within woodland. The larvae feed on rushes.
Heather Bug Rhacognathuspunctatus is found on heathy vegetation, usually in damper areas. The larva is predatory on the Heather beetle Lochmaea suturalis and other beetles.
Juniper Shieldbug Cyphostethus tristriatus, once dependent on juniper berries, is now found on Lawson’s Cypress Chamaecyparis lawsoniana It appears to be extending its range and is widespread in midland and southern England.
The Parent Bug Elasmucha grisea is so-named because the female guards her eggs and young. Both are found in clusters on birch leaves.
Picromerus bidens is a predator of Lepidopteran larvae and other insects, sucking their juices rather than those of plants.
A ground species which climbs White Deadnettle Lamium album and other Lamiaceae. The female cares for her young laying eggs on the ground.
Rhyparochromus pini Although found widely in southern England, south and mid Wales, this is a scarce bug with a scattered distribution. found on heathland or similar habitats often under heather or in leaf litter.
The Woundwort Bug Eysarcoris fabricii is common and widespread in southern and midland England. It forms colonies on Hedge Woundwort Stachys sylvatica.
The Blue Bug Zicrona caerulea is widespread and is a predator leaf beetle(Chysomelidae)and lepidopetran larvae
References
Barnard, P. C. 2011. The Royal Entomological Society book of British Insects. Wiley-Blackwell.
Curran, A. 2011. Box bug Gonocerus acuteangulatus in Edgbaston. Worcestershire Record 30:23.
Evans, M. & Edmondson, R. 2005. A photographic guide to the Shieldbugs and Squashbugs of the British Isles. WGUK in association with WildGuide UK.
Green. H. 2011. Adomerus biguttatus (=Sehirus biguttatus), the Cow-wheat Shieldbug re-found in Worcestershire. Worcestershire Record 30:22.
Hawkins, R. D. 2003. Shieldbugs of Surrey. Surrey Wildlife Trust, Woking.
Holder, J. 2011. Western Conifer Seed Bug Leptoglossus occidentalis in Droitwich. Worcestershire Record 31:21
McGee, K. 2007. Records of note 2006. Worcestershire Record 21:29-33.
McGee, K. 2007. Records of note 2007. Worcestershire Record 23:42-46
McGee, K. 2009. An account of uncommon insects I have recorded at the Mill meadow/Mill Rough complex since 1996. Worcestershire Record 27:7-12
Meiklejohn, J.W. 2003. The squash bug Coreus marginata in Worcestershire. Worcestershire Record 14:34.
Meiklejohn, J.W. 2011. Red and black bugs (Hemiptera –Heteroptera) in Worcestershire. Worcestershire Record 30:23-24.
Partridge, J. 2004. Shieldbugs of Worcestershire. Worcestershire Record17:31-34
Partridge, J. 2006. Hemiptera records for Worcestershire. Worcestershire Record 21:41
Partridge, J. 2007. A rare bug in a tussock [Lasiosomus enervis]. Worcestershire Record 20:3
Partridge, J. 2007. The Pied Shieldbug Sehirus bicolor. Worcestershire Record 22:26
Southwood, T.R.E, & Leston,D. 1959. Land and Water Bugs of the British Isles. Warne.
Southwood, T.R.E. & Leston, D. 2005. Land and Water Bugs of the British Isles. A reprinted version by Pisces Conservation Ltd.
Web sites
Bantock, T. & Botting, J. 2010. British Bugs website: http://www.britishbugs.org.uk/ accessed April 2012.
http://www.agroatlas.ru/en/content/pests/Eurydema_oleracea/ accessed July 2011.
Images

Fig. 01. Fallen’s Leatherbug Arenocris fallen. Brett Westwood.

Fig. 02. Fallen’s Leatherbug Arenocris fallen. Harry Green

Fig. 03. Masked Hunter Reduvius personatus. Brett Westwood

Fig. 04. Ceraleptus lividus Rosemary Winnall

Fig. 05. Stictopleurus punctatonervosus. Rosemary Winnall

Fig. 06. Stictopleurus punctatonervosus. Rosemary Winnall

Fig. 07. Forget-me-not Bug nymph Sehirus luctuosus. Jane Scott

Fig. 08. Forget-me-not Bug Sehirus luctuosus adult. Kevin McGee

Fig. 09. Trapezonotus (Trapezonotus) arenarius. John Bingham

Fig. 10. Dicranocephalus medius. John Bingham

Fig. 11. Dicranocephalus medius. John Bingham

Fig. 12. Corizus hyoscyami. Pete Smith

Fig. 13. Corizus hyoscyami. Note the hairs. Pete Smith

Fig. 14. Brassica Bug Eurydema oleracea with reddish markings. John Bingham

Fig. 15. Brassica Bug Eurydema oleracea with pale yellow markings. Harry Green

Fig. 16. Sloe Bug Dolycoris baccarum, Gorse Bug Piezodorus lituratus, and Green Shieldbug Palomena prasina. Jane Scott

Fig. 17. Green shield bug Palomena prasina. Harry Green

Fig. 18. Tortoise Shieldbug Eurygaster testudinaria. Harry Green

Fig. 19. Tortoise Shieldbug Eurygaster testudinaria. Harry Green

Fig. 20. Heather Bug Rhacognathus punctatus. Brett Westwood

Fig. 21. Juniper Shieldbug Cyphostethus tristriatus. Gary Farmer

Fig. 22. Parent Bug Elasmucha grisea final instar larvae and some newly moulted into adults. Harry Green

Fig. 23. Picromerus bidens feeding on Eurygaster testudinaria. Gary Farmer.

Fig. 24. Pied Shieldbug Tritomegas bicolour. Harry Green

Fig. 25. Rhyparochromus pini. Brett Westwood

Fig. 26. Woundwort bug Eysarcoris fabricii. Harry Green

Fig. 27. Blue Bug Zicrona caerulea. John Bingham
Worcestershire Record | 32 (April 2012) page: 7-11 | Worcestershire Biological Records Centre & Worcestershire Recorders
Crayfish of Wyre Forest – an update
Ann Hill
Introduction
In 2010 a baseline study of the native white-clawed crayfish in two tributaries of Dowles Brook, Wyre Forest was undertaken (Hill 2010a & b). In 2011 the baseline study was repeated and expanded to include a study of the distribution of the species in Dowles Brook and all its associated tributaries that flow within the Wyre Forest.
Methods
The 2011 survey work was undertaken between 3rd August and 4th October following published guidelines and best practice (Peay 2002). A qualitative appraisal of likely white-clawed crayfish habitat (and preliminary daytime safety check) was undertaken of all forty-three waterbodies within the stream network of the Wyre Forest. Night-time torching (using a 15-minute timed search) was undertaken of twenty waterbodies that the habitat appraisal found to have the suitable combination of habitat and flow conditions to support crayfish. A standard daytime manual search was made of all those waterbodies where evidence of white-clawed crayfish was found during the night-time torching survey. A standard daytime manual search was also undertaken of five waterbodies where doubt remained as to the presence of white-clawed crayfish. Baited traps replaced a daytime manual search in those waterbodies where the water was >0.5 m deep. Additionally, the 2010 baseline survey of Bell Brook and Forest Lodge Stream was repeated using the same team of surveyors, the same number and location of sample stretches and the same amount of survey effort.
A total of twenty night-time torching surveys (fifty-six 15-minute timed searches); six trapping sessions (105 baited traps); and ten manual standard surveys (370 refugia searched) were undertaken. The study sampled twenty-three percent of the available channel length (17.2 km from a total channel length of 74.6 km). It was not possible to sample the entire length of every watercourse but by using the recognised survey protocol it was ensured that a representative sample was undertaken. No constraints regarding climatic conditions were identified during the survey and there were no periods of increasing or high river flow.
Results
The 2011 survey effort produced a total of 850 white-clawed crayfish records and 119 American signal crayfish records. Please note that the aim of the study was to map the distribution of white-clawed crayfish and therefore, less survey effort was spent on surveying watercourses found to have non-native crayfish populations and hence lower numbers of non-native crayfish are to be expected. A summary of the records from all the surveys is given in Table 1.
Table 1: Crayfish recorded within Dowles Brook and associated tributaries, Wyre Forest, Worcestershire/Shropshire during August to October 2011 using different survey methods.
| Watercourse | Date | Method | Records | |
| Native Crayfish | Signal Crayfish | |||
| Baveney Brook | 16/08/2011 | Torching | 0 | 6 |
| Bell Brook | 12/08/2011 | Casual | 17 | 0 |
| 15/08/2011 | Casual | 37 | 0 | |
| 24/08/2011 | Standard | 27 | 0 | |
| 19/09/2011 | Torching | 4 | 0 | |
| 31/08/2011 | Torching | 588 | 0 | |
| Bell Brook Pool | 31/08/2011 | Torching | 4 | 0 |
| Doghanging Stream | 15/09/2011 | Standard | 0 | 9 |
| 03/08/2011 | Torching | 0 | 93 | |
| Dowles Brook | 20/08/2011 | Torching | 0 | 2 |
| 15/09/2011 | Casual | 0 | 1 | |
| 02-03/09/11 | Trap | 0 | 3 | |
| 03-04/09/11 | Trap | 0 | 1 | |
| Forest Lodge Stream | 24/08/2011 | Rescue | 46 | 0 |
| 25/08/2011 | Torching | 59 | 0 | |
| 01/09/2011 | Torching | 27 | 0 | |
| Kingswood Stream | 27/08/2011 | Standard | 13 | 0 |
| 23/08/2011 | Torching | 19 | 0 | |
| Lodge Coppice Stream | 17/08/2011 | Torching | 0 | 2 |
| Longdon Stream | 30/08/2011 | Standard | 1 | 0 |
| 30/08/2011 | Torching | 8 | 0 | |
| Mad Brook | 22/08/2011 | Torching | 0 | 2 |
White-clawed crayfish were recorded in four watercourses: Bell Brook (including Bell Brook Pool), Forest Lodge Stream, Kingswood Stream and Longdon Stream, within the downstream tributaries of Dowles Brook, in the east of the Wyre Forest study area, (Fig. 1).
American signal crayfish were detected in five watercourses: Baveney Brook, Doghanging Stream, Dowles Brook, Lodge Coppice Stream and Mad Brook, within the west of the Wyre Forest. Above Furnace Mill, the upstream limit of detection of American signal crayfish was at the confluence of Lodge Coppice Stream and Baveney Brook. The downstream limit of detection of American signal crayfish was where the forestry road crosses Dowles Brook, near to the Mercian Way signpost. Suitable crayfish habitat was present but crayfish were absent or undetected in ten waterbodies: Doghanging Tributary 2, Dowles Tributary 1, Far Forest Stream, Gladder Brook, Lem Brook, Neenshill Stream, Park Brook, River Rea, Tibbet’s Stream and Withybed Stream.
The relative abundance of crayfish in an individual watercourse was calculated as the number of crayfish caught per ten refugia searched (Catch per Unit Effort, CPUE) and were compared with CPUE categories determined from standard surveys undertaken nationally (Peay 2002).
Bell Brook: A CPUE of 5.4 which equates to a very high population of white-clawed crayfish (i.e. very high >5 CPUE, Peay 2002).
Kingswood Stream: A CPUE of 2.17 which equates to a moderate population of white-clawed crayfish (i.e. moderate >=1, <3 CPUE, Peay 2002). Longdon Stream: A CPUE of 0.14 which equates to a low population of white-clawed crayfish (i.e. low >0, <1 CPUE, Peay 2002). No comparison could be made with the numbers of white-clawed crayfish in Forest Lodge Stream because the standard manual day-time survey became a rescue operation to move stranded white-clawed crayfish (and fish) to pools within the same tributary. However, it was likely from the numbers recued in the day-time survey (46 individuals) that the stream would be classed as having a high population (i.e. >=3, <=5 CPUE, Peay 2002) of white-clawed crayfish.
The white-clawed crayfish carapace length (CL) of individuals caught and measured ranged between 8.0 mm and 39.1 mm in Bell Brook, 10.8 mm and 40.2 mm in Forest Lodge Stream and between 12 mm and 35 mm in Kingswood Stream. A juvenile is taken as size <25 mm CL. There were too few observations (<30 observations) to comment on population status in Kingswood Stream and no white-clawed crayfish were measured in Longdon Stream (all escapees). However, analysis of the carapace size distribution (Figs. 2.) indicates that there was a:
Stable population of white-clawed crayfish in Bell Brook (skewness -0.074).
Recruiting population of white-clawed crayfish in Forest Lodge Stream (skewness +0.212).
Figs. 2a, 2b, 2c: Histograms showing size distribution (carapace length) of white-clawed crayfish population in three tributaries of Dowles Brook, Wyre Forest, Worcestershire/Shropshire.
Monitoring Study
There was a non-significant increase in the number of observations of white-clawed crayfish in Bell Brook in 2011 compared with observation made in 2010 (Kruskal-Wallis test, sig. = 0.609, p = n.s., n = 32), (Figs. 3.).
There was a statistically significant increase in white-clawed crayfish observations from the night-time torching in Forest Lodge Stream in 2011 compared with observation made in 2010 (Kruskal-Wallis test, sig. = 0.034, p = 0.05., n = 16). However, the increase in observations may be as a result of low water conditions concentrating the population into a smaller area. Absence of water and low flow conditions prevented replication of the 2010 daytime standard survey in Forest Lodge Stream in 2011. Instead stranded dying crayfish (and fish) were rescued and transferred to small in-stream pools.
Fig. 3a & 3b: Comparison of white-clawed crayfish records observed in Bell Brook and Forest Lodge Tributary, Wyre Forest in 2010 and 2011 using standard manual method and night torching.
The relative abundance of crayfish (CPUE) increased from “high” population abundance in 2010 to “very high” population abundance of white-clawed crayfish in Bell Brook in 2011, Table 2. The relative abundance of white-clawed crayfish in Forest Lodge Stream was “low” population in 2010 and as discussed previously in this article, the population abundance would now be classed as a “high”.
Table 2: Comparison of relative abundance of white-clawed crayfish recorded per unit searched between 2010 and 2011 in Bell Brook and Forest Lodge Stream, Wyre Forest.
| Watercourse | Catch Per Unit Effort (CPUE) | |||
| 2010 | 2011 | |||
| number per15 mins | number per10 refuges | number per15 mins | number per10 refuges | |
| Bell Brook | 42.73 | 3.2 | 85.77 | 5.4 |
| Forest Lodge Stream | 4.59 | 0.4 | 13.15 | n/a |
There is no significant change in carapace lengths of white-clawed crayfish measured between 2010 and 2011 in either Bell Brook (Kruskal-Wallis test, sig. = 0.852, n.s., n=58) or Forest Lodge Stream (Kruskal-Wallis test, sig. = 0.536, n.s., n=48), Figs. 4a and 4b: Comparison of white-clawed crayfish carapace length between 2010 and 2011 in Bell Brook and Forest Lodge Stream, Wyre Forest.
Interestingly, more males than females were recorded in Forest Lodge Stream in both 2010 and 2011 and more females than males were recorded in Bell Brook in 2010 and 2011, Table 3.
Table 3: Ratio of male and female white-clawed crayfish records over two recording years (2010 and 2011) from two tributaries of Dowles Brook, Wyre Forest. Key: M = Male; F= Female.
| Watercourse | Male: Female Ratio | |
| 2010 | 2011 | |
| Bell Brook | 2M:8F (1:4) | 19M:33F (1:1.7) |
| Forest Lodge Stream | 1M:0F (n/a) | 29M:14F (1:0.5) |
Discussion
White-clawed crayfish is the only native species of crayfish (Figs. 5 & 6) in the UK and numbers have declined severely over recent years. Despite conservation efforts across Europe this species became recognised as endangered by the International Union of Conservation of Nature in October 2010 (Füreder et. al. 2010). Historically, white-clawed crayfish were widely distributed in the catchment of the Dowles Brook, in both the main river and the tributaries, until the population was thought to be lost following the outbreak of crayfish plague in 1988.
The 2011 survey has established that the Dowles Brook catchment supports at least four discrete populations of white-clawed crayfish and found that Bell Brook has a very high and stable population of white-clawed crayfish whilst Forest Lodge Stream (a more seasonal and ephemeral stream) has a high and recruiting population. The moderate population in Kingswood Stream is possibly a re-emergence of a relict population: the reasons for the re-emergence are unknown. There are no known historical records for Longdon Stream, so this is a significant new record of a white-clawed crayfish population for the Dowles Brook catchment. However, it is unknown whether the low population in Longdon Stream is a decreasing population, a colonising population or a population suffering from some environmental factor that is limiting population growth.
The main threat to the native white-clawed crayfish in Wyre is the non-native American signal crayfish and the associated crayfish plague. Expansion of the American signal crayfish populations in the catchment is predicted and the substantial physical barriers (i.e. culverts with a large vertical face) across the lower reaches of Bell Brook, Forest Lodge Stream, Kingswood Stream and Longdon Stream play a significant role in conservation action for the native species by preventing upstream invasion by non-native crayfish (or at least by significantly delaying it). It is critical to the safeguarding of white-clawed crayfish population in Wyre that measures are taken for the upkeep, maintenance and monitoring of these barriers.
Other pressures to the native population are from water quality (especially in Kingswood Stream) and to a lesser extent climate change (particularly in Forest Lodge Stream). All crayfish populations are vulnerable to issues regarding water quality and are particularly susceptible to acute pollution incidents caused by spills of organic material with a high biochemical oxygen demand, such as cattle slurry or silage. Water quantity is also a crucial criterion affecting the viability of the crayfish, with prolonged drought having the potential to decimate populations. In the dry summer of 2011 flow conditions in all the sampled watercourses were low, with parts of banks and bed exposed and a reduced width of wetted channel in many areas. During the survey, it is highly likely that the flow in Bell Brook was augmented sufficiently by water leaks from around the retaining walls of the upper pools which prevented the watercourse from drying out. Similarly water seepage around the retaining walls of the upper pools in Forest Lodge Stream assisted water flow. However, flow was insufficient to prevent Forest Lodge Stream drying out in places until only shallow pools were left. Low water levels can also increase crayfish vulnerability to predation and evidence of crayfish (both native and alien species) dismembered by otters (or mink) was found along Dowles Brook and Bell Brook.
On a more positive note, the isolation of the individual native crayfish populations from each other gives each discrete population a degree of protection from any risk of crayfish plague or some unexpected event such as a pollution incident. However, the isolation, as well as giving protection also makes the individual local populations vulnerable to extinction and could eventually lead to the decline of the species.
As a result of the last two years of survey work, records and knowledge of native and non-native crayfish distribution in Wyre are increasing but there are still gaps in data. It is encouraging news that a healthy population of native crayfish is present in Wyre but it is only by the continued monitoring of the Wyre crayfish populations that information on long-term variation will gradually be obtained. We plan to continue to survey and monitor the crayfish populations of Wyre (and Worcestershire) where resources and access permits. The high number of native crayfish and the new population records are both exciting and important to our understanding of the distribution of the native crayfish in Wyre and are fundamental to any research on what can be done to conserve the white-clawed crayfish in Wyre.
White-clawed Crayfish are omnivorous as illustrated in Fig. 7. A piece of apple accidentally dropped into a stream promptly became a meal!
References
Hill, A., 2010a. Atlantic Stream Crayfish in Wyre. In: Winnall, R. (Ed.) Wyre Forest Study Group Review 2010.
Hill, A. 2010b. Atlantic Stream (white-clawed) crayfish Austropotamobius pallipes in Wyre Forest. Worcestershire Record 29:29-31
Füreder, L., Gherardi, F., Holdich, D., Reynolds, J., Sibley, P. & Souty-Grosset, C. 2010. Austropotamobius pallipes. In: IUCN 2011. IUCN Red List of Threatened Species. Version 2011.2. <www.iucnredlist.org>. Downloaded on 18 January 2012.
Linck, K., 2000. Crayfish Survey. A survey of watercourses within Worcestershire. Unpublished report for Worcestershire Wildlife Trust.
Peay, S., 2002. A Standardised Survey and Monitoring Protocol for the White-clawed Crayfish in the UK. Life in UK Rivers LIF 02-11-37. EC LIFE Programme, DG Env.D.1., Brussels, Belgium.
Acknowledgements
We are grateful to the Wyre Forest Study Group and Forestry Commission who provided local knowledge and access details, the landowners for access permissions and the many volunteers who assisted with the survey work. Funding was generously provided through Grow with Wyre, the Heritage Lottery funded Landscape Partnership Scheme.
Images
Fig. 1 Location of native and non-native crayfish populations in Dowles Brook and tributaries
Fig. 2a. Histogram showing size distribution (carapace length) of white-clawed crayfish Bell Brook population
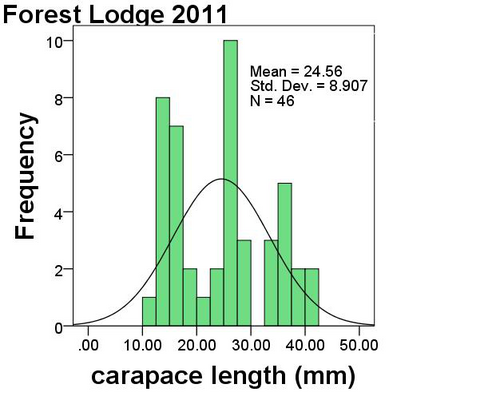
Fig. 2b. Histogram showing size distribution (carapace length) of white-clawed crayfish Forest Lodge population
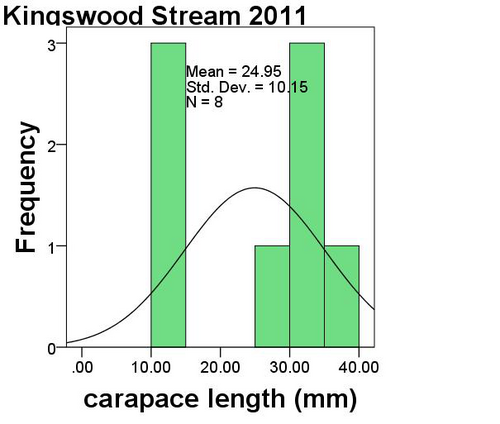
Fig. 2c. Histogram showing size distribution (carapace length) of white-clawed crayfish Kingswood Stream population
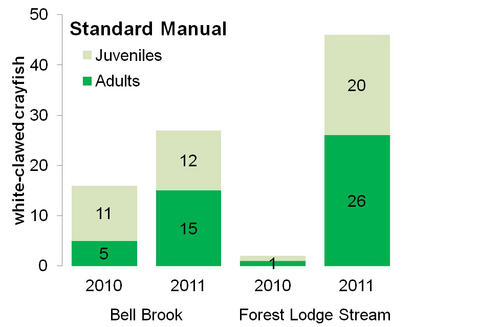
Fig. 3a. White-clawed crayfish records observed in Bell Brook and Forest Lodge Tributary using the standard method.
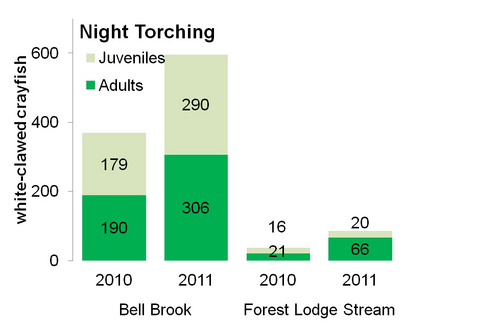
Fig. 3b. White-clawed crayfish records observed in Bell Brook and Forest Lodge Tributary using the night torching method
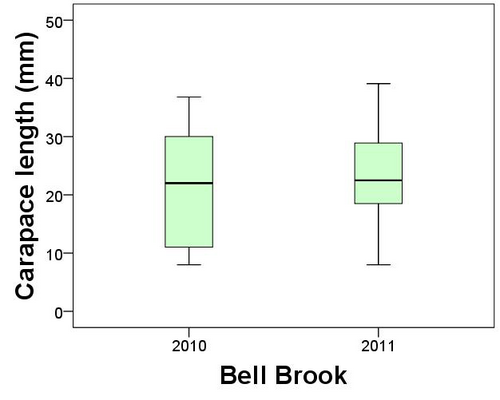
Fig. 4a. Comparison of white-clawed crayfish carapace length between 2010 and 2011 in Bell Brook.
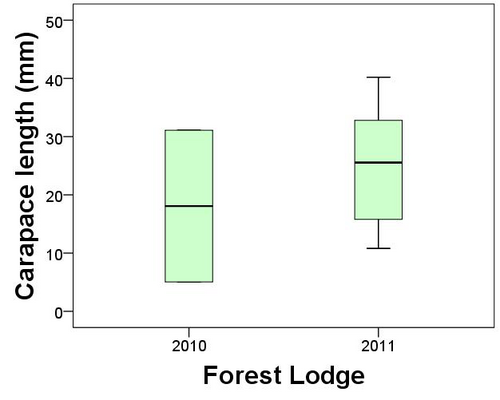
Fig. 4b. Comparison of white-clawed crayfish carapace length between 2010 and 2011 in Forest Lodge Stream.

Fig. 5. Adult White-clawed crayfish. ©Rosemary Winnall.

Fig. 6. Juvenile White-clawed rayfish after capture waiting measurement. ©Rosemary Winnall.

Fig. 7. White-clawed crayfish eating dropped apple. ©Graham Hill
Worcestershire Record | 32 (April 2012) page: 6 | Worcestershire Biological Records Centre & Worcestershire Recorders
Female Blackbird taking adult Poplar Hawkmoth in Worcestershire
Graham Knight
(Little Comberton, Pershore, Worcestershire WR10 3EW)
The 29th July 2011 was a somewhat overcast but warm dry day in the village of Little Comberton near Pershore in Worcestershire (SO94, 28m O.D.). Having reason to examine the inside of a taxi parked in the village at 10.00 hrs BST, I observed a large hawkmoth clinging to the inside of the door frame. From its generally mid-grey colouring and reddish hind wing spots it was identified as a Poplar Hawkmoth Laothoe populi L. which is a localised widely-scattered species throughout the area.
While I was actively discussing the moth with a group of people a female Blackbird Turdus merula L. suddenly appeared and, walking between us, immediately took the moth in its beak and proceeded to walk away with it. Dropped once, the moth was retrieved and finally removed from the scene by the Blackbird which presumably consumed its soft parts.
Many recent summers in Worcestershire have been characterised by drought, of which the summer of 2011 was a more extreme example. This is well-known to have altered the habits and distribution of summer Blackbirds in the area. The few that remained became, in terms of food resources, more opportunistic or more specialised in locating and detecting less usual invertebrate prey. This included regular entry into outbuildings and greenhouses to search for spiders and entrapped flies. Although attest to the wide range of food taken by Blackbirds, this observation, reflecting the boldness and enterprise of one individual, seems worthy of publication.
Reference
Snow, D.W. & Perrins, C.M. 1998. The Birds of the western Palearctic Volume 2. Concise edition. Oxford University Press
Worcestershire Record | 32 (April 2012) page: 6 | Worcestershire Biological Records Centre & Worcestershire Recorders
Worcestershire Record | 32 (April 2012) page: 5 | Worcestershire Biological Records Centre & Worcestershire Recorders
Using a Custom Spell-Checker in Excel and Word
John Partridge
john.partridge@blueyonder.co.uk
Most computer users will be aware that Word (and most other word-processors) have spell-checkers – Word’s is also usable from Excel, and it is possible to make your own list of words, so that your articles for Worcestershire Record and your Excel spreadsheets for Worcestershire Biological Records Centre (WBRC) can have those Scientific Names checked for accuracy.
Pressing F7 brings up the spellchecker, and you can add words to the custom dictionary from here – but it will take you a long time.
The method that I have tried is to produce a list of all the species that have been recorded in Worcestershire, using Recorder and selecting the Preferred Names. Since the spell checker will not check the full Scientific Name, since it is usually two words, I split the names at the space between Species and Genus. (Using Data>Text to Columns), and then got just one of each using Data>Filter>Advanced Filter, checking Unique Values.
A single list is then put together which contains all the used Genera and all the used Species – surprisingly only around 17000 words. This list is then produced as a .txt list, and copied and pasted into the file custom.dic
But first you have to find it. On my computer a search reveals it at:
“C:\Documents and Settings\user1\Application Data\Microsoft\Proof\custom.dic”
Your new spell-checker can be used on your Excel sheet by highlighting all the species list, and pressing F7. If a mismatch is found, there are alternative suggestions made for replacement values – but whether these are useful depends on the extent of your mis-spelling.
Unfortunately, it will not be much use if you have paired up the wrong Genus & Species, because it will pass both of them, and it will not tell you if you have used castanea instead of castaneum or castaneus, for they are all in the dictionary.
A possible way round would be to make all the names into one word e.g. Misumena_vatia, both for the dictionary, and in your Excel spreadsheet. Unfortunately this seems to produce error messages because some of the entries are more than 65 characters long. Perhaps someone can come with another alternative.
If anyone would like a copy of my custom.dic, I will happily supply it.
Worcestershire Record | 32 (April 2012) page: 5 | Worcestershire Biological Records Centre & Worcestershire Recorders
Worcestershire Record | 32 (April 2012) page: 21 | Worcestershire Biological Records Centre & Worcestershire Recorders
Vascular Plant Records – Update February 2012
Bert Reid
In the last Worcestershire Record my article on vascular plant records included a record for Greater Broomrape Orobanche rapum-genistae at Birchfield, Upper Rochford (Fig. 1.). John Day, Bert Reid and Brett Westwood were shown as the recorders but unfortunately I omitted to mention that the plants were initially spotted by John Bingham, who drew them to the attention of at least seven recorders including the three listed. I apologise for my careless error. John Bingham’s sharp eyes gave all who attended the field meeting the opportunity to enjoy the sight of this rare plant.
In the same article, I also floated the idea of setting up an e-mail group to co-ordinate botanical recording in Worcestershire. I asked anyone who wanted to join in and help to e-mail me at bert_reid@talk21.com. Only two people replied to the invitation, so I repeat it here. You don’t have to be an expert to join in. If you can recognise a few easy plants and make occasional lists for your local area you will create useful data that I can use within both national and local projects. But I need more people if we are to set up specific botany field meetings, training days and other activities. If you are unsure what we might do, or whether your contribution would be of any use, please contact me. I will be attending all the general field meetings this year, and will be pleased to see any of you there.
Recording in winter is often ignored by botanists, with most people content to wait for the full flush of spring flowers before venturing out in the field. This is unfortunate because some plant recording is often rewarding in the winter months. Some plants, like Mistletoe Viscum album, Winter Heliotrope Petasites fragrans and Winter Aconite Eranthis hyemalis can be easier to spot before other vegetation comes into leaf. Another advantage is that the restricted range of vegetation enables the recorder to look more closely at the vegetative characteristics of what is around and to practice their skills of identifying plants when they are not in flower. Two examples, both from February 2012, show what can be seen in the ‘dead’ months.
The first example comes from Wyre Forest where John Bingham found about a dozen small clumps of Floating Club-rush Eleogiton fluitans in a small pool backwater in Hitterhill Coppice. This native plant is scarce in Worcestershire, only known six tetrads since 1987 and only three believed to be still extant. This is only the second in the Wyre Forest area (John was also responsible the other Wyre record in 2008, about 0.5km from the new record). Many aquatic plants are available in the vegetative state throughout the year, and although some are very difficult to identify, others are much more straightforward, so they should not be ignored.
The second example is very different, being a non-native plant in full flower in February. The author was seeing what he could find in the early months of the year when he saw abundant well-naturalised Snowdrops Galanthus nivalis growing in Strensham Churchyard. The great majority of the plants were typical examples of Snowdrops, with narrow, glaucous, more or less flat leaves, but a few clumps looked very different, with broad, shiny green leaves standing out clearly from the majority of the plants (Figs. 2 & 3.). Although I have seen a few non-native Snowdrop species in Worcestershire, these churchyard plants were completely new to me. Checking through the literature led me to think that the characteristics of the plants were consistent with Green Snowdrop Galanthus woronowii native to the Aegean. This had never been recorded in Worcestershire, so I sent photos of the plant to Dr Aaron Davis at Kew, who confirmed the identification, saying “You are spot on: G. woronowii. This does naturalize in the UK (I’ve also seen it in Holland) but it is rather infrequent/rare.”
So don’t think that the winter months are a waste of time for the botanist. You too can find a new site for a rare native or a new county record for a naturalised introduction.
Images

Fig. 1. Greater Broomrape Orobanche rapum-genistae at Birchfield, Upper Rochford. John Bingham.

Fig. 2. Green Snowdrop Galanthus woronowii in Strensham Churchyard, February 2012.Bert Reid.

Fig. 3. Green Snowdrop Galanthus woronowii in Strensham Churchyard, February 2012.Bert Reid
Worcestershire Record | 32 (April 2012) page: 33-39 | Worcestershire Biological Records Centre & Worcestershire Recorders
The autecology of the Noble Chafer Gnorimus nobilis within Worcestershire
Jenni Schenke
[A summary of a project undertaken for MSc in Countryside Management, Manchester Metropolitan University]1. Introduction
The Noble Chafer (Fig. 1.) is a relatively large and distinctive metallic green beetle which I am sure many Worcestershire Record readers will be aware of thanks to the numerous appeals for sightings made to readers over the years from Harry Green (‘An edition of Worcestershire Record without Noble Chafer is almost unthinkable!’ (April 2007). However, just in case you would like a quick reminder or you are a new reader I thought I should include some brief background information on the species.
Noble Chafers are saproxylic (dependent upon dead/decaying timber) as larvae, feeding upon the wood mould of trees (Alexander, 2008a; Mann, 2006). Fortunately this makes searching for the species relatively easy as larvae produce distinctive faecal pellets known as frass which can be surveyed for all year around. Usually the elusive beetles develop after two years and if you are lucky you can find them nectaring upon pale/white flowers such as hogweed and meadowsweet on hot summer days (although interestingly this year Simon Wood discovered one on a plum trunk on a rather grey June day).
The species is considered to be an old forest relic (Alexander, 2002b; Whitehead, 2000) having moved over time from pioneer birch and willow, to oak trees, to cultivated fruit trees within traditional orchards which it has been proposed may be a response to climate change (Whitehead, 2003). Unfortunately the shift towards cultivated fruit trees may have been a bad move for the Noble Chafer as their saproxylic nature means that they require the dead/decaying wood associated with mature, well-spaced, open-grown trees. Changes in farming practices have meant that cultivating such trees is no longer economically viable and many traditional orchards have been grubbed up, replaced with dwarf/bush trees or destroyed by development. The situation is such that traditional orchards are now one of the rarest Biodiversity Action Plan (BAP) priority habitats). I t is therefore not surprising to find that the English range of Noble Chafers has declined significantly and that considerable concern exists for the survival of the species.
The main population stronghold has become largely confined to the Severn Basin (Herefordshire, Worcestershire and Gloucestershire) (Figs. 2. & 3.) with Worcestershire appearing to be a particular hotspot. Outlying populations exist in south Oxfordshire, Kent and interestingly the New Forest where the beetle has been found outside of traditional orchards (maps available on NBN Gateway). The Vale of Evesham and Wyre Forest are thought to contain two of the population centres within Worcestershire, with the Teme Valley area also appearing to be a favoured area (this has become more apparent recently since the Teme Valley Orchards Project) (Fig. 4.). There is potential for the Teme Valley and Wyre Forest areas to form one large centre, but further survey work is required to determine this.
Conservation of the Noble Chafer is important both locally and nationally (Worcestershire/UK Biodiversity Action Plan (BAP) species, Red Data Book 2), and also within most of the rest of its European range where it is largely considered to be of conservation concern. With its distinctive appearance, ease of survey and conservation importance it is not surprising that the Noble Chafer has become somewhat of an icon for conservation of the traditional orchards it inhabits and other species associated with such habitats.
My study aimed to build upon the annual PTES survey work, which have reinforced concern for the species’ survival (Green, 2004), and find out more about the habitat preferences of the Noble Chafer in the hope that findings could be used to direct future research and conservation. In order to achieve this, comments made by May (1994) that ecological studies must be pursued at many different levels i.e. spatial scales in order to understand what is going on ecologically, were taken on board and formed the basis of the study. Therefore environmental variables were investigated at host-plant, site, and landscape scales in an effort to discover factors and scales influential on Noble Chafer distribution.
2. Methods
All known existing records of Noble Chafer within Worcestershire were mapped and overlaid across the national traditional orchards habitat layer created by PTES so that potential survey sites could be short-listed.
The River Severn naturally dissects Worcestershire and was used to form two study areas; east and west. Ten short-listed sites were selected from each area for survey, including a variety of different orchards i.e. those with sparse and multiple records, isolated and well-connected orchards, private and protected sites (Fig. 5., Table 1).
Tree and orchard characteristics data were collected in the field while landscape scale data was obtained using Mapinfo GIS. Evidence of Noble Chafer was determined using the accepted method of searching for larval frass using a long handled spoon. This method is considered much less damaging to the larval pabulum than searching for larvae (Smith, 2003; Whitehead, 2003) and is much easier to identify than the larvae themselves (Alexander 2004b; Hammond & Hine 1999). Also frass can be found throughout the year unlike the elusive adult beetles which are rarely observed (Alexander 2002b; Mann 2006; Whitehead 2003).
The data was analysed using various statistical tests including Independent samples t-tests, Chi-square, Regression tree analysis, Oneway Analysis of Variance and the G-test of independence.
| Name | Grid Ref | Size(ha) | Description |
| East Area | |||
| Churchill Orchard | SO922532 | 0.46 | Mature plum orchard; many trees dead and fallen |
| Cleeve Prior Community Orchard | SP076488 | 1.29 | Managed by Cleeve Prior Heritage Trust; apple & plum |
| Earl’s Lane Orchard | SP082492 | 1.47 | Managed by Vale Landscape Heritage Trust; apple & plum |
| Field Barn Orchard | SP075488 | 0.32 | Plum orchard adjacent to Cleeve Prior Community Orchard |
| Hampton Community Orchard | SP020421 | 0.85 | Managed by Vale Landscape Heritage Trust; plum |
| Mill Pond Avon Farm | SO895423 | 0.51 | Derelict and overgrown apple orchard |
| New Farm Orchard | SO905470 | 1.38 | Mixed orchard |
| Phepson Farm | SO942599 | 0.14 | Derelict damson orchard, isolated noble chafer site |
| Tiddesley Wood | SO930461 | 0.62 | Wildlife Trust reserve; plum and damson near to ancient woodland |
| Yessell Lane Orchard | SP010448 | 7.55 | Soon to be in Higher Level Stewardship; large plum |
| West Area | |||
| Alfrick Pound; orchard 2 | SO743525 | 0.78 | Plum orchard with very high density of noble chafer |
| Birchenhall Farm | SO750515 | 2.89 | Large cherry orchard close to woodland reserves |
| Bowcastle Farm; A | SO769750 | 1.65 | Cherry orchard on bank within SSSI |
| Bowcastle Farm; C | SO766753 | 1.96 | Mixed orchard within SSSI adjacent to woodland |
| Cherry Orchard, Blissgate | SO746732 | 0.86 | Cherry orchard on steep bank next to SSSI |
| Huntlands Farm | SO723561 | 2.42 | Apple & plum orchard next to Leigh Brook |
| Knapp & Papermill | SO748520 | 1.65 | Worcestershire Wildlife Trust reserve; apple |
| Knightwick Manor; 2 | SO723549 | 1.67 | Cherry & plum orchard within cluster of old orchards |
| Lowe Farm; A | SO702682 | 2.02 | Apple orchard (close to orchard B) |
| Lowe Farm; B | SO704683 | 1.47 | Plum orchard on steep slope |
Table 1. Summary of orchard sites surveyed
3. Results – summary of significant findings (Table 2)
| Variable |
Significant |
Comments |
| Host Plant Scale | ||
| Girth (at breast height) |
Yes |
Apple & plum trees with NC evidence had significantly greater girths than those without. Cherry trees exhibited the opposite |
| Species |
Yes |
The effect of tree species appeared to be reflected across several host plant variables |
| Proximity to next tree |
Yes |
Damson trees with NC evidence were closer to nearest fruit tree than those without |
| Proximity to next NC tree |
No |
Possible indicator of dispersal ability. NC can fly >700m to nectar but can also spend its life within a tree without ever leaving it |
| Tree density within 10m |
Yes |
NC presence was positively associated with tree density |
| Tree health (alive/dead/hollowing etc.) |
Yes |
Cherry trees with NC evidence had higher % dead branches |
| Rot hole (size/orientation etc.) |
No |
Weak associations for higher holes & South/West orientations. Recording temp. within rot holes may have been more useful |
| Height |
Yes |
Cherry trees with NC evidence were shorter than those without |
| Location within orchard |
No |
Thought to influence micro-climate & sun-exposure. Weak +ve association with bottom of slopes (possibly more sheltered) |
| Site Scale | ||
| Area |
Yes |
Smaller orchards had a positive association with high NC tree density |
| Tree density |
Yes |
Orchards with highest fruit tree density had highest densities of NC trees |
| Dominant tree type |
Yes |
Stone fruits were positively associated with high NC tree density |
| Average tree girth |
No |
Not a good indicator of tree age/dead wood availability as high density NC orchards were mostly composed of plums/damsons which are much smaller than apples & cherries. More indicative of fruit species composition |
| Aspect, Elevation & Topography |
No |
Thought to influence shelter/sun-exposure but anthropogenic nature of orchards may override the effects of such variables e.g. orchards were probably originally planted in warmer more sheltered parts of people’s land and therefore such variables would be very similar across visited sites. May have been more significant difference if sites without NC were also included in study. |
| Orchard management |
No |
Boundary, vegetation, grassland & tree management thought to influence various environmental factors mostly related to micro-climate, availability of food supplies & nectar sources. Possible indication that these factors may not be as influential at site-scale. |
| Adjacent habitat |
Yes/No |
Sites located next to known NC orchards had significantly higher NC tree densities. Other habitat types had less of an effect |
| Landscape Scale | ||
| Proximity to ancient woodland |
No |
Thought to be important due to NC’s historical association with woodland. However recording past and present woodland areas may have been more relevant |
| Location in relation to River Severn |
No |
Predicted would have impact on habitat preferences of NC due to different geographical factors e.g. climate, local distinctiveness. Also proposed as a dispersal barrier by Alexander, 2005, this may not be the case in Worcestershire. |
| Surrounding traditional orchard density |
No |
Recorded as an indicator of habitat connectivity, but data probably not representative as only orchards were included whereas other habitats may provide suitable connecting habitats i.e. rides in woodland may provide nectaring sites |
| Surrounding NC orchard density |
Yes |
Low density NC orchards had larger areas of NC orchards within 2.5km radius than those with intermediate NC tree density |
Table 1 – Summary table of variables investigated, their significance and brief explanation of results.
4. Discussion of significant findings
4.1 Host-plant scale
Trunk girth
Trunk girth was a significant characteristic in all but the damson trees recorded (damson were the least frequently recorded fruit species and had more been sampled the result may have become significant). These findings are comparable with other studies where increased trunk girth was to be positively related to saproxylic invertebrate presence and diversity (Alexander 2002; Grove,2002; Lindhe & Lindelöw 2004; Lush et. al. 2009; Oleksa 2009; Ranius 2002; Ranius & Jansson 2000).
Trunk girth is a key factor influencing Noble Chafers as girth increases as the tree ages, therefore there is likely to be more deadwood, fungal decay and rot holes in larger girthed trees (Alexander 2008b; Green 2004; Ranius 2002) (providing they are not hollow). Larger girthed trunks will also have a relatively smaller surface area compared to smaller counterparts and so in theory will be better at retaining heat and providing a more stable environment retaining heat and better moisture conditions suitable for larval development (Alexander, 2008b; Begon et. al., 1990; Schiegg, 2001; Ranius & Jansson, 2000; Schiegg, 2001) for the ‘warmth-loving’ Noble Chafer (Alexander, 2008b).
The cherry tree result is interesting and requires further investigation as Noble Chafer trees had significantly smaller girths. Out of all the trees surveyed, the cherries were by far the tallest and largest and displayed the ‘healthiest’ appearance (solid trunks, fewer rot-holes). It may be that cherry trees rot differently and rot holes may occur higher up in the branches which were not searched. Bark thickness can also be a limiting factor for saproxylic species (Abrahamsson & Lindbladh, 2006; Buse et. al., 2007; Hedgren, 2007) and with bark thickness and trunk diameter being highly correlated it is possible that smaller cherry trees may be more penetrable.
Tree species
Noble Chafer evidence was only recorded in four fruit species all belonging to either Prunus or Malus genus thus agreeing with Whitehead’s comment in 2003 that ‘[Noble Chafer] seem to prefer senile Prunus & Malus species’. The narrow range of host species recorded and relatively strong preference for plum trees indicates a rather specialised nature, typical of saproxylic species (Alexander 2008a, Alexander 2004a; Bouget et. al. 2008b; Dodelin 2008). However, this survey was restricted to orchards and no other fruit species were recorded (it is possible that Worcs contains a relatively high proportion of plum trees).
Preferences for certain host species may be explained by differences in microhabitat dynamics (Dubois et. al. 2009b; Ranius 2002) i.e. the amount of wood mould, aspect of rot-holes (Ranius 2002; Ranius & Nilsson 1997) which may be the result of natural characteristics i.e. growth speed, wood quality; or management e.g. pruning (Dubois et. al. 2009b, Lachat et. al. 2006). Therefore investigating tree characteristics e.g. bark depth, rot-hole temperature/moisture conditions within different regions of the species’ UK and European range may provide more answers explaining host tree preferences and the geographic differences that occur (i.e. traditional orchards within Britain (with the exception of the New Forest) and a wider range of broadleaved species on the continent).
Proximity to other fruit trees
This was recorded as an indicator of micro-climate with the assumption that the more widely spaced trees would receive the greatest amount of sun-exposure (Dubois et. al.,2009b; Green 2004) and would be more likely to provide a warmer larval pabulum, aiding survival and development of larvae (Alexander 2002b; Green 2004; Whitehead, 2003). Sun-exposure of the host wood substrate is known to positively influence saproxylic invertebrate presence and can be of more importance than trunk diameter (Buse et. al. 2007; Djupström et. al.,2008; Dubois et. al. 2009b; Lindhe & Lindelöw 2004; Lindhe et. al. 2005; Ranius & Nilsson 1997).
Interestingly the opposite association to that expected was apparent where trees without Noble Chafer evidence had the most open space around them. Occupied trees were also closer to the nearest tree than unoccupied trees (except apple which showed a very weak opposite trend). These results suggest that Noble Chafer preferred semi-exposed conditions similar to findings by Jonsell et. al. 2004, Ranius 2000 and Ranius 2002. Such conditions may be more suitable for fungal activity and rot-hole development (Bouget & Duelli 2004; Lindhe et. al. 2004). The smaller size of fruit trees (as opposed to larger broadleaves often included in saproxylic studies) may mean that some shading helps to stabilise internal trunk conditions. Another factor to consider is that orchards are planted to maximise fruit productivity with full canopy growth therefore trunks may still receive relatively high amounts of sun-exposure even if other trees are nearby. They are also often planted in sheltered, sunny or south-west facing areas (Wedge 2007b) and sometimes pruned (Green 2004). Alternative methodology i.e. insolation index based on sunshine intensity, measuring evaporation (Buse et. al. 2007; Lindhe & Lindelöw 2004) may have provided better comparison of micro-climate.
Proximity to other Noble Chafer trees
Distance to nearest Noble Chafer tree was not significantly different between occupied and unoccupied trees for any tree species. This may suggest that Noble Chafers are good dispersers and is comparable to the findings by Gibb et. al. 2006a who found that specific red-listed forest beetles had lower wing-loadings and superior flying ability than more common species. The fact that Noble Chafers have been observed nectaring and copulating on flowers up to 700metres away from known orchard sites (Alexander 2004b; Whitehead 2003) indicates that they have the ability to disperse relatively far away to find suitable host trees.
However, Noble Chafers are generally only observed flying when temperatures are really warm (Alexander 2002b; Mann 2006; Whitehead 2003) and it is proposed that they can spend their whole life within one tree without leaving it at all (Whitehead 2003). This also appears to fit with the findings that distance to nearest host tree is not significant.
It is therefore difficult to establish the scale at which metapopulations exist i.e. site scale or landscape scale (Krauss et. al., 2005). Ranius, 2006 appeared to have the same difficulty in his study of O. eremita. He proposed that the extremely low dispersal rate between trees could mean that each tree hosted a local population, making the stand of trees a metapopulation. He then later suggested that metapopulation dynamics may also arise at a larger scale, with each stand of hollow trees sustaining a local population and the individuals in an entire landscape forming a metapopulation. Perhaps, as proposed by Krauss et. al. 2004 in their study of the Small Blue Butterfly Cupido minimus Fuessly, the majority of Noble Chafers are sedentary and only a few adults disperse widely, helping to maintain genetic diversity within local populations (Harrison 1993; Ranius 2006). Dispersal is a key characteristic in species conservation and major contributor to metapopulation viability (Dubois et. al. 2009a) therefore further investigation into the dispersal ability of this elusive beetle is required.
In contrast to this study’s findings Buse et. al. 2007,Grove, 2002 and Ranius 2006 all found distance to nearest occupied tree to be a significant host plant-scale factor. However Buse et. al. 2007 classified distance to next nearest host plant as a landscape-scale factor as opposed to host plant-scale which demonstrates the difference between orchard habitats which in theory have higher microhabitat densities (i.e. more potential host trees with more rot-holes) than isolated oak trees (Dubois et. al. 2009b).
Tree condition
Findings indicate that Noble Chafer trees had higher percentages of dead branches than those without Noble Chafer evidence, although this difference was only significant between cherry trees (and very nearly apple trees). These findings are comparable with studies by Buse et. al. 2007 and Lachat et. al.,2006 where the proportion of dead branches was found to be a positive indicator of saproxylic invertebrates. Nordèn et. al. 2004 also recognised the importance of attached dead wood but recommended that further investigation of its biodiversity value was required. Proposed reasons for this positive association include greater sun-exposure to the trunk with higher numbers of dead branches giving less shading from foliage (Green, 2004), and more opportunities (i.e. rot-holes) to enter the tree (Alexander, 2004b).
Tree height
The effect of host-plant height on invertebrates has been well documented; however there has been less study of the effect of tree height on saproxylic species. In this study only cherry trees showed a significant difference in height and similarly to other factors investigated (girth, rot-hole height and distance to nearest occupied tree) exhibited the opposite trend to the other fruit species i.e. shorter trees were preferred. This may be because cherries were the largest of the fruit trees investigated and so it is possible that shorter trees are preferred as they represent the weaker, more accessible specimens (i.e. reduced bark thickness).
4.2 Site Scale
Fruit tree density and orchard area
Orchards with the highest fruit tree densities had significantly higher densities of Noble Chafer trees, a finding which is frequently found in invertebrate studies (Krauss et. al. 2005; Krauss et. al. 2004; Müller et. al.in press) because a greater food resource will support more individuals (Yee et. al., 2007).
These findings may again reflect the importance of tree species at site scale (Dubois et. al. 2009b; Oleksa et. al. 2007) as orchards dominated by plum had the highest fruit tree densities. This may also indicate a preference for warmer, more stable microclimates as higher densities of fruit trees may provide shelter against wind and retain heat thus speeding up larval development (Green 2004).
Habitat patch size is frequently strongly and positively correlated with host-plant density (Krauss et. al. 2005) and invertebrate density with larger patches providing more resources and having lower extinction risks (Krauss et. al. 2004; Ranius 2002; Sahlin & Schroeder 2010; Tikkanen et. al. 2009). However, in comparison this study found that smaller orchards had the highest Noble Chafer densities. This finding may again be linked to fruit tree composition as plum trees, (the preferred species) are much smaller than apple and cherry trees meaning a greater number can be planted in a smaller area.
Whitehead (2003) noted the resilience of the Noble Chafer and its ability to form small-scale populations in restricted areas of acceptable habitat. Therefore it is possible Noble Chafers can live in high densities where habitat quality is optimal even if the sites are relatively small. However if this is the case then these sites will need to have good links with other favourable habitats (Kindlmann et. al. 2005; Krauss et. al. 2005; Webb & Thomas 1994), a number of dispersive individuals (Dubois et. al. 2009a; Kindlmann et. al. 2005; Krauss et. al. 2004) or a continued supply of mature fruit trees (Alexander,2008b; Lush et. al. 2009; Webb et. al. 2010) otherwise the populations will eventually collapse.
Adjacent habitats
Adjacent habitats were recorded because they are believed to have an important microclimatic role on the sites invertebrates inhabit (Dubois et. al. 2009b; Dubois & Vignon 2008; Lindbladh et. al. 2003; Lindhe et. al2005; Ranius & Nilsson,1997). The strong positive association between adjacent orchards and the relatively strong negative association with woodlands could be seen to support the microclimate theory even though they exhibit opposite trends. This is because although both would provide shelter, the more widely spaced trees in orchards would also allow the sun to penetrate thus creating more suitable conditions (Dubois et. al. 2009b; Green 2004) i.e. warmer rot holes to aid larval maturation (Alexander 2002b; Green 2004; Whitehead 2003), warmer air temperatures to aid dispersal (Dubois et. al. 2009a).
The positive association with adjacent orchard habitats could also be linked to the increased the patch size and reduced edge effects (Johansson et. al. 2007; Johnson et. al. 2002).
It is also possible the negative woodland association supports the theory that adjacent habitats affects dispersal efficiency as the tall, dense surrounding vegetation creates an obstacle for movement (Dubois et. al, 2009b; Dubois & Vignon 2008; Jeanneret et. al, 2003; Kindlmann et. al. 2005).
4.3 Landscape Scale
Density of Noble Chafer orchards in surrounding area
It is well known theory that the distribution and continuity of suitable habitats is a limiting factor for invertebrate populations and that the most well-connected sites support the greatest populations due to factors such as high rates of immigration and low extinction risk (Krauss et. al. 2005; Lush et. al. 2009; Ranius 2002; Sahlin & Schroeder 2010; Sverdrup-Thygeson et. al. 2010; Webb,2006; Webb & Thomas 1994).
Low density orchards had the greatest areas of Noble Chafer orchards at both scales; the effect being more pronounced at the larger scale (2.5km) (analysis of surrounding traditional orchards produced similar patterns, but were not significant). This result is interesting and may indicate the negative effect of habitat fragmentation whereby more isolated sites become more densely populated because only a small proportion of the population is able to disperse to more distant orchards (although this proportion may be greater than that of low density orchards) thus supporting some of the metapopulation theories mentioned earlier and highlighting the importance of the species’ dispersal ability.
The importance of dispersal ability for determining the scale at which metapopulation dynamics such as connectivity becomes relevant has been raised by a number of studies (Krauss et. al. 2004; Krauss et. al. 2005; Ranius, 2002). Findings by other studies suggest that the Noble Chafer’s dispersal ability could be relatively good due to the beetle’s relatively large size (Webb et. al. 2006) and distribution within fragmented habitats (Merckx & Dyck 2007). Therefore further investigation into the Noble Chafer’s dispersal ability may provide useful information.
However, it is also important to consider that occurrence patterns may also be related to historical habitat distribution i.e. before agricultural intensification when orchards were less fragmented and isolated (Lush et. al. 2010; Ranius 2002; Sahlin & Schroeder 2010). Therefore the landscape matrix may have become more inhospitable during this time with fewer corridors and stepping stones (Krauss et. al.,2005; Tikkanen et. al. 2009; Webb 2006) as Alexander (2004a; 2005b) believed to be the case in Gloucestershire. Further analysis including the comparison of historical OS maps with present orchard distribution may provide information to support/reject this theory.
Also the species distribution data used in this study is limited to the holdings of Noble Chafer records by Worcestershire Biological Records Centre at the time of the study. Therefore some positive orchards may have been missed because they had not been surveyed or because records had not been supplied to WBRC. Although it is difficult to conclusively determine negative Noble Chafer orchards/trees, more robust testing of this theory would require the survey of every orchard in both radii around sites to determine both presence/absence as well as Noble Chafer density. Potential nectaring sites should also be included as they can be important breeding sites (Alexander 2004b; Whitehead 2003) and may also provide important stepping stones between apparently isolated orchards.
5. Summary
Factors affecting Noble Chafer distribution occurred across all spatial scales with host-plant appearing to be the most influential (trunk girth being particularly important). I t is proposed that this may reinforce the highly specialised nature of saproxylic species and importance of the dead wood provided by host-plants, although biases within the study meant this cannot be decisively concluded. The findings do demonstrate the complex nature of interactions that exist between Noble Chafers and traditional orchards, and the interconnectedness of factors between scales. Finding out more about the dispersal ability of Noble Chafer and investigating a greater range of landscape-scale variables is essential for informing long-term conservation management. In the mean time ensuring that the landscape provides a mosaic of well-connected traditional orchards and nectaring sites is important.
6. Recommendations
Further research
Developing a more reliable non-invasive method to determine presence/absence of Noble Chafer would be extremely useful. Deborah Harvey’s current study (part funded by PTES) on nitrogen fixation of the Stag Beetle, Noble Chafer and Variable Chafer may provide some leads for this kind of development in the future.
Recording tree characteristics in more detail i.e. rot-hole temperature, moisture could help to explain host-plant preferences.
Surveys which determine both presence/absence and assess habitat suitability for Noble Chafer (using Alexander s 2002a grading system) should continue and the data should be mapped using GIS to enable large scale analysis.
Mapping other important habitats e.g. nectaring sites, historical orchards may also produce some interesting results and help to inform long-term conservation management i.e. identifying linkages/obstacles between sites (Alexander, 2002a), and priorities for conservation i.e. isolated orchards.
Finding out more about the dispersal ability of Noble Chafer will also be essential, but could prove rather challenging due to the elusive nature of the beetle!
Noble Chafer conservation
Apply a landscape-scale approach maintaining a mosaic if well connected habitats (Alexander,2002a; Webb et. al.,2010; Webb 2006; Wedge 2007c) e.g. creating rides through woodlands separating orchards.
Increase the availability of mature fruit trees in the surrounding landscape by establishing replanting programmes, especially within known Noble Chafer orchards (Lush et. al. 2009).
Provide continued encouragement for landowners to participate in agri-environment schemes so that existing orchard habitats are protected and enhanced (Alexander, 2002a). Hopefully the inclusion of traditional orchards as a priority in the BAP Habitat Action Plan will help to ensure that this happens (Lush et. al. 2009).
Hopefully conservation action applied across a range of scales as suggested should not only help to conserve the Noble Chafer, but also numerous other important wildlife species which have come to depend on such habitats.
Acknowledgements
Thank you to everyone who helped me during my project.
References
Abrahamsson, M. and Lindbladh, M. 2006. A comparison of saproxylic beetle occurrence between man-made high- and low-stumps of spruce (Picea abies). Forest Ecology and Management. 226 (1-3):230-237.
Alexander, K. N. A. 2002a. The Noble Chafer Gnorimus nobilis in Worcestershire – a report on the 2002 survey. Unpublished report for The People’s Trust for Endangered Species.
Alexander, K. N. A. 2002b. The invertebrates of living and decaying timber in Britain and Ireland – a provisional annotated checklist. Research Report No. 467, English Nature, Peterborough,.
Alexander, K. N. A. 2004a. The Noble Chafer Gnorimus nobilis in Gloucestershire – a report on the 2004 survey. Unpublished report for People’s Trust for Endangered Species.
Alexander, K.N.A. 2004b. Revision of the Index of Ecological Continuity as used for saproxylic beetles. Research Report No. 574. English Nature Peterborough.
Alexander, K. N. A. 2008. Tree biology and saproxylic Coleoptera: Issues of definitions and conservation language. In: Vignon V.,and Asmodé J.-F. (Eds). Proceedings of the 4th Symposium on the Conservation and Workshop of Saproxylic Beetles 27-29th June 2006. p. 1-6. Vivoin Sarthe Department, France.
Begon, M., Harper., J.L. and Townsend, C.R. 1990. Ecology Individuals, Populations and Communities (2nd ed.). Blackwell Scientific Publications, Oxford
Bouget, C., Brustel, H. and Zagatti, P. 2008b. The French Information System on Saproxylic Beetle Ecology (FRISBEE): an ecological and taxonomical database to help with the assessment of forest conservation status. In: Vignon V., Asmodé J.-F. (Eds). Proceedings of the 4th Symposium on the Conservation and Workshop of Saproxylic Beetles 27-29th June 2006. p. 25-28. Vivoin Sarthe Department, France.
Bouget, C. and Duelli, P. 2004. The effects of windthrow on forest insect communities: a literature review. Biological Conservation 118:281–299.
Buse, J., Schröder, B., and Assmann, T. 2007 Modelling habitat and spatial distribution of an endangered longhorn beetle – A case study for saproxylic insect conservation. Biological Conservation. 137 (3):372-381.
Djupström, L.B., Weslein, J. and Schroeder, L.M. (2008) Dead wood and saproxylic beetles in set-aside and non set-aside forests in a boreal region. Forest Ecology and Management 255 (8-9):3340-3350
Dodelin, B. 2008 Aspects of the repartition of the saproxylic beetles in forests (French Alps). In: Vignon V., Asmodé J.-F. (Eds). Proceedings of the 4th Symposium on the Conservation and Workshop of Saproxylic Beetles. p. 47-52. Vivoin Sarthe Department, France
Dubois, G. and Vignon, V. 2008 First results of radio-tracking of Osmoderma eremita (Coleoptera: Cetoniidae) in French chestnut orchards. In: Vignon V., Asmodé J.-F. (Eds). Proceedings of the 4th Symposium on the Conservation and Workshop of Saproxylic Beetles 27-29th June 2006. p. 123-130. Vivoin Sarthe Department, France
Dubois, G.F., Vernon, P. and Brustel, H. 2009a. A flight mill for large beetles such as Osmoderma eremite (Coleoptera: Cetoniidae). In: Buse, J., Alexander, K.N.A., Ranius, T. and Assman, T. (Eds). Saproxylic beetles: their role and diversity in European woodland and tree habitats – proceedings of the 5th symposium and workshop 14-16th June 2008. p. 219-224. Leuphana University, Institute of Ecology and Environment, Luneburg, Germany.
Dubois, G.F., Vignon, V., Delettre, Y.R., Rantier, Y., Vernon, P. and Burel, F. 2009b. Factors affecting the occurrence of the endangered saproxylic beetle Osderma eremita (Scopoli, 1763) (Coleoptera: Cetoniidae) in an agricultural landscape. Landscape and Urban Planning 91 (3):152-159.
Gibb, H., Hjältén, J., Ball, J.P., Pettersson, R.B., Landin, J. and Alvini, O. 2006a. Wing loading and habitat selection in forest beetles: are red-listed species poorer dispersers or more habitat-specific than common congenerics? Biological Conservation 132:250–260
Green, G.H. 2004. The noble chafer Gnorimus nobilis – Records needed! Worcestershire Record 16:10-11
Grove, S.J. 2002. Tree basal area and dead wood as surrogate indicators of saproxylic insect faunal integrity: a case study from the Australian lowland tropics. Ecological Indicators. 1 (3):171-188.
Hammond, P.M. and Hine, S.J. 1999. Coleoptera; the beetles. In: Barnard, P.C. (Eds.) Identifying British Insects and Arachnids An annotated bibliography of key works. Cambridge University Press, Cambridge. p. 80-138
Harrison, S. 1993. Metapopulations and conservation. In: Edwards, P.J., May, R.M. and Webb, N.R. (Eds.) Large-Scale Ecology and Conservation Biology. Blackwell Scientific Publications, Oxford p. 111-128
Hedgren, P.O. 2007. Early arriving saproxylic beetles (Coleoptera) and low and high stumps of Norway spruce. Forest Ecology and Management. 241 (1-3):155-161
Jeanneret, Ph., Schϋpbach, B. and Luka, H. 2003. Quantifying the impact of landscape and habitat features on biodiversity of cultivated landscapes. Agriculture Ecosystems and Environment 98:311-320
Johansson, T., Hjältén, J., Gibb, H., Hilszczanski, J., Stenlid, J., Ball, J.P., Alinvi, O. and Danell, K. 2007. Variable response of different functional groups of saproxylic beetles to substrate manipulation and forest management; Implications for conservation strategies. Forest Ecology and Management. 424 (2-3):496-510(2)
Johnson, C. M., Johnson, L.B., Richards, C. and Beasley, V. 2002. Predicting the Occurrence of Amphibians: An Assessment of Multiple-scale Models. In: Scott, M.J., Heglund, P.J., Morrison, M.L., Raphael, M.G., Wall, W.A. and Samson, F.B. (eds.). Predicting species occurrences: issues of accuracy and scale. Island Press. Washington. p.157-170
Jonsell, M., Nittèrus, K. and Stighäll, K. 2004. Saproxylic beetles in natural and man-made deciduous high stumps retained for conservation. Biological Conservation 118 (2):163-173
Kindlmann, P., Aviron, S. and Burel, F. 2005. When is landscape matrix important for determining animal fluxes between resource patches? Ecological Complexity 2 (2):150-158
Krauss, J., Steffan-Dewenter, I., Mϋller, C.B. and Tscharntke. 2005. Relative importance of resource quantity, isolation and habitat quality for landscape distribution of a monophagous butterfly. Ecography. 28 (4):465-474
Krauss, J., Steffan-Dewenter, I. and Tschanrntke, T. 2004. Landscape occupancy and local population size depends on host plant distributions in the butterfly Cupido minimus. Biological Conservation 120 (3):355, 361.
Lachat, T., Nagel, P. Cakpo, Y., Attignon, S., Goergen, G., Sinsin, B. and Peveling, R. 2006. Dead wood and saproxylic beetle assemblages in a semi-deciduous forest in Southern Benin. Forest Ecology and Management 225 (1-3):27-38.
Lindhe, A. and Lindelöw, Å. 2004. Cut high stumps of spruce, birch, aspen and oak as breeding substrates for saproxylic beetles. Forest Ecology and Management. 203:1-20.
Lindhe, A., Åsenblad, N. and Toresson, H-G. 2004. Cut logs and high stumps fo spruce, birch, aspen and oak – nine years of saproxylic fungi succession. Biological Conservation 119 (4):443-454.
Lindhe, A., Lindelöw, Å. and Åsenblad, N. 2005. Saproxylic beetles in standing dead wood density in relation to substrate sun-exposure and diameter. Biodiversity and Conservation. 14 (12):3033–3053.
Lush, M., Robertson, H.J., Alexander, K.N.A., Giavarini, V., Hewins, E., Mellings, J.,Stevenson, C.R., Storey, M. and Whitehead, P.F., 2009. Biodiversity studies of six traditional orchards in England. Natural England ,Peterborough. Natural Engand Research Report 025.
Mann, D.J. 2006. The Beetle Families Scarabaedeoidea. In: Cooter, J. and Barclay, M.V.L. (Eds.) A Coleopterist’s Handbook .4th ed. The Amateur Entomologists’ Society, Kent p.47-58.
Merckx, T. and Dyck, H. V. 2007. Habitat fragmentation affects habitat-finding ability of the speckled wood butterfly, Pararge aegeria L. Animal Behaviour 74 (4):1029-1037.
Müller, J., Noss, R.F., Bussler, H. and Brandl, R. (in press) Learning from a “benign neglect strategy” in a national park: Response of saproxylic beetles to dead wood accumulation. Biological Conservation.
Nordèn, B., Götmark, F., Tönnberg, M. and Ryberg, M. 2004. Dead wood in semi-natural temperate broadleaved woodland: contribution of coarse and fine dead wood , attached dead wood and stumps. Forest Ecology and Management 194 (1-3):235-248.
Oleksa, A. 2009. Conservation and ecology of the hermit beetle Osmoderma eremita s.l. in Poland. In: Buse, J., Alexander, K.N.A., Ranius, T. and Assman, T. (Eds). Saproxylic beetles: their role and diversity in European woodland and tree habitats – proceedings of the 5th symposium and workshop 14-16th June 2008. p. 177-188. Leuphana University, Institute of Ecology and Environment, Luneburg, Germany.
Oleksa, A., Ulrich, W. and Gawroński, R. 2007. Host tree preferences of hermit beetles Osmoderma eremita Scop., Coleoptera: Scarabaeidae in a network of rural avenues in Poland. Polish Journal of Ecology 55 (2):315-323.
Ranius, T. (2002) Influence of stand size and quality of tree hollows on saproxylic beetles in Sweden. Biological Conservation 103 (1):85-91
Ranius, T. and Jansson, N. 2000. The influence of forest regrowth, original canopy cover and tree size on saproxylic beetles associated with old oaks. Biological Conservation 95:85-94.
Ranius, T. and Nilsson, S.G. (997. Habitat of Osmoderma eremita Scop (Coleoptera: Scarabaeidae), a beetle living in hollow trees. Journal of Insect Conservation 1, p. 193-204
Sahlin, E. and Schroeder, L.M. (2010) Importance of habitat patch size for occupancy and density of aspen-associated saproxylic beetles. Biodiversity and Conservation 19:1325-1339
Schiegg, K. 2001. Saproxylic insect diversity of beech: limbs are richer than trunks. Forest Ecology and Management 149 (1-3):295-304.
Smith, M. 2003. Noble Chafer Gnorimus nobilis Wyre Forest District, Worcestershire 2003 Survey Worcestershire. Unpublished report for People’s Trust for Endangered Species
Sverdrup-Thygeson, A., Skarpass, O. and Odegaard, F. 2010. Hollow oaks and beetle conservation: the significance of the surroundings. Biodiversity and Conservation 19 (3):837-852.
Tikkanen, O., Punttila, P. and Heikkila, R. 2009. Species-area relationships of red-listed species
Webb, J.R. 2006. Conservation. In: Cooter, J. and Barclay, M.V.L. (Eds.) A Coleopterist’s Handbook. 4th ed. The Amateur Entomologists’ Society, Kent p.47-58.
Webb, J.R., Drewitt, A.L., Measures, G.H. 2010. Managing for species: Integrating the needs of England’s priority species into habitat management. Part 1 Report. Peterborough: English Nature, NERR024.
Webb, N.R. and Thomas, J.A.1994. Conserving insect habitats in heathland biotopes: a question of scale. In: Edwards, P.J., May, R.M. and Webb, N.R. (Eds.) Large-Scale Ecology and Conservation Biology. Blackwell Scientific Publications, Oxford p. 129-151.
Wedge, C. 2007b. Traditional orchards: site and tree selection. Natural England, Bristol.
Wedge, C. 2007c. Traditional orchards: a summary. Natural England, Bristol.
Whitehead, P.F. 2003. The noble chafer Aleurostictus nobilis (L., 1758) (Col., Scarabaeidae) in Britain. In: People’s Trust for Endangered Species. Proceedings of the second pan-European conference on Saproxylic Beetlesp.17-31. People’s Trust for Endangered Species, London.
Yee, D.A. and Juliano, S.A. 2007. Abundance matters: a field experiment testing the more individuals hypothesis for richness – productivity relationships. Oecologia 153 (1):153-162.
Images

Fig. 1. Fig. 1. The Noble Chafer Gnorimus nobilis. ©Harry Green
Fig. 2. Pre 1980 records of Noble Chafer Gnorimus nobilis in the UK. (data provided by PTES March 2010)
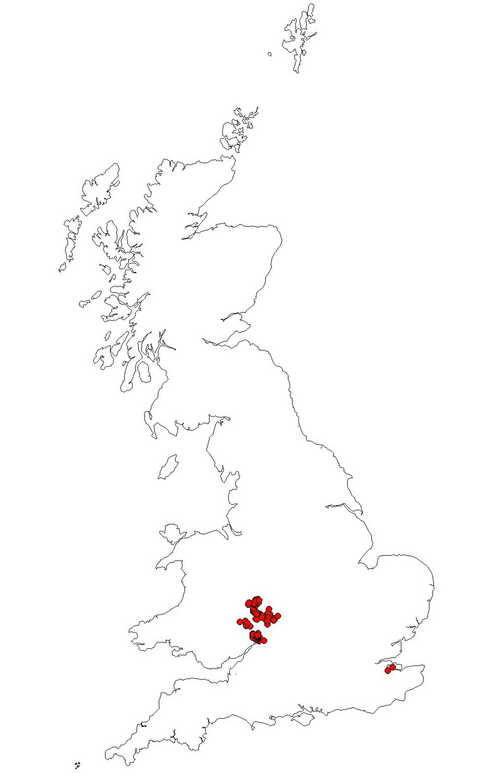
Fig. 3. Post 1980 records of Noble Chafer Gnorimus nobilis within the UK. (data provided by PTES March 2010)
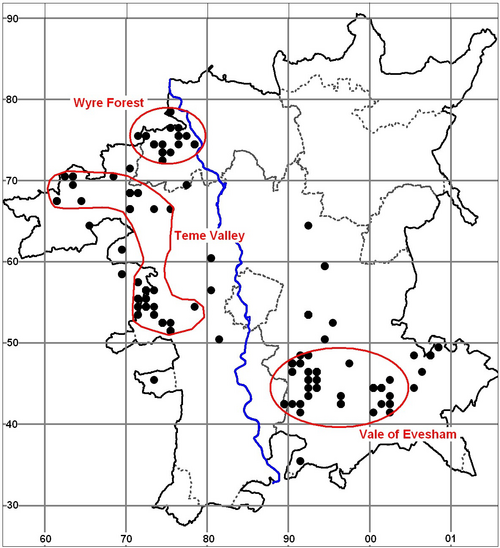
Fig. 4. Noble Chafer Gnorimus nobilis distribution in Worcestershire – all records. (1901-2011) in relation to the River Severn (records provided by Worcestershire Biological Records Centre 2011)
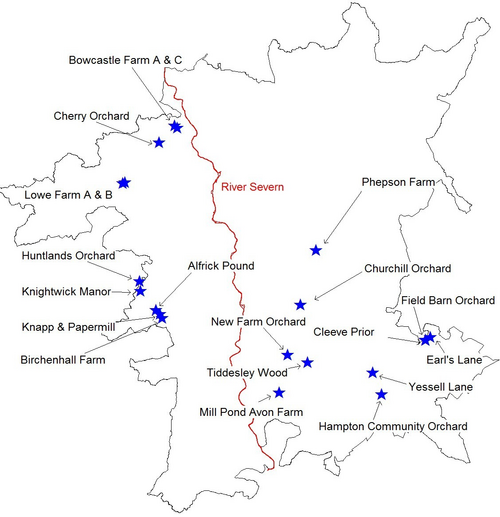
Fig. 5. Orchards surveyed in Worcestershire for this project.
Worcestershire Record | 32 (April 2012) page: 24-25 | Worcestershire Biological Records Centre & Worcestershire Recorders
The National Bechstein’s Bat Survey and Beyond
Jane Sedgeley, James Hitchcock & Johnny Birks
In Worcestershire Record 14:38, April 2011, James Hitchcock reported on the Bat Conservation Trust’s Bechstein’s Bat Project launch in Worcestershire and presented preliminary survey results from 2010. Since then we’ve completed our second survey season and the National Project has come to an end – but with some very exciting results for Worcestershire.
The Project aimed to map the UK distribution of the rare and elusive Bechstein’s bat, Myotis bechsteinii (Fig. 1.); to gather more information about the species’ range and habitat associations; and to identify conservation hotspots. Based on pilot work in south-east England (by Dr David Hill and Frank Greenaway), selected woodlands in southern England and south Wales were surveyed using a specialised trapping technique (Fig. 2.) operated by county based voluntary bat groups. Ten local bat groups took part in the project: North Buckinghamshire, Cornwall, Devon, Dorset, Gloucestershire, Kent, Oxfordshire, Somerset, Surrey and Worcestershire. In each county the targeted woodlands (one per 10×10 km square) were surveyed over two summer seasons.
Overall, between 2009 and 2011, a total of 199 target woodlands were surveyed in which 838 bats of 12 species were caught, identified and released. These included 57 Bechstein’s Bats (29 females and 28 males) at 37 sites. The records collected in Buckinghamshire and Worcestershire are now the most northerly UK records for breeding females of this species (Miller 2011).
In Worcestershire 18 woodlands were surveyed in 2010/2011. Eight Bechstein’s Bats were caught at four different sites. Breeding females and juvenile bats (born that season) were caught at Grafton Wood (owned and managed by Worcestershire Wildlife and Butterfly Conservation), Romers Wood (owned and managed by Herefordshire Nature Trust), and Oakley Wood near Droitwich. A further adult male was caught at a fourth site, but this woodland is not being named as it is in private ownership and has no public access.
Based on this success, the Bat Conservation Trust gave permission to extend the survey to investigate potential breeding hotspots. Could we find breeding female Bechstein’s Bats using other woodlands close to Grafton and Oakley Woods in the old ‘Forest of Feckenham’ area? Yes we could! We surveyed a further five woodlands and found Bechsteins’s at two of them. An adult male and juvenile female were caught at Trench Wood (owned and managed by Worcestershire Wildlife Trust) and an elderly male and a non-breeding female at the privately owned Little Goosehill Wood (no public access).
These additional surveys brought our grand total in Worcestershire to23 woodlands surveyed, 160 bats caught, comprising at least 11 bat species with 12 Bechstein’s Bats found in six different woodlands (Fig. 3.). Not only did the project reveal that Bechstein’s Bats are more widespread in Worcestershire than previously known, it also provided valuable information on a range of other rare or under-recorded woodland bat species frequently overlooked by more conventional survey techniques. For example, we now have 25 new records for Whiskered Bat Myotis mystacinus, a species very under-recorded in the county because its echolocation calls are practically indistinguishable from those of other Myotis species so it can only be indentified with any confidence in the hand.
So what next? Worcestershire Wildlife Trust has secured a grant from the People’s Trust for Endangered Species to work in conjunction with ecologists Johnny Birks and Eric Palmer, in conjunction with the Worcestershire Bat Group, to conduct a radio-tracking project this summer entitled: Bechstein’s Bat – a study on the population size, foraging range and roosting ecology at Grafton Wood SSSI, Worcestershire. The main aims of the project are to build on the findings of the initial Bechstein’s Bat Surveys in order to expand our knowledge of the foraging and roosting patterns of the speciesin managed woodlands.
Management at Grafton Wood is currently very active, with wide rides and 12 acres of coppice providing a good deal of open space among the predominantly closed-canopy. Results from the forthcoming radio-tracking study will be used to produce a report to inform future management decisions at Grafton and other woodlands. The report will offer habitat management options designed to balance current conservation objectives with the desire to establish and maintain the favourable conservation status of Bechstein’s Bat populations.
It’s certainly going to be a very interesting and busy summer. We hope to present our results in a future issue of Worcestershire Record, so watch this space for the next instalment and see how we got on.
Reference
Miller, H. 2011.Bechstein’s Bat survey– final report. Bat Conservation Trust. Downloadable from: http://www.bats.org.uk/data/files/publications/Bechsteins_bat_survey_final_report.pdf(accessed 9th April 2012).
Images

Fig. 1. Bechstein’s Bat Myotis bechsteinii. Jane Sedgeley

Fig. 2. Setting the harp trap used to catch Bechstein’s Bat. Jane Sedgeley.
Fig. 3. Sites in Worcestershire where Bechstein’s Bat has been recorded. Mike Glyde
Worcestershire Record | 32 (April 2012) page: 14-16 | Worcestershire Biological Records Centre & Worcestershire Recorders
New moth species recorded in Worcestershire in 2011
Tony Simpson
(County moth recorder).
2011 was a “bumper year” with 16 new species of moths recorded in Worcestershire (VC37).
There were three new Macro species:
Danny Arnold had a single Double Line Mythimna turca at Upper Rochford on 5/6/11 which might be a new resident or possibly a migrant.
Steve Whitehouse had a Rannoch Looper Itame brunneata of the migrant form at Trench Wood on 14/6/11.
In early July there was an influx of Small Marbled Eublemma parva, a rare migrant which has not been recorded in the county before, with specimens recorded at Abberton by Gavin Peplow, at Bewdley by Ian Machin, and at Stoke Prior by Jeff Rush. Later in October I recorded one at Bransford.
There were thirteen new Micro species:
I found a number of vacated leaf mines of Ectoedemia minimella on Birch in Rock Coppice on 8/10/2011.
I found two larval cases of Coleophora lithargyrinella on Stitchwort Stellaria holostea at Cobb’s Cross on 29/4/2011from which I bred one moth.
Oliver Wadsworth had a Coleophora sternipennella come to light at St. Johns, Worcester: a species which feeds on Atriplex and Chenopodiumspecies.
Oliver also recorded Cosmiotes consortella at Monkwood on 1/4/2011 and at Tiddesley Wood on 16/8/2011.
Patrick Clement caught a specimen of Tuta absoluta at Windmill Hill on 2/7/2011, the first free flying record in the UK of a recently arrived pest species. There had been previous DEFRA records from Tomato greenhouses in the Evesham area in 2010 and it remains a notifiable pest. It has the potential to live on any Solanaceous plant outdoors, such as Woody Nightshade Solanum dulcamara.
Martin Skirrow caught a moth at light on 27/7/11 at Carpenter’s Farm – Pendock later confirmed by me to be Dichomeris alacella, which is a rare species whose larva feeds on lichens on tree trunks, and may have come from nearby orchard trees.
Ian Machin caught a pretty little moth at light in Bewdley on 15/9/11, which Oliver Wadsworth confirmed as Mompha divisella, whose larva makes a stem gall in smaller Willow Herb species.
Mike Dale and Oliver Wadsworth had specimens of Cnephasia communanaat Feckenham Wylde Moor come to light on 17/5/2011 and at Windmill Hill on 24/5/2011. This is either an overlooked or more probably a recently arrived species in the county.
When we were light trapping at Monk Wood on 22/4/11 Patrick Clement caught a worn grey Tortrix which was confirmed as Acleris logiana, and Steve Whitehouse had another at Worcester on 24/12/2011. This Birch feeding species used to be confined to Scotland, but there have been an increasing number of records in the south of England, some in Warwickshire in 2010, and now it has reached us. This probably represents a new wave of immigration from the continent unrelated to the Scottish specimens.
Danny Arnold had an unusual Tortrix come to light at Upper Rochford early in the year, on 14/1/11, which on dissection turned out to be Acleris abietana. This is a moth which first appeared in Scotland in 1965. There were some records from the North of England and then one in Herefordshire in 2008. It feeds on conifers especially Abies species and seems now to be spreading southwards in the UK.
The last three species to reach Worcestershire are all migrants to the United Kingdom:
Danny Arnold had our first European Corn Borer Ostrinia nubilalis at Upper Rochford to light on 1/10/11. This species can be a pest species on Maize in hotter climates and is now resident in South East England feeding on Mugwort Artemesia vulgaris.
Gavin Peplow had a Hymenia recurvalis at Abberton at light on 10/10/11. There was a small influx of this Mediterranean species into the UK this autumn.
Lastly Jeff Rush had a beautiful Palpita vitrealis to light at Stoke Prior on 28/10/11.
This large number of new species were recorded in 2011when despite a hot sunny start, summer turned into a generally cold and miserable season in which the commoner migrant and resident species were in low numbers. This good result reflects the generally increased number of recorders with light traps and the fact that we now have five proficient micromoth recorders in the county. It will be difficult to match these records unless we get another major migration year in 2012.
Images

Fig. 1. Double Line Mythimna turca. Danny Arnold.

Fig, 2. Small marbled moth Eublemma parva. Tony Simpson

Fig. 3. Cosmiotes consortella. Oliver Wadsworth

Fig. 4. Tuta absoluta. Patrick Clement.

Fig. 5. Mompha divisella. Oliver Wadsworth.

Fig. 6. Acleris logiana. Oliver Wadsworth.

Fig. 7. European Corn Borer Ostrinia nubilalis. Danny Arnold.

Fig. 8. Hymenia recurvalis. Gavin Peplow.

Fig. 9. Palpita vitrealis. Oliver Wadsworth
Worcestershire Record | 32 (April 2012) page: 12-13 | Worcestershire Biological Records Centre & Worcestershire Recorders
Close encounters with hornets in 2011
Roger Umpelby
Hornet numbers in south-east Worcestershire around Ashton-under-Hill have fluctuated over the past ten years, but overall they seem to be doing very well. Numbers seemed to dip in 2008 and 2009, but 2010 saw a sharp rise in sightings, and in 2011 they seemed to be everywhere from early spring until late autumn.
Their unjustified reputation for being aggressive is confounded by how easy it was to take some of these close-up photographs. My faith in their relatively placid nature was tested on one occasion when I was asked for advice on what to do about a hornets’ nest under some wooden steps into the back door of a house. The householder, who was obviously a nature-lover, took the advice not to annoy them but to carry on as normal whilst avoiding sudden movements or loud noises close to the nest. I was pleased to hear subsequently, and perhaps a little relieved, that the hornets proved to be model neighbours for the rest of the year.
I was fascinated to see Douglas Gregor’s photograph in ‘Hornet stories’ in the April 2011 Worcestershire Record as, although late in the year hornets frequently feed on ripe fruit and flowers, I had not seen the sap feeding. Then in early October having heard several hornets, I found a young willow tree on which numerous hornets were feeding. They had stripped considerable lengths of bark to release the sap flow, dropping the chewed bark like sawdust. As with Douglas’s picture, several flies were feeding alongside the hornets, but none were attacked while I watched. Earlier in the year in the same location, hornets were often to be seen searching for prey in open grassland. Their searching appeared to be quite systematic, but most prospective prey appeared to either hear or see them approaching and make their escape in good time.
Reference
Gregor, D. 2011. ‘Hornets stripping bark and also catching fly’. A note within ‘Hornet Stories’. Worcestershire Record, 30:33.
Images

Fig. 1. Hornet head on. Roger Umpelby

Fig. 2. Hornets feeding and damage on willow. Roger Umpelby

Fig. 3. Hornet and greenbottle feeding on willow. Roger Umpelby

Fig. 4. Hornet drinking water. Roger Umpelby

Fig. 5. Hornet feeding on ivy pollen and pollinating. Roger Umpelby

Fig. 6. Hornets aggressive – surely not. Roger Umpelby
Worcestershire Record | 32 (April 2012) page: 19 | Worcestershire Biological Records Centre & Worcestershire Recorders
Rooks nesting on pylons
Roger Umpelby
Full house! Rooks nesting on a pylon near Ashton-under-Hill, March 2011. There were about five nests in an oak about 100m and a rookery in trees about 300 m away.
Images

Fig.1. Rookery pylon 8April2012 Ashton under Hill.Roger Umpelby

Fig.2. Rookery pylon nest 5 with occupant 8April2012 Ashton under Hill.Roger Umpelby
Worcestershire Record | 32 (April 2012) page: 23 | Worcestershire Biological Records Centre & Worcestershire Recorders
Two uncommon Ascomycota fungi in Worcestershire, Geopora sumneriana(Cooke) M. Torre. (Cedar Cup) and Helvella leucomelaena (Pers.) Nannf. (White-footed Elfin Cup)
Brett Westwood & John Bingham
A visit to Shavers End quarry on the Abberley Hills on March 11 2012 BW discovered a number of small cup fungi growing on old quarry waste under Scots pine, a species he tentatively identified as Helvella leucomelaena. This identification was later confirmed by John Bingham. The fruit-bodies were dark grey in colour inside about 2-4cm across with a pale grey outer colour with short ribbed stalk. The photograph shows the cups somewhat dry and faded a little.
The fungus is said to grow on sandy soils in woods or heaths but the quarry waste appears to have been a suitable substrate. It is a rare species thinly scattered across southern Britain with apparently just two previous records for Worcestershire from the south of the county. (British Mycological Society Checklist).
On March 21st 2012 following a report by Roger Stickland of unusual fungi, BW visited his garden in Kidderminster in search of Geopora (Sepultaria)sumneriana an uncommon and large cup fungusi that grows under cedar trees. There was a remarkable mass fruiting of over 100 fruit-bodies scattered on the northern side of the tree. The owner reported that the tree was 30 years old and this was the first year it had produced any cup fungi. It was also the first year it had produced cones, whether that is significant remains unknown.
The fungus forms an underground sphere, an ascocarp that breaks the surface to form a crown-shaped cup, pale on the inside with a roughly hairy dark brown outside layer. The cups are 5-7 cm across and up to 5cm tall. Like most cup fungi the flesh is brittle. It is a vernal species associated with cedar trees and very rarely possibly with yew.
Uncommon in Britain and mostly southern south of a line from the Severn to Humber. There are a few records for Worcestershire , most around the Malvern area, no doubt due to the presence of cedar trees in larger gardens and grounds in that area. (BMS Checklist).
Both fungi were reported to John Bingham who supplied further information and visited both sites to obtain the pictures.
References
Sterry, P. & Hughes, B. 2009. Collins Complete Guide to British Mushrooms & Toadstools. Collins
British Mycological Society Checklist of Fungi http://www.fieldmycology.net/GBCHKLST/gbchklst.asp
Images

Fig. 1. Helvella leucomelaena.John Bingham

Fig. 2. Geopora sumneriana.John Bingham
Worcestershire Record | 32 (April 2012) page: 22-23 | Worcestershire Biological Records Centre & Worcestershire Recorders
Horn Stalkball Onygena equina(Willd.) Pers. A recent record for Worcestershire
Brett Westwood
On 29th January 2012 I found an old decomposing cattle horn in willow and alder carr at Hurcott Wood near Kidderminster. On closer examination, many small pin-like fungi with clubbed heads were found growing on the horn. I took a photograph and later sent the image to John Bingham who identified it as Onygena equina, a rare species not often seen and now listed in the Red Data Book Edition 2. as ‘Near Threatened’. A brief description can be found in the Collins New Naturalist book Fungi. (Spooner & Roberts 2005)..
Onygena equina, the Horn Stalkball. is one of a small group of fungi species that grow on keratin found in horn and hooves or sometimes on deer antlers. Another Onygena species grows on feathers. Potentially it can grow on human finger nails, so it needs to be handled and treated with some care!
Quite how long the horn had been in Hurcott Wood or how it got there is a mystery. The horn was so decomposed it had a strong pungent smell possibly due to the fungus or bacteria.
John Bingham’s research revealed that the most recent records for Worcestershire were pre-1909, the date that the Botany of Worcestershirewas published. There Carleton Rea noted two sites for the county, Shrawley Wood and Ombersley, and the species was given as ‘not common’ and “on owl pellets and decayed horse hoof” (Amphett & Rea 1909). No more recent records appear to exist for Worcestershire. Nationally its distribution is local and it is found mainly in the upland areas of the United Kingdom growing on old sheep horns.
Over the years the fungus has declined as farm hygiene and the disposal of dead animals has been improved and controlled by legislation. Dead beasts no longer litter the local woods and I su spect that the fungus is destined to remain a rarity.
References.
Amphett, J. & Rea, C. 1909. The Botany of Worcestershire. Cornish Brothers Ltd.
Spooner, B. & Roberts, P. 2005. Fungi. New Naturalist Series No.96. Collins, London.
Evans, S. (Unpublished, dating about 2008) Preliminary Assessment. The Red Data List of Threatened British Fungi. British Mycological Society.
Images

Fig. 1. Oxygena equina on cow horn. Brett Westwood.

Fig. 2. Onygena equina on cow horn. John Bingham

Fig. 3. Onygena equina on cow horn. John Bingham.
Worcestershire Record | 32 (April 2012) page: 20 | Worcestershire Biological Records Centre & Worcestershire Recorders
Grey squirrels (Sciurus carolinensisGmelin) debarking a mature Coast Redwood (Sequioa sempervirensEndlicher) in Evesham town, Worcestershire during the 2010-2011 winter
P.F. Whitehead
During March 2011 my attention was drawn to the fact that Grey Squirrels Sciurus carolinensis had debarked an ornamental Coast Redwood Sequoia sempervirens of Victorian age in Evesham town, Worcestershire. When I visited the site I observed, quite remarkably, that the outer layers of the outer bark had been removed for the entire height of the twin-stemmed tree (Fig. 1) estimated to be in the order of 16 metres. The bark removals that littered the ground varied from about 3 cms in length to long strips some 30 cms long and up to 2.5 cms thick. Observers noticed that the squirrels were active during February 2011 and were concerned that the tree might in some way be impacted on. Providing there is no repetition that can be dismissed.
I have no previous experience of Grey Squirrels intensively debarking trees in the family Taxodiaceae, certainly not in this area. Normally Grey Squirrels debark trees which are relatively thin-barked or sappy to gain access to nutrients, especially in the phloem. It is well-known that Sycamore Acer pseudoplatanus L. is favoured, especially when the sap is rising in the spring, and that most British Sycamore woodlands are affected. The question remains as to why Grey Squirrels would debark a Coast Redwood tree so intensively when the tannin-rich bark is stringy, fibrous, has no food value, and would scarcely impact on incisor tooth condition.
Coast Redwoods have a highly developed ability to reproduce themselves vegetatively; it is not infrequent to observe mature trees surrounded by basal suckers, which are able to develop into new trees when the parent tree ages. Buried deep in the outer bark of this tree were numerous semi-dormant adventitious buds, some of which had been exposed. It is contended that the squirrels had actively searched for these by removing the bark systematically and then consumed the developing buds with their rich store of nutrients and auxins. Such damage had not been observed at the site before and it seems probable that this urban population was faced with two choices once their range of food stocks was nearing exhaustion in a severe winter. One was to individually disperse into urbanised surroundings with much more limited available food; the other was to stay put and make creative choices in terms of food resource usage. That is my explanation for why this tree was debarked.
The interlocking history of all these non-native organisms warrants comment. The Grey Squirrel was introduced into Britain sometime around 1828 (Fitter, 1959). The Coast Redwood was introduced into Britain in 1846 (Gilbert-Carter, 1936), i.e. about the same time, although the genus was native in the pre-Pleistocene of Europe. Sycamore is likely to have been introduced into Britain in the early medieval period (Whitehead, 2005). This note is, in essence therefore, a tale of man the unwitting miscreant.
References
FITTER, R.S.R. 1959. The ark in our midst. William Collins Sons & Co. Ltd, London.
GILBERT-CARTER, H. 1936. British trees and shrubs. Clarendon Press, Oxford.
WHITEHEAD, P.F., 2005. The Coleoptera of Sycamore (Acer pseudoplatanusL.) with reference to Sooty Bark Disease and Bredon Hill, Worcestershire. Entomologist’s Gazette 56:107-127
Image

(C)PFWHITEHEAD
Fig. 1. Mature twin-stemmed Coast Redwood Sequioa sempervirensdebarked by Grey Squirrels Sciurus carolinensis, Evesham town, Worcestershire, 31 March 2011. Paul Whitehead.
Worcestershire Record | 32 (April 2012) page: 18-19 | Worcestershire Biological Records Centre & Worcestershire Recorders
Ants and Extra-floral Nectaries
Rosemary Winnall
We know that many plants produce nectar within their flowers to attract insect pollinators. However, some plants have extra-floral nectaries (EFNs) which are situated in parts of the plant outside the calyx. These are glands that produce water and sugars but they are not designed to attract and reward pollinators. The nectar produced by EFNs is sweeter than phloem and is made up of a mixture of glucose, fructose, sucrose and some protein, amino acids and organic acids.
These EFNs may be found on leaf margins, leaf axils, petioles, stipules, flower bracts, sepals and flower stalks. They may be conspicuous as raised glands or recessed basins and are sometimes coloured differently than the surrounding plant material.
Ants are often attracted to these to feed, as are occasionally other insects. The presence of the ants helps to protect the plant from browsing and from insects laying eggs on the foliage, and experiments have shown that plants with ants do better than plants without. Ants at nectaries on buds can protect the flower from nectar robbing.
Post-floral nectaries are found after the flower has died back and the fruits or seedpods are developing. The presence of ants in this situation help to protect the developing seeds from being eaten. So far I have seen this on Ivy and Garlic Mustard.
It is well known that ants visit aphids to feed on the honeydew that they excrete. So when ants are seen on plants, it is essential to check what they are doing. Are the ants feeding, and is so, are there aphids present or are nectaries involved?
I first noticed ants on extra-floral nectaries in a greenhouse where many Lasius niger ants were feeding on extra-floral nectaries on nectarines and peach plants. As these plants are Prunus species I quickly checked out the nearest Wild Cherry Prunus avium, and yes, ants were present there too! This stimulated some internet research and I found that although there has been dedicated work done in Florida, little has been recorded about EFNs from the UK. This started me looking more closely and documenting evidence. I photographed the ants feeding, identified the ant and the plant and started to keep a list of what I discovered. This is an ongoing project, but my results so far can be seen in the table 1. It is interesting to note that I have not to date found the common ant Formica fusca feeding in this way.
There are other plants noted for their EFNs, such as Elder Sambucus nigra , Dog’s Mercury Mercurialis perennis, willows Salix sp., Cherry Laurel Prunus laurocerasus, Acacia, Polygonum, Passiflora, Cucurbita, vines, Helianthus, Hibiscus, Robinia. I have yet to check these out. So there is plenty more work to be done in the search for ants using extra-floral nectaries in our gardens and countryside!
Plant |
Lasius niger |
Myrmica rubra |
Myrmica ruginodis |
| Alliaria petiolata, Garlic Mustard | |||
| Centaurea sp. Garden Knapweed | |||
| Fraxinus excelsior, Ash | |||
| Paeonia sp., Peony | |||
| Prunus armeniaca, Apricot | |||
| Prunus avium, Wild Cherry | |||
| Prunus persica, Nectarine | |||
| Pteridium aquilinum, Bracken | |||
| Vibernum opulus, Guelder Rose | |||
| Vicia sativa, Common Vetch | |||
| Vicia sepium, Bush Vetch | |||
| Vicia faba, Broad Bean |
Table 1. Ant species found feeding on extra-floral nectaries.
Images

Fig. 1. Lasius niger on Garlic Mustard. Rosemary Winnall

Fig. 2. Lasius niger on Centaurea. Rosemary Winnall

Fig. 3. Myrmica rubra on Peony. Rosemary Winnall

Fig. 4. Myrmica ruginodis on Wild Cherry. Rosemary Winnall

Fig. 5. Lasius niger on Nectarine. Rosemary Winnall

Fig. 6. Myrmica rubra on bracken. Rosemary Winnall

Fig. 7. Lasius niger on Common Vetch. Rosemary Winnall

Fig. 8. Lasius niger on Broad Bean. Rosemary Winnall
Worcestershire Record | 32 (April 2012) page: 5-6 | Worcestershire Biological Records Centre & Worcestershire Recorders
Mystery deaths of Tawny Owls
Rosemary Winnall
Ever since we erected two Tawny Owl boxes in 2000 on our land at Bliss Gate near Bewdley, we have had a nesting pair in one box or the other during most years.
On 2nd April 2012 my farmer neighbour telephoned me to tell me that there were two dead Tawny Owls lying close together in his field and he could not tell how they had died. He said that they had certainly not been there when he had visited the field five days previously. Although the birds had been moved up off the ground, I could see the feathers on the grass indicating where they had been found originally (see photograph). Examination showed that these had been plucked and not chewed off.
Close examination of the birds indicated that one had lost its head and both had had their breast meat removed (see photograph). In addition there were broken sections taken out of the sternum indicating that a bird of prey had been involved. There was no other damage that I could see and no feathers on the back of the birds appeared to have been disturbed.
I have never known a bird of prey take Tawny Owls before, although Golden Eagles, Goshawks and Buzzards are documented to have done so. We have plenty of Buzzards around and for the last three years a pair has nested in a large oak tree about 200m away from where the dead owls were found.
I have previously written in Worcestershire Record (2004) how I’d recorded Buzzards flying across our valley before sunrise whilst it was still dark, so I know that there is a possibility that these two species could be flying time at the same time. I was interested in the note from Graham and Marie-Anne Martin (2007) of a Buzzard attacking a Barn Owl. But have we any other local records of Tawny Owls being killed by Buzzards? It would be interesting to know!
References
Martin, Graham & Marie-Anne. 2007. What do buzzards eat? Worcestershire Record 21:21-22
Winnall, R. 2004. What do buzzards eat …..! Worcestershire Record 16:8-9
Images

Fig. 1. Site where two dead Tawny Owls were found showing plucked feathers. Rosemary Winnall

Fig. 2. Two dead Tawny Owls. Rosemary Winnall
Worcestershire Record | 32 (April 2012) page: 5 | Worcestershire Biological Records Centre & Worcestershire Recorders
Buzzard Stories
Ravens at crow’s nest previously attacked by buzzards
Craig Wright
On 27th February 2012 Craig Wright of Pershore wrote:
Forgot to say that further to the incident I mentioned to you some time back about the Buzzard making off with a Carrion Crow chick from a nest in the top of my garden Leylandii. I had another incident the other morning just after first light but this time it was Ravens and the Carrion Crows. An almighty racket awoke me from my slumber and as I peered out the window I could see a pair of Ravens perched in the leyandii “gronking” and the two Crows buzzing and diving at them to drive them off, which they eventually succeeded in doing. Can’t imagine the Ravens were checking the tree for their nest site and I don’t think the crows have started nesting yet. Anyway another garden tick none the less!
Reference
Wright, C. 2011. Buzzards and crows. Worcestershire Record 31:51-54 |
Worcestershire Record | 32 (April 2012) page: 5 | Worcestershire Biological Records Centre & Worcestershire Recorders
